What Are Marine Sanctuaries?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Sustainable Seas Expeditions and Recognizes the Capabilities of BACKGROUND INFORMATION Deepworker
INVESTIGATION 3 Planning an Expedition Club-tipped anemone LAURA FRANCIS lzlzlzlzlzlzlzlzlz n this Investigation, students design LEARNING OBJECTIVES I their own scientific expedition to the Students will: sea using the goals of Sustainable Seas • Conduct a simulated vertical transect Expeditions as a guide. They begin by in the sea as one method of doing conducting a simulated vertical transect research; as an example of one method scientists • Plan and design a scientific investiga- tion that meets the goals of use in oceanic research. Sustainable Seas Expeditions and recognizes the capabilities of BACKGROUND INFORMATION DeepWorker. Exploring—For Answers Sustainable Seas Expeditions Research STANDARDS Meet DeepWorker Geography Standard 3 NOAA’s National Marine Sanctuaries How to analyze the spatial organization National Marine Sanctuary Sites of people, places, and environments on the Earth’s surface ACTIVITIES Geography Standard 18 Sample Mission: Vertical Transect How to apply geography to interpret Planning Your SSE Mission the present and plan for the future Science Education Standards Recognizing and developing abilities necessary to design and conduct scientific investigations •47• INVESTIGATION 3 Sample Mission: ACTIVITY Vertical Transect Guiding Question In this activity, students conduct a simulated vertical transect, providing them with an example What is a vertical transect? How do scientists use of one sampling method that they may consider this method to determine the layering system of for their scientific investigations. The data repre- physical and biological parts in the ocean? sents a vertical transect in Monterey Bay, but may be replaced with data from other locations. Discussion Materials Scientists often face the problem of trying to ac- curately interpret a natural system based upon ❑ Dive Mission Cards, one set for the class the limited data they are able to collect in the ❑ Water Sample Cards, one set for the class field. -

The Evolution of Siphonophore Tentilla for Specialized Prey Capture in the Open Ocean
The evolution of siphonophore tentilla for specialized prey capture in the open ocean Alejandro Damian-Serranoa,1, Steven H. D. Haddockb,c, and Casey W. Dunna aDepartment of Ecology and Evolutionary Biology, Yale University, New Haven, CT 06520; bResearch Division, Monterey Bay Aquarium Research Institute, Moss Landing, CA 95039; and cEcology and Evolutionary Biology, University of California, Santa Cruz, CA 95064 Edited by Jeremy B. C. Jackson, American Museum of Natural History, New York, NY, and approved December 11, 2020 (received for review April 7, 2020) Predator specialization has often been considered an evolutionary makes them an ideal system to study the relationships between “dead end” due to the constraints associated with the evolution of functional traits and prey specialization. Like a head of coral, a si- morphological and functional optimizations throughout the organ- phonophore is a colony bearing many feeding polyps (Fig. 1). Each ism. However, in some predators, these changes are localized in sep- feeding polyp has a single tentacle, which branches into a series of arate structures dedicated to prey capture. One of the most extreme tentilla. Like other cnidarians, siphonophores capture prey with cases of this modularity can be observed in siphonophores, a clade of nematocysts, harpoon-like stinging capsules borne within special- pelagic colonial cnidarians that use tentilla (tentacle side branches ized cells known as cnidocytes. Unlike the prey-capture apparatus of armed with nematocysts) exclusively for prey capture. Here we study most other cnidarians, siphonophore tentacles carry their cnidocytes how siphonophore specialists and generalists evolve, and what mor- in extremely complex and organized batteries (3), which are located phological changes are associated with these transitions. -

Trophic Ecology of Gelatinous Zooplankton in Oceanic Food Webs of the Eastern Tropical Atlantic Assessed by Stable Isotope Analysis
Limnol. Oceanogr. 9999, 2020, 1–17 © 2020 The Authors. Limnology and Oceanography published by Wiley Periodicals LLC on behalf of Association for the Sciences of Limnology and Oceanography. doi: 10.1002/lno.11605 Tackling the jelly web: Trophic ecology of gelatinous zooplankton in oceanic food webs of the eastern tropical Atlantic assessed by stable isotope analysis Xupeng Chi ,1,2* Jan Dierking,2 Henk-Jan Hoving,2 Florian Lüskow,3,4 Anneke Denda,5 Bernd Christiansen,5 Ulrich Sommer,2 Thomas Hansen,2 Jamileh Javidpour2,6 1CAS Key Laboratory of Marine Ecology and Environmental Sciences, Institute of Oceanology, Chinese Academy of Sciences, Qingdao, China 2Marine Ecology, GEOMAR Helmholtz Centre for Ocean Research Kiel, Kiel, Germany 3Department of Earth, Ocean and Atmospheric Sciences, University of British Columbia, Vancouver, British Columbia, Canada 4Institute for the Oceans and Fisheries, University of British Columbia, Vancouver, British Columbia, Canada 5Institute of Marine Ecosystem and Fishery Science (IMF), Universität Hamburg, Hamburg, Germany 6Department of Biology, University of Southern Denmark, Odense M, Denmark Abstract Gelatinous zooplankton can be present in high biomass and taxonomic diversity in planktonic oceanic food webs, yet the trophic structuring and importance of this “jelly web” remain incompletely understood. To address this knowledge gap, we provide a holistic trophic characterization of a jelly web in the eastern tropical Atlantic, based on δ13C and δ15N stable isotope analysis of a unique gelatinous zooplankton sample set. The jelly web covered most of the isotopic niche space of the entire planktonic oceanic food web, spanning > 3 tro- phic levels, ranging from herbivores (e.g., pyrosomes) to higher predators (e.g., ctenophores), highlighting the diverse functional roles and broad possible food web relevance of gelatinous zooplankton. -

New Zealand Fishes a Field Guide to Common Species Caught by Bottom, Midwater, and Surface Fishing Cover Photos: Top – Kingfish (Seriola Lalandi), Malcolm Francis
New Zealand fishes A field guide to common species caught by bottom, midwater, and surface fishing Cover photos: Top – Kingfish (Seriola lalandi), Malcolm Francis. Top left – Snapper (Chrysophrys auratus), Malcolm Francis. Centre – Catch of hoki (Macruronus novaezelandiae), Neil Bagley (NIWA). Bottom left – Jack mackerel (Trachurus sp.), Malcolm Francis. Bottom – Orange roughy (Hoplostethus atlanticus), NIWA. New Zealand fishes A field guide to common species caught by bottom, midwater, and surface fishing New Zealand Aquatic Environment and Biodiversity Report No: 208 Prepared for Fisheries New Zealand by P. J. McMillan M. P. Francis G. D. James L. J. Paul P. Marriott E. J. Mackay B. A. Wood D. W. Stevens L. H. Griggs S. J. Baird C. D. Roberts‡ A. L. Stewart‡ C. D. Struthers‡ J. E. Robbins NIWA, Private Bag 14901, Wellington 6241 ‡ Museum of New Zealand Te Papa Tongarewa, PO Box 467, Wellington, 6011Wellington ISSN 1176-9440 (print) ISSN 1179-6480 (online) ISBN 978-1-98-859425-5 (print) ISBN 978-1-98-859426-2 (online) 2019 Disclaimer While every effort was made to ensure the information in this publication is accurate, Fisheries New Zealand does not accept any responsibility or liability for error of fact, omission, interpretation or opinion that may be present, nor for the consequences of any decisions based on this information. Requests for further copies should be directed to: Publications Logistics Officer Ministry for Primary Industries PO Box 2526 WELLINGTON 6140 Email: [email protected] Telephone: 0800 00 83 33 Facsimile: 04-894 0300 This publication is also available on the Ministry for Primary Industries website at http://www.mpi.govt.nz/news-and-resources/publications/ A higher resolution (larger) PDF of this guide is also available by application to: [email protected] Citation: McMillan, P.J.; Francis, M.P.; James, G.D.; Paul, L.J.; Marriott, P.; Mackay, E.; Wood, B.A.; Stevens, D.W.; Griggs, L.H.; Baird, S.J.; Roberts, C.D.; Stewart, A.L.; Struthers, C.D.; Robbins, J.E. -

Copyrighted Material
06_250317 part1-3.qxd 12/13/05 7:32 PM Page 15 Phylum Chordata Chordates are placed in the superphylum Deuterostomia. The possible rela- tionships of the chordates and deuterostomes to other metazoans are dis- cussed in Halanych (2004). He restricts the taxon of deuterostomes to the chordates and their proposed immediate sister group, a taxon comprising the hemichordates, echinoderms, and the wormlike Xenoturbella. The phylum Chordata has been used by most recent workers to encompass members of the subphyla Urochordata (tunicates or sea-squirts), Cephalochordata (lancelets), and Craniata (fishes, amphibians, reptiles, birds, and mammals). The Cephalochordata and Craniata form a mono- phyletic group (e.g., Cameron et al., 2000; Halanych, 2004). Much disagree- ment exists concerning the interrelationships and classification of the Chordata, and the inclusion of the urochordates as sister to the cephalochor- dates and craniates is not as broadly held as the sister-group relationship of cephalochordates and craniates (Halanych, 2004). Many excitingCOPYRIGHTED fossil finds in recent years MATERIAL reveal what the first fishes may have looked like, and these finds push the fossil record of fishes back into the early Cambrian, far further back than previously known. There is still much difference of opinion on the phylogenetic position of these new Cambrian species, and many new discoveries and changes in early fish systematics may be expected over the next decade. As noted by Halanych (2004), D.-G. (D.) Shu and collaborators have discovered fossil ascidians (e.g., Cheungkongella), cephalochordate-like yunnanozoans (Haikouella and Yunnanozoon), and jaw- less craniates (Myllokunmingia, and its junior synonym Haikouichthys) over the 15 06_250317 part1-3.qxd 12/13/05 7:32 PM Page 16 16 Fishes of the World last few years that push the origins of these three major taxa at least into the Lower Cambrian (approximately 530–540 million years ago). -

CNIDARIA Corals, Medusae, Hydroids, Myxozoans
FOUR Phylum CNIDARIA corals, medusae, hydroids, myxozoans STEPHEN D. CAIRNS, LISA-ANN GERSHWIN, FRED J. BROOK, PHILIP PUGH, ELLIOT W. Dawson, OscaR OcaÑA V., WILLEM VERvooRT, GARY WILLIAMS, JEANETTE E. Watson, DENNIS M. OPREsko, PETER SCHUCHERT, P. MICHAEL HINE, DENNIS P. GORDON, HAMISH J. CAMPBELL, ANTHONY J. WRIGHT, JUAN A. SÁNCHEZ, DAPHNE G. FAUTIN his ancient phylum of mostly marine organisms is best known for its contribution to geomorphological features, forming thousands of square Tkilometres of coral reefs in warm tropical waters. Their fossil remains contribute to some limestones. Cnidarians are also significant components of the plankton, where large medusae – popularly called jellyfish – and colonial forms like Portuguese man-of-war and stringy siphonophores prey on other organisms including small fish. Some of these species are justly feared by humans for their stings, which in some cases can be fatal. Certainly, most New Zealanders will have encountered cnidarians when rambling along beaches and fossicking in rock pools where sea anemones and diminutive bushy hydroids abound. In New Zealand’s fiords and in deeper water on seamounts, black corals and branching gorgonians can form veritable trees five metres high or more. In contrast, inland inhabitants of continental landmasses who have never, or rarely, seen an ocean or visited a seashore can hardly be impressed with the Cnidaria as a phylum – freshwater cnidarians are relatively few, restricted to tiny hydras, the branching hydroid Cordylophora, and rare medusae. Worldwide, there are about 10,000 described species, with perhaps half as many again undescribed. All cnidarians have nettle cells known as nematocysts (or cnidae – from the Greek, knide, a nettle), extraordinarily complex structures that are effectively invaginated coiled tubes within a cell. -

Idstorical Diver
Historical Diver, Number 3, 1994 Item Type monograph Publisher Historical Diving Society U.S.A. Download date 09/10/2021 13:15:37 Link to Item http://hdl.handle.net/1834/30846 IDSTORICAL DIVER Number 3 Summer 1994 The Official Publication of the Historical Diving Society U.S.A As you will by now know, the Society has relocated to Santa Barbara, California and this move, along with various other Society developments has delayed the publication of the Spring '94 issue of HISTORICAL DIVER. By way of catching up, we have produced a Summer double issue and have the good fortune to be able to publish with a color cover. Coinciding with the Santa Barbara relocation is the appointment, by the Board of Directors, of the first members of the HDS USA Advisory Board. This distinguished group of senior diving professionals, with extensive backgrounds in diving medicine, technical development, commercial, military and sports diving, bring in excess of 300 years of diving experience to the Society. Most of their biographies are the size of town phone directories, and have had to be severely edited for publication. We are honored and gratefulfortheir willing offers of service, and hope that we have done their biographies justice. Details start on page 4. The recently introduced, Founding Benefactor class of membership has proven to be very popular with over half of the thirty available memberships already taken. An opportunity still exists to acquire one of these unique memberships and details of it's benefits are noted on page 9. On the international front, the ongoing formation of the HDS USA as a nonprofit corporation has, by law, changed the conditions that govern our relationship with the HDS in UK. -

Articles and Plankton
Ocean Sci., 15, 1327–1340, 2019 https://doi.org/10.5194/os-15-1327-2019 © Author(s) 2019. This work is distributed under the Creative Commons Attribution 4.0 License. The Pelagic In situ Observation System (PELAGIOS) to reveal biodiversity, behavior, and ecology of elusive oceanic fauna Henk-Jan Hoving1, Svenja Christiansen2, Eduard Fabrizius1, Helena Hauss1, Rainer Kiko1, Peter Linke1, Philipp Neitzel1, Uwe Piatkowski1, and Arne Körtzinger1,3 1GEOMAR, Helmholtz Centre for Ocean Research Kiel, Düsternbrooker Weg 20, 24105 Kiel, Germany 2University of Oslo, Blindernveien 31, 0371 Oslo, Norway 3Christian Albrecht University Kiel, Christian-Albrechts-Platz 4, 24118 Kiel, Germany Correspondence: Henk-Jan Hoving ([email protected]) Received: 16 November 2018 – Discussion started: 10 December 2018 Revised: 11 June 2019 – Accepted: 17 June 2019 – Published: 7 October 2019 Abstract. There is a need for cost-efficient tools to explore 1 Introduction deep-ocean ecosystems to collect baseline biological obser- vations on pelagic fauna (zooplankton and nekton) and es- The open-ocean pelagic zones include the largest, yet least tablish the vertical ecological zonation in the deep sea. The explored habitats on the planet (Robison, 2004; Webb et Pelagic In situ Observation System (PELAGIOS) is a 3000 m al., 2010; Ramirez-Llodra et al., 2010). Since the first rated slowly (0.5 m s−1) towed camera system with LED il- oceanographic expeditions, oceanic communities of macro- lumination, an integrated oceanographic sensor set (CTD- zooplankton and micronekton have been sampled using nets O2) and telemetry allowing for online data acquisition and (Wiebe and Benfield, 2003). Such sampling has revealed a video inspection (low definition). -
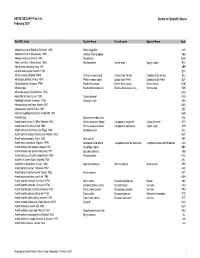
ASFIS ISSCAAP Fish List February 2007 Sorted on Scientific Name
ASFIS ISSCAAP Fish List Sorted on Scientific Name February 2007 Scientific name English Name French name Spanish Name Code Abalistes stellaris (Bloch & Schneider 1801) Starry triggerfish AJS Abbottina rivularis (Basilewsky 1855) Chinese false gudgeon ABB Ablabys binotatus (Peters 1855) Redskinfish ABW Ablennes hians (Valenciennes 1846) Flat needlefish Orphie plate Agujón sable BAF Aborichthys elongatus Hora 1921 ABE Abralia andamanika Goodrich 1898 BLK Abralia veranyi (Rüppell 1844) Verany's enope squid Encornet de Verany Enoploluria de Verany BLJ Abraliopsis pfefferi (Verany 1837) Pfeffer's enope squid Encornet de Pfeffer Enoploluria de Pfeffer BJF Abramis brama (Linnaeus 1758) Freshwater bream Brème d'eau douce Brema común FBM Abramis spp Freshwater breams nei Brèmes d'eau douce nca Bremas nep FBR Abramites eques (Steindachner 1878) ABQ Abudefduf luridus (Cuvier 1830) Canary damsel AUU Abudefduf saxatilis (Linnaeus 1758) Sergeant-major ABU Abyssobrotula galatheae Nielsen 1977 OAG Abyssocottus elochini Taliev 1955 AEZ Abythites lepidogenys (Smith & Radcliffe 1913) AHD Acanella spp Branched bamboo coral KQL Acanthacaris caeca (A. Milne Edwards 1881) Atlantic deep-sea lobster Langoustine arganelle Cigala de fondo NTK Acanthacaris tenuimana Bate 1888 Prickly deep-sea lobster Langoustine spinuleuse Cigala raspa NHI Acanthalburnus microlepis (De Filippi 1861) Blackbrow bleak AHL Acanthaphritis barbata (Okamura & Kishida 1963) NHT Acantharchus pomotis (Baird 1855) Mud sunfish AKP Acanthaxius caespitosa (Squires 1979) Deepwater mud lobster Langouste -

RACE Species Codes and Survey Codes 2018
Alaska Fisheries Science Center Resource Assessment and Conservation Engineering MAY 2019 GROUNDFISH SURVEY & SPECIES CODES U.S. Department of Commerce | National Oceanic and Atmospheric Administration | National Marine Fisheries Service SPECIES CODES Resource Assessment and Conservation Engineering Division LIST SPECIES CODE PAGE The Species Code listings given in this manual are the most complete and correct 1 NUMERICAL LISTING 1 copies of the RACE Division’s central Species Code database, as of: May 2019. This OF ALL SPECIES manual replaces all previous Species Code book versions. 2 ALPHABETICAL LISTING 35 OF FISHES The source of these listings is a single Species Code table maintained at the AFSC, Seattle. This source table, started during the 1950’s, now includes approximately 2651 3 ALPHABETICAL LISTING 47 OF INVERTEBRATES marine taxa from Pacific Northwest and Alaskan waters. SPECIES CODE LIMITS OF 4 70 in RACE division surveys. It is not a comprehensive list of all taxa potentially available MAJOR TAXONOMIC The Species Code book is a listing of codes used for fishes and invertebrates identified GROUPS to the surveys nor a hierarchical taxonomic key. It is a linear listing of codes applied GROUNDFISH SURVEY 76 levelsto individual listed under catch otherrecords. codes. Specifically, An individual a code specimen assigned is to only a genus represented or higher once refers by CODES (Appendix) anyto animals one code. identified only to that level. It does not include animals identified to lower The Code listing is periodically reviewed -

16.2 the WORLD of PERPETUAL DARKNESS the Lack of Food
Final PDF to printer 376 Part Three Structure and Function of Marine Ecosystems O2 abyssopelagic zone lies from 4,000 to 6,000 m (13,000 to 20,000 ft). The hadopelagic, or hadal pelagic, zone consists of the waters of the ocean trenches, from below 6,000 m to Amount of dissolved oxygen just above the sea floor, as deep as 11,000 m (36,000 ft). Low High Each of the depth zones supports a distinct community of animals, but they also have much in common. Here we hotosynthesi CO P s Organic 2 stress the similarities, rather than the differences, among the + matter H O + Epipelagic depth zones of the deep sea. 2 n O Respiratio 2 The conditions of life in the deep pelagic environment Decomposition change very little. Not only is it always dark, it is always 200 m cold: The temperature remains nearly constant, typically at 1 to 2 °C (35 °F). Salinity and other chemical properties of the water are also remarkably uniform. Oxygen minimum zone CO Organic 2 matter The deep sea also includes the ocean bottom beyond + H O Respiration + the continental shelf. Bottom-living organisms are covered 2 O Decomposition 2 Mesopelagic separately (see “The Deep-Ocean Floor,” below). The deep sea includes the bathypelagic, from 1,000 to 4,000 m; the abyssopelagic, 4,000 to 6,000 m; and the hadopelagic, 6,000 m to 1,000 m the bottom of trenches. The physical environment in these zones is quite constant. The deep sea also includes the deep-sea floor. -

Time Series Analysis and Visualization of Midwater Zooplankton Ecology: Automating Long-Term Studies of the VARS Dataset
Time series analysis and visualization of midwater zooplankton ecology: Automating long-term studies of the VARS dataset Alexander L. Jaffe, Harvard University, Department of Organismic and Evolutionary Biology Mentor: Steven Haddock Summer 2013 Abstract: This study uses multi-decadal time series from the Monterey Bay Aquarium Research Institute’s unique VARS database to illustrate spatio-temporal patterns of abundance in mesopelagic zooplankton. In situ observations from approximately 900 ROV dives over 25 years reveal both long and short-term fluctuations in abundance and diversity of these organisms, which heretofore have been largely understudied. Correlations between normalized data for midwater siphonophores, krill, and ctenophores indicated potential ecological relationships between taxa as well as sensitivity to oceanographic parameters like temperature, salinity, spiciness, and oxygen. We discuss evidence of niche partitioning between Praya dubia and Resomia ornicephala, as well as evidence of a bimodal distribution for the ctenophore Bolinopsis infundibulum. We also present trends in seasonality and periodicity within the context of environmental shift, and a method of reproduction for similar studies is set forth. Keywords: time-series, pelagic ecology, ROV, deep sea, VARS, spiciness 1. Introduction While it may be one of the largest habitats on Earth, the deep sea and its associated fauna have remained largely understudied (Schlining, 1999). The mesopelagic zone (~ 200-1000 m) in particular is home to a wide variety of complex organisms that are only recently known to science and whose biology and ecology is mostly a mystery (Robison, 2004). Despite this, recent studies (Buecher, 1999; Robison, 2004) have shown that these organisms, many of which are gelatinous, can have major impacts as both predators and competitors with repercussions for trophic networks throughout the pelagic realm.