Structures of Selected Boranes and Carboranes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
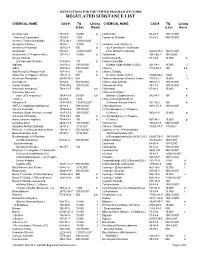
Regulated Substance List
INSTRUCTIONS FOR THE UNIFIED PROGRAM (UP) FORM REGULATED SUBSTANCE LIST CHEMICAL NAME CAS # TQ Listing CHEMICAL NAME CAS # TQ Listing (Lbs) Basis (Lbs) Basis Acetaldehyde 75-07-0 10,000 g Cantharidin 56-25-7 100/10,0001 * Acetone Cyanohydrin 75-86-5 1,000 Carbachol Chloride 51-83-2 500/10,0001 Acetone Thiosemicarbazide 1752-30-3 1,000/10,0001 Acetylene (Ethyne) 74-86-2 10,000 f Carbamic Acid, Methyl-,o- Acrolein (2-Propenal) 107-02-8 500 b (((2,4-Dimethyl-1,3-Dithiolan- Acrylamide 79-06-1 1,000/10,0001 2-YL) Methylene)Amino)- 26419-73-8 100/10,0001 Acrylonitrile (2- Propenenitrile) 107-13-1 10,000 b Carbofuran 1563-66-2 10/10,0001 Acrylyl Chloride Carbon Disulfide 75-15-0 10,000 b (2-Propenoyl Chloride) 814-68-6 100 b Carbon Oxysulfide Aldicarb 116-06-3 100/10,0001 (Carbon Oxide Sulfide (COS)) 463-58-1 10,000 f Aldrin 309-00-2 500/10,0001 Chlorine 7782-50-5 100 a,b Allyl Alcohol (2-Propen-1-ol) 107-18-6 1,000 b Chlorine Dioxide Allylamine (2-Propen-1-Amine) 107-11-9 500 b (Chlorine Oxide (ClO2)) 10049-04-4 1,000 c Aluminum Phosphide 20859-73-8 500 Chlorine Monoxide (Chlorine Oxide) 7791-21-1 10,000 f Aminopterin 54-62-6 500/10,0001 Chlormequat Chloride 999-81-5 100/10,0001 Amiton Oxalate 3734-97-2 100/10,0001 Chloroacetic Acid 79-11-8 100/10,0001 Ammonia, Anhydrous 2 7664-41-7 500 a,b Chloroform 67-66-3 10,000 b Ammonia, Aqueous Chloromethyl Ether (conc 20% or greater) 7664-41-7 20,000 a,b (Methane,Oxybis(chloro-) 542-88-1 100 b * Aniline 62-53-3 1,000 Chloromethyl Methyl Ether Antimycin A 1397-94-0 1,000/10,0001 (Chloromethoxymethane) -

Thermodynamic Hydricity of Small Borane Clusters and Polyhedral Closo-Boranes
molecules Article Thermodynamic Hydricity of Small Borane Clusters y and Polyhedral closo-Boranes Igor E. Golub 1,* , Oleg A. Filippov 1 , Vasilisa A. Kulikova 1,2, Natalia V. Belkova 1 , Lina M. Epstein 1 and Elena S. Shubina 1,* 1 A. N. Nesmeyanov Institute of Organoelement Compounds and Russian Academy of Sciences (INEOS RAS), 28 Vavilova St, 119991 Moscow, Russia; [email protected] (O.A.F.); [email protected] (V.A.K.); [email protected] (N.V.B.); [email protected] (L.M.E.) 2 Faculty of Chemistry, M.V. Lomonosov Moscow State University, 1/3 Leninskiye Gory, 119991 Moscow, Russia * Correspondence: [email protected] (I.E.G.); [email protected] (E.S.S.) Dedicated to Professor Bohumil Štibr (1940-2020), who unfortunately passed away before he could reach the y age of 80, in the recognition of his outstanding contributions to boron chemistry. Academic Editors: Igor B. Sivaev, Narayan S. Hosmane and Bohumír Gr˝uner Received: 6 June 2020; Accepted: 23 June 2020; Published: 25 June 2020 MeCN Abstract: Thermodynamic hydricity (HDA ) determined as Gibbs free energy (DG◦[H]−) of the H− detachment reaction in acetonitrile (MeCN) was assessed for 144 small borane clusters (up 2 to 5 boron atoms), polyhedral closo-boranes dianions [BnHn] −, and their lithium salts Li2[BnHn] (n = 5–17) by DFT method [M06/6-311++G(d,p)] taking into account non-specific solvent effect (SMD MeCN model). Thermodynamic hydricity values of diborane B2H6 (HDA = 82.1 kcal/mol) and its 2 MeCN dianion [B2H6] − (HDA = 40.9 kcal/mol for Li2[B2H6]) can be selected as border points for the range of borane clusters’ reactivity. -

Minutesbasel Approved
Approved minutes, Basel 2018 International Union of Pure and Applied Chemistry Division VIII Chemical Nomenclature and Structure Representation Approved minutes for Division Committee Meeting Basel, Switzerland, 13–14 August, 2018 1. Welcome, introductory remarks and housekeeping announcements Alan Hutton (ATH) welcomed everybody to the meeting, extending a special welcome to those who were attending the Division Committee (DC) meeting for the first time, and noting the presence of three former Presidents of the Division, as well as the current Secretary General of IUPAC. He described the house rules and arrangements for the course of the meeting. 2. Attendance and apologies Present: Alan T. Hutton (President, ATH), Karl-Heinz Hellwich (Past-President, KHH), Risto S. Laitinen (Secretary, RSL), Michael A. Beckett (MAB), Edwin C. Constable (ECC), Ture Damhus (TD), Richard M. Hartshorn (RMH), Elisabeth Mansfield (EM), Gerry P. Moss (GPM), Michelle M. Rogers (MMR), Molly A. Strausbaugh (MAS), Clare Tovee (CT), Andrey Yerin (AY) (see also Appendix 1) Apologies: Fabio Aricò (FA), Maria Atanasova Petrova (MA), Neil Burford (NB), (JC), Ana Maria da Costa Ferreira (ACF), Safiye Erdem (SE), Rafał Kruszyński (RK), Robin Macaluso (RM), , Ladda Meesuk (LM), Ebbe Nordlander (EN), Amelia P. Rauter (APR), Erik Szabó (ES), Keith T. Taylor (KTT), Jiří Vohlídal (JV) Invited observer: G. Jeffery Leigh (GJL) No replies: József Nagy, Sangho Koo 3. Introduction of attendees A short round of introductions was made. A new titular member, Prof. Edwin C. Constable and a new Associate Member, Dr. Clare Tovee, were attending the meeting of the Division Committee for the first time in this function. KHH informed the meeting that the following persons had passed away during recent years: Peter A. -

Boranes: Physical & Chemical Properties, Encyclopaedia of Occupational Health and Safety, Jeanne Mager Stellman, Editor-In
Boranes: Physical & chemical properties, Encyclopaedia of Occupational Health and Safety, Jeanne Mager Stellman, Editor-in-Chief. International Labor Organization, Geneva. 2011. Chemical Name Colour/Form Boiling Point Melting Molecular Solubility in Relative Density Relative Vapour Inflam. Flash Auto CAS-Number (°C) Point (°C) Weight Water (water=1) Vapour Pressure/ Limits Point (°C) Ignition Density (Kpa) Point (°C) (air=1) BORON polymorphic: alpha- 2550 2300 10.81 insol Amorphous, 1.56x 580 3 -5 7440-42-8 rhombohedral form, clear 2.3 g/cm ; 10 red crystals; beta- alpha-- @ 2140 °C rhombohedral form, black; rhombohedral, - alpha-tetragonal form, 2.46 g/cm3; - black, opaque crystals with alpha-- metallic luster; amorphous tetragonal, - form, black or dark brown 2.31 g/cm3; - powder; other crystal beta-rhom- forms known bohedral, - 2.35 g/cm3 BORIC ACID, DISODIUM powder or glass-like 1575 741 201.3 2.56 g/100 g 2.367 SALT plates; white, free-flowing 1330-43-4 crystals; light grey solid BORON OXIDE rhombic crystals; 1860 450 69.6 2.77 g/100 g 1.8 1303-86-2 colourless, (amorphous); semitransparent lumps or 2.46 hard, white crystals (crystalline) BORON TRIBROMIDE colourless liquid 90 -46.0 250.57 reacts 2.6431 8.6 5.3 10294-33-4 @ 18.4 °C/4 °C @ 14 °C BORON TRICHLORIDE 12.5 -107 117.16 1.35 4.03 2.99 Pa 10294-34-5 @ 12 °C/4 @ 12.4 °C BORON TRIFLUORIDE colourless gas -99.9 -126.8 67.82 reacts 3.08g/1.57 l 2.4 10 mm Hg 7637-07-2 @ 4 °C @ -141 °C. -

Boranes: Structure & Bonding
18-Aug-20 Boranes: Structure & Bonding PROF. K.K. UPADHYAY DEPARTMENT OF CHEMISTRY INSTITUTE OF SCIENCE, BHU VARANASI - 221005 An Introduction to Boranes . For undergraduate students, boranes means only diborane which is not true. Boranes means electron deficient molecules but probably that is not true. Boranes are a class of compounds comprising of several hundreds of compounds ranging from simple to polyhedral to macro polyhedral types having intriguing structures. 1 18-Aug-20 10 11 . Boron is an element with atomic no. 5 having two isotopes 5B (20%) and 5B (80%). It occupies group XIII and is a p-block element. .Boranes are binary compounds of boron and hydrogen and are the fourth most extensive group of hydrides after the Carbon, Phosphorous and Silicon hydrides. BH 3 is the simplest of all the boranes but non-existent. B2H6 is the dimer of BH 3 and is the most primitive among the existing boranes. Boranes are not found in the nature. These are always synthesised in the laboratory. Very first synthesis was carried out in 19 th century by protolysis of metal borides. 2Mg 3B2 + 12HCl 6MgCl 2 + mixture of boranes . But neither correctly analysed nor identified. 2 18-Aug-20 . The first systematic study of boranes was performed by Alfred stock during the year 1912-1936. He used Schlenk line technique for the synthesis of boranes in a systematic way. Alfred Stock . He studied nature, stoichiometry and reactivity of boranes in a systematic way. 3 NaBH 4 + 4 BF 3→ 3 NaBF 4 + mixture of boranes BCl 3 + 3 H 2 → mixture of boranes + 3 HCl - 3- BH 4 + BX 3 → mixture of boranes + HBX (X = Cl, Br) Schlenk line 2x4 + 1x6 = 14 electrons 2x3 + 1x6 = 12 electrons . -

UC Riverside Electronic Theses and Dissertations
UC Riverside UC Riverside Electronic Theses and Dissertations Title Carboranes: Building Blocks for Materials and Ligand Development Permalink https://escholarship.org/uc/item/2vp9m2z6 Author Estrada, Jess Steven Publication Date 2017 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA RIVERSIDE Carboranes: Building Blocks for Materials and Ligand Development A Dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Chemistry by Jess Steven Estrada September 2017 Dissertation Committee: Dr. Vincent Lavallo, Chairperson Dr. Richard Hooley Dr. Pingyun Feng Copyright by Jess Steven Estrada 2017 The Dissertation Jess Steven Estrada is approved: Committee Chairperson University of California, Riverside Acknowledgements I would like to thank Dr. Vincent Lavallo for giving me the opportunity to join his lab and for all of his help and support throughout graduate school. I owe a lot of my success to you. I would also like to thank the amazing faculty at UCR for all of their help and knowledge they have provided. I truly enjoyed every class I took here at UCR. In addition to the faculty, the staff in the chemistry department has been amazing over the last five years and played a huge role in my success as well so I would specifically like to thank Dr. Borchardt, the NMR genius, Dr. Fook for all of his help with my great looking X-ray structures, Christina Youhas for answering literally every question I ever had and being so kind about answering. My lab mates, who created the most unique group of people probably in the history of UCR and that I’ve ever had the pleasure of working with. -

Reactions of Titanium Imides and Hydrazides with Boranes Simona Mellino,† Laura C
This is an open access article published under a Creative Commons Attribution (CC-BY) License, which permits unrestricted use, distribution and reproduction in any medium, provided the author and source are cited. Article pubs.acs.org/Organometallics Reactions of Titanium Imides and Hydrazides with Boranes Simona Mellino,† Laura C. Stevenson,† Eric Clot,*,‡ and Philip Mountford*,† † Chemistry Research Laboratory, Department of Chemistry, University of Oxford, Mansfield Road, Oxford OX1 3TA, U.K. ‡ Institut Charles Gerhardt Montpellier, UMR 5253 CNRS-UM-ENSCM, Universitéde Montpellier, cc 1501, Place Eugené Bataillon, F-34095 Montpellier Cedex 5, France *S Supporting Information ABSTRACT: We report the first reactions of titanium imido, alkylidene hydrazido, and dimethylhydrazido compounds with the F F * i boranes H2BTex, 9-BBN, HBAr 2, and HBPin (Tex = tert-hexyl; Ar =C6F5). Reactions of Cp Ti{MeC(N Pr)2}(NTol) with F * i ′ − H2BTex, 9-BBN, or HBAr 2 resulted in the hydride-bridged adducts Cp Ti{MeC(N Pr2)2}{N(Tol)HBRR } without B H bond * i F β cleavage. Cp Ti{MeC(N Pr)2}(NNCPh2)(4) reacted with HBAr 2 via a sequence of steps involving adducts at the - and then α − -nitrogen of the NNCPh2 ligand, before slow 1,2-addition of B H across the N CPh2 double bond of 4, forming * i F Cp Ti{MeC(N Pr)2}{NN(BAr 2)CHPh2}(17). The other boranes reacted immediately with 4 to form homologues of 17. fi * i These products are the rst examples of borylhydrazido(2-) complexes. Reaction of Cp Ti{MeC(N Pr2)2}(NNMe2)(2) with F * i F − HBAr 2 gave the hydride-bridged adduct Cp Ti{MeC(N Pr2)2}{N(NMe2)HBAr 2}, whereas with HBPin B H bond cleavage * i occurred to form the borylhydrazide(1-)-hydride Cp Ti{MeC(N Pr)2}(H){N(BPin)NMe2}. -

United States Patent Office Patented Jan
3,489,812 United States Patent Office Patented Jan. 13, 1970 2 3,489,812 and sufficient dialkyl sulfide to permit convenient stirring METHOD OF PREPARING BIS-(DIETHYL of the reactant mixture. The reaction should be carried SULFIDE)-DECABORANE out in the absence of air or other oxidizing gas, suitably Mervin D. Marshall, Fombell, and Richard M. Hunt and by using a vacuum or nonoxidizing cover gas such as Gerald T. Heiferan, Butler, Pa., assignors to Mine nitrogen or argon. Safety Appliances Company, a corporation of Pennsyl vania tion:The following examples are illustrative of this inven No Drawing. Filed Jan. 15, 1968, Ser. No. 697,596 Int, C. C07f 5/02 EXAMPLE I U.S. C. 260-606.5 1 Claim O A glass reactor equipped with a magnetic stirrer and connected to a glass vacuum apparatus was charged with ABSTRACT OF THE DISCLOSURE 3.513 g. of (NH4)2BioHo (22.6 millimols) and 40 ml. of diethyl sulfide, cooled to about 4° C. and evacuated. Bis-(diethyl sulfide)-decaborane is prepared by reac Measured aliquots of anhydrous hydrogen chloride (92. tion of ammonium decahydrodecarborate, HCl and di 15 immoles) were added in two equal increments. This reac ethyl sulfide. tion mixture was allowed to warm to room temperature and stirred for 72 hours. A white precipitate formed which Bis-(dialkyl sulfide)-decaboranes, such as bis-(dimethyl was filtered from the diethyl sulfide solution. The evapora sulfide)-decaborane, BioH12S (CH3)2)2, and bis-(diethyl tion of excess diethyl sulfide from the solution gave 4.596 sulfide)-decaborane, BioH12S (C2H5)2)2, have been used g. -

Innovation Infosheet Preparation of Amine Boranes, Ammonia Borane, and Phosphine Boranes
Innovation Infosheet Downloaded September 29, 2021 Preparation of Amine Boranes, Ammonia Borane, and Phosphine Boranes Track Code: 2014-RAMA-66892 Categories: - Chemistry and Chemical Analysis Keywords: - Amine-Boranes - Chemistry and Chemical Analysis - Hydrogen Storage Amine and phosphine-boranes, the classic Lewis base-Lewis acid (LB-LA) pairs have been known and valued for over a century, with uses that include hydrogen storage applications. There are major drawbacks associated with current procedures for making amine-boranes, including low efficiency and other issues such as formation of highly unpleasant odors from the necessary reactions. In addition, removal of dimethyl sulfide for reuse of the solvent THF adds additional steps in the synthetic process. Therefore, as the demand for large quantities of amine- boranes increases, there is a need for more effective methods for making ammonia borane and amine-boranes. Researchers at Purdue University have developed several new procedures to efficiently make ammonia borane and amine boranes in large quantities. In addition, due to the stability of sodium borohydride (SBH) and ammonia borane in water, water can be used in the production process, allowing for an efficient, cost-effective synthesis of ammonia borane from SBH. Advantages: -Simplest possible synthesis of ammonia borane from sodium borohydride -Prepare ammonia-borane in large quantities Potential Applications: -Method for making a more pure ammonia borane and amine borane People: - Ramachandran, Padinjaremadhom V (Project -

Nite States Patent O?Ice Patented Apr
3,030,423 nite States Patent O?ice Patented Apr. 17, 1962 1 2 cuprous chloride catalyst employed also can be varied 3,030,423 REACTION PRODUCTS 0F ACETYLENES widely, generally being in the range of from 0.005 to 0.5 AND DECABORANE mole of cuprous chloride per mole of decaborane and Starling K. Alley, Beverly Hills, Calif., and Otto Fuchs, preferably from 0.01 to 0.2 mole of cuprous chloride per Tonawanzia, N.Y., assignors to Olin Mathieson Chemi- 5 mole of decaborane. The reaction temperature can vary cal Corporation, a corporation of Virginia from 100 to 350° C. and preferably from 150 to 250° C. No Drawing. Filed Feb. 13, 1959, Ser. No. 793,211 The pressure can vary from atmospheric pressure to about 3 Claims. (Cl. 260-6065) 700 p.s.i.g., although closed system reactions are usually conducted at about 100 to 600 p.s.i.g. The reaction gen This invention relates to solid products obtained by 10 erally requires about 1 to 10 hours depending upon the the reaction of acetylene or methylacetylene with deca~ ratio of reactants and the temperature and pressure em borane. ployed. The solid products of this invention, when incorporated Although the reaction will proceed in the absence of a with suitable oxidizers such as ammonium perchlorate, solvent, a solvent common for the reactants but inert with potassium perchlorate, sodium perchlorate, etc., yield 15 respect to the reactants under the reaction conditions is solid propellants generally suitable for rocket power usually employed. Such solvents include aliphatic hydro plants and other jet propelled devices. -

The Use of Carboranes in Cancer Drug Develop-Ment
ISSN: 2378-3419 Zargham et al. Int J Cancer Clin Res 2019, 6:110 DOI: 10.23937/2378-3419/1410110 Volume 6 | Issue 2 International Journal of Open Access Cancer and Clinical Research REviEW ARtiCLE The Use of Carboranes in Cancer Drug Development Emilia O Zargham, Christian A Mason and Mark W Lee Jr* Check for Department of Chemistry, University of Missouri, Columbia, Missouri, USA updates *Corresponding author: Mark W Lee Jr., Department of Chemistry, University of Missouri, 601 South College Avenue, Columbia, Missouri, 65211, USA, Tel: 573-884-2424 erwise rapidly metabolize [5]. Furthermore, selective Abstract chemical substitution of each carbon or boron atom Over the past decade, there has been a rising interest in these clusters allows for their use as rigid, three di- in the use of carboranes as a potential pharmacophoric moiety in the development of new drugs for the treatment of mensional scaffolds upon which to construct new drug various types of cancer. The unique physical and chemical molecules. properties of carboranes make their use attractive in drug development. In several instances, the inclusion of Nearly all past biomedical research involving carbo- carboranes into a drug structure has increased the agent’s ranes has focused on their use in the design of boron de- binding affinity, potency, or bioavailability. The purpose of livery agents for boron neutron capture therapy (BNCT) this review is to highlight applications of carboranes to the [2]. This binary radiation therapy depends on the selec- medicinal chemistry of cancer. tive delivery of a high concentration of boron-10 atoms to targeted tissues. -

Prep Aration and Pro Perties of Trifluoromethyl• Boranes Everett Duane Baker a Thesis Oregon State College Master of Science
PREP ARATION AND PRO PERTIES OF TRIFLUOROMETHYL• BORANES by EVERETT DUANE BAKER A THESIS submitted to OREGON STATE COLLEGE in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE June 1961 APTBOVEDT Redacted for Privacy Arroolat. Profilts sf Chrrl. tlt In 0hrr.gr of, l{rt or Redacted for Privacy Ghrlrarn of Drpuhcat of Chralttry Redacted for Privacy Chr lrun Sobool Orrdurtr Comtttrr Redacted for Privacy Dorn of &rrductr Scbool Drtr thrrlr 1r prrcntod )g Au2a2t l%O Iypod by LrAnna Ermlr ACKNOWLEDGMENT Tbe author wishes to express his sincere appre ciation to Dr- Theran D. Parsons tor h1s guidance and encouragement throughout tha course of this inves tigation. The material aid ot the Office ot Naval Research fellowship is acknowledged w1th appreciation. TABLE OF CONTENTS Page I. INTRODUCTION ' • 1 • • ... • • • • • • • • • • • • II. EXPERIMENTAL • •• ,, ... • ••• • • • .... • • • 1) A~ Appara tue and Eqatpme.nt. .. • • • • • • • • • • 1) B. Reagen t8 • • • • • • • • • • • • • • • • • • .1) c. P~epal!lt1ona and ·lteactiona • • • • • , , • • .15 Figure I , • • • • ·• ... ·• • • • • • • • • • .16 Table I • •· • • • • • • , • • • • • • • • ~ Figure 2 • • • • • • • • • • • • • • • • • •. :~~ lli. DISCUSSION AND CONCLUSIONS ·• .. • • . ·• . .29 IV, BIB.LIOGRAl?iY • • • • .• • • • • • • • • • • • • • •34 PREPARATION AND PROPERTIES OF TRIFLUROMETHYL-BOR ANES I. INTRODUCTION The elucidatio.n or the struct\ll'es of the boron hydrides and their ~erivatives, and of certain compounds of aluminum, has caused great interest in electron deficient bonding. Lipscomb (14) reviewed the s t ructures ot the boron hydrides. Evidence was cited which s how s that diborane has a structure in which. two of the hydrogen atoms form a bridge between the two boron atoms: H H H \. 1\ I B B / \. / \ H H H The grouping of the atoms bonde d to a boron a tom is ap prox imately tetrahedral in configuration.