Unit 12 Inborn Errors of Metabolism
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Clinical Applications and Implications of Common and Founder Mutations in Indian Subpopulations
REVIEW OFFICIAL JOURNAL Clinical Applications and Implications of Common and Founder Mutations in Indian Subpopulations www.hgvs.org Arunkanth Ankala,1∗ † Parag M. Tamhankar,2 † C. Alexander Valencia,3,4 Krishna K. Rayam,5 Manisha M. Kumar,5 and Madhuri R. Hegde1 1Department of Human Genetics, Emory University School of Medicine, Atlanta, Georgia; 2ICMR Genetic Research Center, National Institute for Research in Reproductive Health, Mumbai, Maharashtra, India; 3Division of Human Genetics, Cincinnati Children’s Hospital Medical Center, Cincinnati, Ohio; 4Department of Pediatrics, University of Cincinnati Medical School, Cincinnati, Ohio; 5Department of Biosciences, CMR Institute of Management Studies, Bangalore, Karnataka, India Communicated by Arupa Ganguly Received 24 October 2013; accepted revised manuscript 16 September 2014. Published online 27 November 2014 in Wiley Online Library (www.wiley.com/humanmutation). DOI: 10.1002/humu.22704 that include a lack of widespread awareness about genetic disorders in the general population and the scarcity of specialized medical ABSTRACT: South Asian Indians represent a sixth of the world’s population and are a racially, geographically, and professionals and affordable genetic tests. Seeking a molecular di- genetically diverse people. Their unique anthropological agnosis and understanding the risk estimates are critical to making structure, prevailing caste system, and ancient religious sound reproductive choices, especially in families with an affected practices have all impacted the genetic composition of most individual. Adding to this adversity is the absence of a properly func- of the current-day Indian population. With the evolving tioning social health care system and insufficient encouragement socio-religious and economic activities of the subsects and of individual health insurance by the government. -

Fructose As an Endogenous Toxin
HEPATOCYTE MOLECULAR CYTOTOXIC MECHANISM STUDY OF FRUCTOSE AND ITS METABOLITES INVOLVED IN NONALCOHOLIC STEATOHEPATITIS AND HYPEROXALURIA By Yan (Cynthia) Feng A thesis submitted in the conformity with the requirements for the degree of Master of Science Graduate Department of Pharmaceutical Sciences University of Toronto © Copyright by Yan (Cynthia) Feng 2010 ABSTRACT HEPATOCYTE MOLECULAR CYTOTOXIC MECHANISM STUDY OF FRUCTOSE AND ITS METABOLITES INVOLVED IN NONALCOHOLIC STEATOHEPATITIS AND HYPEROXALURIA Yan (Cynthia) Feng Master of Science, 2010 Department of Pharmaceutical Sciences University of Toronto High chronic fructose consumption is linked to a nonalcoholic steatohepatitis (NASH) type of hepatotoxicity. Oxalate is the major endpoint of fructose metabolism, which accumulates in the kidney causing renal stone disease. Both diseases are life-threatening if not treated. Our objective was to study the molecular cytotoxicity mechanisms of fructose and some of its metabolites in the liver. Fructose metabolites were incubated with primary rat hepatocytes, but cytotoxicity only occurred if the hepatocytes were exposed to non-toxic amounts of hydrogen peroxide such as those released by activated immune cells. Glyoxal was most likely the endogenous toxin responsible for fructose induced toxicity formed via autoxidation of the fructose metabolite glycolaldehyde catalyzed by superoxide radicals, or oxidation by Fenton’s hydroxyl radicals. As for hyperoxaluria, glyoxylate was more cytotoxic than oxalate presumably because of the formation of condensation product oxalomalate causing mitochondrial toxicity and oxidative stress. Oxalate toxicity likely involved pro-oxidant iron complex formation. ii ACKNOWLEDGEMENTS I would like to dedicate this thesis to my family. To my parents, thank you for the sacrifices you have made for me, thank you for always being there, loving me and supporting me throughout my life. -

Fructose in Medicine Jussi K. HUTTUNEN
Postgrad Med J: first published as 10.1136/pgmj.47.552.654 on 1 October 1971. Downloaded from Poitgraduate Medical Journal (October 1971) 47, 654-659. CURRENT SURVEY Fructose in medicine A review with particular reference to diabetes mellitus Jussi K. HUTTUNEN M.D. Third Department of Medicine, University of Helsinki, Finland Summary and in experimental animals. Earlier suggestions con- A review is given of the metabolism of fructose in the cerned with the atherogenic properties of fructose mammalian organism, and its significance in medicine. have recently been challenged. The apparent increase Emphasis is laid upon the absorption and assimilation in the incidence of coronary disease among sucrose of fructose through pathways not identical with those users seems to be a statistical artefact, caused by the of glucose. The metabolism of fructose is largely increased ingestion of coffee and soft drinks by insulin-independent, although the ultimate fate of cigarette smokers. fructose carbons is determined by the presence or the absence of insulin. Introduction Clinical and experimental work has suggested that The metabolism of fructose has engaged the atten- fructose may exert beneficial effects as a component tion of clinicians since 1874, when Kulz suggestedProtected by copyright. of the diet for patients with mild and well-balanced that diabetic patients can assimilate fructose better diabetes. Fructose is absorbed slowly from the gut, than glucose. These observations have been con- and does not induce drastic changes in blood sugar firmed in a number of experimental and clinical levels. Secondly, fructose is metabolized by insulin- studies (Minkowski, 1893; Naunyn, 1906; Joslin, independent pathways in the liver, intestinal wall, 1923), it being further shown that in some patients kidney and adipose tissue. -

Fructose and Mannose in Inborn Errors of Metabolism and Cancer
H OH metabolites OH Review Fructose and Mannose in Inborn Errors of Metabolism and Cancer Elizabeth L. Lieu †, Neil Kelekar †, Pratibha Bhalla † and Jiyeon Kim * Department of Biochemistry and Molecular Genetics, University of Illinois, Chicago, IL 60607, USA; [email protected] (E.L.L.); [email protected] (N.K.); [email protected] (P.B.) * Correspondence: [email protected] † These authors contributed equally to this work. Abstract: History suggests that tasteful properties of sugar have been domesticated as far back as 8000 BCE. With origins in New Guinea, the cultivation of sugar quickly spread over centuries of conquest and trade. The product, which quickly integrated into common foods and onto kitchen tables, is sucrose, which is made up of glucose and fructose dimers. While sugar is commonly associated with flavor, there is a myriad of biochemical properties that explain how sugars as biological molecules function in physiological contexts. Substantial research and reviews have been done on the role of glucose in disease. This review aims to describe the role of its isomers, fructose and mannose, in the context of inborn errors of metabolism and other metabolic diseases, such as cancer. While structurally similar, fructose and mannose give rise to very differing biochemical properties and understanding these differences will guide the development of more effective therapies for metabolic disease. We will discuss pathophysiology linked to perturbations in fructose and mannose metabolism, diagnostic tools, and treatment options of the diseases. Keywords: fructose and mannose; inborn errors of metabolism; cancer Citation: Lieu, E.L.; Kelekar, N.; Bhalla, P.; Kim, J. Fructose and Mannose in Inborn Errors of Metabolism and Cancer. -

Antenatal Diagnosis of Inborn Errors Ofmetabolism
816 ArchivesofDiseaseinChildhood 1991;66: 816-822 CURRENT PRACTICE Arch Dis Child: first published as 10.1136/adc.66.7_Spec_No.816 on 1 July 1991. Downloaded from Antenatal diagnosis of inborn errors of metabolism M A Cleary, J E Wraith The introduction of experimental treatment for Sample requirement and techniques used in lysosomal storage disorders and the increasing prenatal diagnosis understanding of the molecular defects behind By far the majority of antenatal diagnoses are many inborn errors have overshadowed the fact performed on samples obtained by either that for many affected families the best that can amniocentesis or chorion villus biopsy. For be offered is a rapid, accurate prenatal diag- some disorders, however, the defect is not nostic service. Many conditions remain at best detectable in this material and more invasive only partially treatable and as a consequence the methods have been applied to obtain a diagnos- majority of parents seek antenatal diagnosis in tic sample. subsequent pregnancies, particularly for those disorders resulting in a poor prognosis in terms of either life expectancy or normal neurological FETAL LIVER BIOPSY development. Fetal liver biopsy has been performed to The majority of inborn errors result from a diagnose ornithine carbamoyl transferase defi- specific enzyme deficiency, but in some the ciency and primary hyperoxaluria type 1. primary defect is in a transport system or Glucose-6-phosphatase deficiency (glycogen enzyme cofactor. In some conditions the storage disease type I) could also be detected by biochemical defect is limited to specific tissues this method. The technique, however, is inva- only and this serves to restrict the material avail- sive and can be performed by only a few highly able for antenatal diagnosis for these disorders. -

Severe Child Form of Primary Hyperoxaluria Type 2
Konkoľová et al. BMC Medical Genetics (2017) 18:59 DOI 10.1186/s12881-017-0421-8 CASE REPORT Open Access Severe child form of primary hyperoxaluria type 2 - a case report revealing consequence of GRHPR deficiency on metabolism Jana Konkoľová1,2*, Ján Chandoga1,2, Juraj Kováčik3, Marcel Repiský2, Veronika Kramarová2, Ivana Paučinová3 and Daniel Böhmer1,2 Abstract Background: Primary hyperoxaluria type 2 is a rare monogenic disorder inherited in an autosomal recessive pattern. It results from the absence of the enzyme glyoxylate reductase/hydroxypyruvate reductase (GRHPR). As a consequence of deficient enzyme activity, excessive amounts of oxalate and L-glycerate are excreted in the urine, and are a source for the formation of calcium oxalate stones that result in recurrent nephrolithiasis and less frequently nephrocalcinosis. Case presentation: We report a case of a 10-month-old patient diagnosed with urolithiasis. Screening of inborn errors of metabolism, including the performance of GC/MS urine organic acid profiling and HPLC amino acid profiling, showed abnormalities, which suggested deficiency of GRHPR enzyme. Additional metabolic disturbances observed in the patient led us to seek other genetic determinants and the elucidation of these findings. Besides the elevated excretion of 3-OH-butyrate, adipic acid, which are typical marks of ketosis, other metabolites such as 3- aminoisobutyric acid, 3-hydroxyisobutyric acid, 3-hydroxypropionic acid and 2-ethyl-3-hydroxypropionic acids were observed in increased amounts in the urine. Direct sequencing of the GRHPR gene revealed novel mutation, described for the first time in this article c.454dup (p.Thr152Asnfs*39) in homozygous form. The frequent nucleotide variants were found in AGXT2 gene. -

Metabolism of Disaccharides: Fructose and Galactose
Metabolism of disaccharides: Fructose and Galactose BCH 340 lecture 10 Fructose metabolism • Diets containing large amounts of sucrose (a disaccharide of glucose and fructose) can utilize the fructose as a major source of energy. • Fructose is found in foods containing sucrose (fruits), high-fructose corn syrups, and honey. • The pathway to utilization of fructose differs in muscle and liver. • In liver, dietary fructose is converted to Fructose-1-P by fructokinase (also in kidney and intestine). • Then, by the action of Fructose-1-P adolase (aldolase B), Fructose-1-P is converted to DHAP and glyceraldehyde. The utilization of fructose by fructokinase then aldolase bypass the steps of glucokinase and PFK-1 which are activated by insulin Glyceraldehyde is conveted to glyceraldehyde-3-P by triose kinase which together with DHAP may undergo: 1. Combine together and converted into glucose (main pathway) 2. They may be oxidized in glycolysis This explains why fructose disappears from blood more rapidly than glucose in diabetic subjects Fructose metabolism • The aldolase B is the rate-limiting enzyme for fructose metabolism. • Muscle which contains only hexokinase can phosphorylate fructose to F6P which is a direct glycolytic intermediate • However, hexokinase has a very low affinity to fructose compared to glucose, So it is not a significant pathway for fructose metabolism. Unless it is present in very high concentration in blood. Fructose-6-P can be converted to glucosamine-6-P which is the precursor of all other amino sugars glutamine amidotransferase glutamate Galactose Metabolism • The major source of galactose is lactose (a disaccharide of glucose and galactose) obtained from milk and milk products • Galactose enters glycolysis by its conversion to glucose-1-phosphate (G1P). -

Diagnose a Broad Range of Metabolic Disorders with a Single Test, Global
PEDIATRIC Assessing or diagnosing a metabolic disorder commonly requires several tests. Global Metabolomic Assisted Pathway Screen, commonly known as Global MAPS, is a unifying test GLOBAL MAPS™ for analyzing hundreds of metabolites to identify changes Global Metabolomic or irregularities in biochemical pathways. Let Global MAPS Assisted Pathway Screen guide you to an answer. Diagnose a broad range of metabolic disorders with a single test, Global MAPS Global MAPS is a large scale, semi-quantitative metabolomic profiling screen that analyzes disruptions in both individual analytes and pathways related to biochemical abnormalities. Using state-of-the-art technologies, Global Metabolomic Assisted Pathway Screen (Global MAPS) provides small molecule metabolic profiling to identify >700 metabolites in human plasma, urine, or cerebrospinal fluid. Global MAPS identifies inborn errors of metabolism (IEMs) that would ordinarily require many different tests. This test defines biochemical pathway errors not currently detected by routine clinical or genetic testing. IEMs are inherited metabolic disorders that prevent the body from converting one chemical compound to another or from transporting a compound in or out of a cell. NORMAL PROCESS METABOLIC ERROR These processes are necessary for essentially all bodily functions. Most IEMs are caused by defects in the enzymes that help process nutrients, which result in an accumulation of toxic substances or a deficiency of substances needed for normal body function. Making a swift, accurate diagnosis -

JCI96702.Pdf
Downloaded from http://www.jci.org on February 6, 2018. https://doi.org/10.1172/JCI96702 The Journal of Clinical Investigation REVIEW Fructose metabolism and metabolic disease Sarah A. Hannou,1 Danielle E. Haslam,2 Nicola M. McKeown,2 and Mark A. Herman1 1Division of Endocrinology and Metabolism and Duke Molecular Physiology Institute, Duke University Medical Center, Durham, North Carolina, USA. 2Nutritional Epidemiology Program, Jean Mayer US Department of Agriculture Human Nutrition Research Center on Aging, Tufts University, Boston, Massachusetts, USA. Increased sugar consumption is increasingly considered to be a contributor to the worldwide epidemics of obesity and diabetes and their associated cardiometabolic risks. As a result of its unique metabolic properties, the fructose component of sugar may be particularly harmful. Diets high in fructose can rapidly produce all of the key features of the metabolic syndrome. Here we review the biology of fructose metabolism as well as potential mechanisms by which excessive fructose consumption may contribute to cardiometabolic disease. Introduction recent prospective study showed that daily SSB consumers had a Glucose is the predominant form of circulating sugar in animals, 29% greater increase in visceral adipose tissue volume over 6 years while sucrose, the disaccharide composed of equal portions of glu- compared with nonconsumers (9). A causal association is sup- cose and fructose, is the predominant circulating sugar in plants. ported by evidence that intake of 1 liter of SSB daily for 6 months As plants form the basis of the food chain, herbivores and omni- increased visceral and liver fat, but increases were not observed in vores are highly adapted to use sucrose for energetic and biosyn- those consuming isocaloric semiskim milk, noncaloric diet soda, or thetic needs. -

Increased Prevalence of Hereditary Metabolic Diseases Among Native Indians in Manitoba and Northwestern Ontario
CLINICAL AND COMMUNITY STUDIES ETUDES CLINIQUES ET COMMUNAUTAIRES Increased prevalence of hereditary metabolic diseases among native Indians in Manitoba and northwestern Ontario James C. Haworth, MD, FRCPC; Louise A. Dilling, ART; Lorne E. Seargeant, PhD Objective: To compare the prevalence of hereditary metabolic diseases in the native and non-native populations of Manitoba and northwestern Ontario. Design: Retrospective analysis. Setting: Children's Hospital, Winnipeg. Patients: Patients were selected by three methods: laboratory tests designed to screen patients suspected of having a metabolic disease, laboratory investigation of newborn infants with abnormalities detected through screening, and investigation of near relatives of probands with disease. Results: A total of 138 patients with organic acid, amino acid and carbohydrate disorders were seen from 1960 to 1990. Of these, 49 (36%) were native Indians (Algonkian linguistic group). This was in sharp contrast to the proportion of native Indians in the total study population (5.8%). Congenital lactic acidosis due to pyruvate carboxylase deficiency (13 patients), glutaric aciduria type I (14 patients) and primary hyperoxaluria type II (8 patients) were the most common disorders detected. Other rare disorders included glutaric aciduria type II (one patient), 2-hydroxyglutaric aciduria (one patient) and sarcosinemia (one patient). Underreporting, especially of glutaric aciduria type I and hyperoxaluria type II, was likely in the native population. Conclusions: Hereditary metabolic diseases are greatly overrepresented in the native population of Manitoba and northwestern Ontario. We recommend that native children who present with illnesses involving disturbances of acid-base balance or with neurologic, renal or liver disease of unknown cause be investigated for a possible metabolic disorder. -
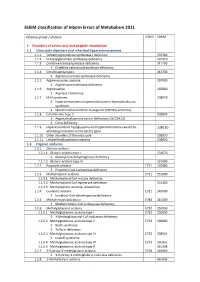
SSIEM Classification of Inborn Errors of Metabolism 2011
SSIEM classification of Inborn Errors of Metabolism 2011 Disease group / disease ICD10 OMIM 1. Disorders of amino acid and peptide metabolism 1.1. Urea cycle disorders and inherited hyperammonaemias 1.1.1. Carbamoylphosphate synthetase I deficiency 237300 1.1.2. N-Acetylglutamate synthetase deficiency 237310 1.1.3. Ornithine transcarbamylase deficiency 311250 S Ornithine carbamoyltransferase deficiency 1.1.4. Citrullinaemia type1 215700 S Argininosuccinate synthetase deficiency 1.1.5. Argininosuccinic aciduria 207900 S Argininosuccinate lyase deficiency 1.1.6. Argininaemia 207800 S Arginase I deficiency 1.1.7. HHH syndrome 238970 S Hyperammonaemia-hyperornithinaemia-homocitrullinuria syndrome S Mitochondrial ornithine transporter (ORNT1) deficiency 1.1.8. Citrullinemia Type 2 603859 S Aspartate glutamate carrier deficiency ( SLC25A13) S Citrin deficiency 1.1.9. Hyperinsulinemic hypoglycemia and hyperammonemia caused by 138130 activating mutations in the GLUD1 gene 1.1.10. Other disorders of the urea cycle 238970 1.1.11. Unspecified hyperammonaemia 238970 1.2. Organic acidurias 1.2.1. Glutaric aciduria 1.2.1.1. Glutaric aciduria type I 231670 S Glutaryl-CoA dehydrogenase deficiency 1.2.1.2. Glutaric aciduria type III 231690 1.2.2. Propionic aciduria E711 232000 S Propionyl-CoA-Carboxylase deficiency 1.2.3. Methylmalonic aciduria E711 251000 1.2.3.1. Methylmalonyl-CoA mutase deficiency 1.2.3.2. Methylmalonyl-CoA epimerase deficiency 251120 1.2.3.3. Methylmalonic aciduria, unspecified 1.2.4. Isovaleric aciduria E711 243500 S Isovaleryl-CoA dehydrogenase deficiency 1.2.5. Methylcrotonylglycinuria E744 210200 S Methylcrotonyl-CoA carboxylase deficiency 1.2.6. Methylglutaconic aciduria E712 250950 1.2.6.1. Methylglutaconic aciduria type I E712 250950 S 3-Methylglutaconyl-CoA hydratase deficiency 1.2.6.2. -

Volume 15 Johannes Zschocke K. Michael Gibson Garry Brown Eva Morava Verena Peters Editors
Johannes Zschocke K. Michael Gibson Garry Brown Eva Morava Verena Peters Editors JIMD Reports Volume 15 JIMD Reports Volume 15 . Johannes Zschocke • K. Michael Gibson Editors-in-Chief Garry Brown • Eva Morava Editors Verena Peters Managing Editor JIMD Reports Volume 15 Editor-in-Chief Editor Johannes Zschocke Eva Morava Division of Human Genetics Tulane University Medical School Medical University Innsbruck New Orleans Innsbruck Louisiana Austria USA Editor-in-Chief Managing Editor K. Michael Gibson Verena Peters WSU Division of Health Sciences Center for Child and Adolescent Clinical Pharmacology Unit Medicine Spokane Heidelberg University Hospital USA Heidelberg Germany Editor Garry Brown University of Oxford Department of Biochemistry Genetics Unit Oxford United Kingdom ISSN 2192-8304 ISSN 2192-8312 (electronic) ISBN 978-3-662-43750-6 ISBN 978-3-662-43751-3 (eBook) DOI 10.1007/978-3-662-43751-3 Springer Heidelberg New York Dordrecht London # SSIEM and Springer-Verlag Berlin Heidelberg 2015 This work is subject to copyright. All rights are reserved by the Publisher, whether the whole or part of the material is concerned, specifically the rights of translation, reprinting, reuse of illustrations, recitation, broadcasting, reproduction on microfilms or in any other physical way, and transmission or information storage and retrieval, electronic adaptation, computer software, or by similar or dissimilar methodology now known or hereafter developed. Exempted from this legal reservation are brief excerpts in connection with reviews or scholarly analysis or material supplied specifically for the purpose of being entered and executed on a computer system, for exclusive use by the purchaser of the work. Duplication of this publication or parts thereof is permitted only under the provisions of the Copyright Law of the Publisher’s location, in its current version, and permission for use must always be obtained from Springer.