University of Cincinnati
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Dysregulated Hepatic Methionine
Virginia Commonwealth University VCU Scholars Compass Internal Medicine Publications Dept. of Internal Medicine 2015 Dysregulated Hepatic Methionine Metabolism Drives Homocysteine Elevation in Diet-Induced Nonalcoholic Fatty Liver Disease Tommy Pacana Virginia Commonwealth University, [email protected] Sophie Cazanave Virginia Commonwealth University Aurora Verdianelli Virginia Commonwealth University See next page for additional authors Follow this and additional works at: http://scholarscompass.vcu.edu/intmed_pubs Part of the Medicine and Health Sciences Commons Copyright: © 2015 Pacana et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited Downloaded from http://scholarscompass.vcu.edu/intmed_pubs/98 This Article is brought to you for free and open access by the Dept. of Internal Medicine at VCU Scholars Compass. It has been accepted for inclusion in Internal Medicine Publications by an authorized administrator of VCU Scholars Compass. For more information, please contact [email protected]. Authors Tommy Pacana, Sophie Cazanave, Aurora Verdianelli, Viashali Patel, Hae-Ki Min, Faridoddin Mirshahi, Eoin Quinlavin, and Arun J. Sanyal This article is available at VCU Scholars Compass: http://scholarscompass.vcu.edu/intmed_pubs/98 RESEARCH ARTICLE Dysregulated Hepatic Methionine Metabolism Drives Homocysteine Elevation in Diet-Induced Nonalcoholic Fatty -

Review Article Cystathionine -Synthase in Physiology and Cancer
Hindawi BioMed Research International Volume 2018, Article ID 3205125, 11 pages https://doi.org/10.1155/2018/3205125 Review Article Cystathionine �-Synthase in Physiology and Cancer Haoran Zhu,1,2 Shaun Blake,1,2 Keefe T. Chan,1 Richard B. Pearson ,1,2,3,4 and Jian Kang 1 1 Division of Research, Peter MacCallum Cancer Centre, 305 Grattan Street, Melbourne, Victoria 3000, Australia 2Sir Peter MacCallum Department of Oncology, Australia 3Department of Biochemistry and Molecular Biology, University of Melbourne, Parkville, Victoria 3052, Australia 4Department of Biochemistry and Molecular Biology, Monash University, Clayton, Victoria 3168, Australia Correspondence should be addressed to Richard B. Pearson; [email protected] Received 23 March 2018; Accepted 29 May 2018; Published 28 June 2018 Academic Editor: Maria L. Tornesello Copyright © 2018 Haoran Zhu et al. Tis is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Cystathionine �-synthase (CBS) regulates homocysteine metabolism and contributes to hydrogen sulfde (H2S) biosynthesis through which it plays multifunctional roles in the regulation of cellular energetics, redox status, DNA methylation, and protein modifcation. Inactivating mutations in CBS contribute to the pathogenesis of the autosomal recessive disease CBS-defcient homocystinuria. Recent studies demonstrating that CBS promotes colon and ovarian cancer growth in preclinical models highlight a newly identifed oncogenic role for CBS. On the contrary, tumor-suppressive efects of CBS have been reported in other cancer types, suggesting context-dependent roles of CBS in tumor growth and progression. Here, we review the physiological functions of CBS, summarize the complexities regarding CBS research in oncology, and discuss the potential of CBS and its key metabolites, including homocysteine and H2S, as potential biomarkers for cancer diagnosis or therapeutic targets for cancer treatment. -

Supplementary Table S4. FGA Co-Expressed Gene List in LUAD
Supplementary Table S4. FGA co-expressed gene list in LUAD tumors Symbol R Locus Description FGG 0.919 4q28 fibrinogen gamma chain FGL1 0.635 8p22 fibrinogen-like 1 SLC7A2 0.536 8p22 solute carrier family 7 (cationic amino acid transporter, y+ system), member 2 DUSP4 0.521 8p12-p11 dual specificity phosphatase 4 HAL 0.51 12q22-q24.1histidine ammonia-lyase PDE4D 0.499 5q12 phosphodiesterase 4D, cAMP-specific FURIN 0.497 15q26.1 furin (paired basic amino acid cleaving enzyme) CPS1 0.49 2q35 carbamoyl-phosphate synthase 1, mitochondrial TESC 0.478 12q24.22 tescalcin INHA 0.465 2q35 inhibin, alpha S100P 0.461 4p16 S100 calcium binding protein P VPS37A 0.447 8p22 vacuolar protein sorting 37 homolog A (S. cerevisiae) SLC16A14 0.447 2q36.3 solute carrier family 16, member 14 PPARGC1A 0.443 4p15.1 peroxisome proliferator-activated receptor gamma, coactivator 1 alpha SIK1 0.435 21q22.3 salt-inducible kinase 1 IRS2 0.434 13q34 insulin receptor substrate 2 RND1 0.433 12q12 Rho family GTPase 1 HGD 0.433 3q13.33 homogentisate 1,2-dioxygenase PTP4A1 0.432 6q12 protein tyrosine phosphatase type IVA, member 1 C8orf4 0.428 8p11.2 chromosome 8 open reading frame 4 DDC 0.427 7p12.2 dopa decarboxylase (aromatic L-amino acid decarboxylase) TACC2 0.427 10q26 transforming, acidic coiled-coil containing protein 2 MUC13 0.422 3q21.2 mucin 13, cell surface associated C5 0.412 9q33-q34 complement component 5 NR4A2 0.412 2q22-q23 nuclear receptor subfamily 4, group A, member 2 EYS 0.411 6q12 eyes shut homolog (Drosophila) GPX2 0.406 14q24.1 glutathione peroxidase -

Anti-AHCY / Adenosylhomocysteinase Antibody (ARG57333)
Product datasheet [email protected] ARG57333 Package: 100 μl anti-AHCY / Adenosylhomocysteinase antibody Store at: -20°C Summary Product Description Rabbit Polyclonal antibody recognizes AHCY / Adenosylhomocysteinase Tested Reactivity Hu, Ms Tested Application WB Host Rabbit Clonality Polyclonal Isotype IgG Target Name AHCY / Adenosylhomocysteinase Antigen Species Human Immunogen Recombinant Protein of Human AHCY / Adenosylhomocysteinase. Conjugation Un-conjugated Alternate Names adoHcyase; AdoHcyase; SAHH; Adenosylhomocysteinase; EC 3.3.1.1; S-adenosyl-L-homocysteine hydrolase Application Instructions Application table Application Dilution WB 1:500 - 1:2000 Application Note * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Positive Control SW480 Calculated Mw 48 kDa Properties Form Liquid Purification Affinity purification with immunogen. Buffer PBS (pH 7.3), 0.02% Sodium azide and 50% Glycerol. Preservative 0.02% Sodium azide Stabilizer 50% Glycerol Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. Note For laboratory research only, not for drug, diagnostic or other use. www.arigobio.com 1/2 Bioinformation Gene Symbol AHCY Gene Full Name adenosylhomocysteinase Background S-adenosylhomocysteine hydrolase belongs to the adenosylhomocysteinase family. It catalyzes the reversible hydrolysis of S-adenosylhomocysteine (AdoHcy) to adenosine (Ado) and L-homocysteine (Hcy). Thus, it regulates the intracellular S-adenosylhomocysteine (SAH) concentration thought to be important for transmethylation reactions. -

A State of the Art of Antioxidant Properties of Curcuminoids in Neurodegenerative Diseases
International Journal of Molecular Sciences Review A State of the Art of Antioxidant Properties of Curcuminoids in Neurodegenerative Diseases Serena Silvestro † , Cinzia Sindona †, Placido Bramanti and Emanuela Mazzon * IRCCS Centro Neurolesi “Bonino-Pulejo”, Via Provinciale Palermo, Contrada Casazza, 98124 Messina, Italy; [email protected] (S.S.); [email protected] (C.S.); [email protected] (P.B.) * Correspondence: [email protected]; Tel.: +39-090-60128172 † These authors contributed equally as first author. Abstract: Neurodegenerative diseases represent a set of pathologies characterized by an irreversible and progressive, and a loss of neuronal cells in specific areas of the brain. Oxidative phosphorylation is a source of energy production by which many cells, such as the neuronal cells, meet their energy needs. Dysregulations of oxidative phosphorylation induce oxidative stress, which plays a key role in the onset of neurodegenerative diseases such as Alzheimer’s disease (AD), Parkinson’s disease (PD), and amyotrophic lateral sclerosis (ALS). To date, for most neurodegenerative diseases, there are no resolute treatments, but only interventions capable of alleviating the symptoms or slowing the course of the disease. Therefore, effective neuroprotection strategies are needed. In recent years, natural products, such as curcuminoids, have been intensively explored and studied for their therapeutic potentials in several neurodegenerative diseases. Curcuminoids are, nutraceutical compouns, that owen several therapeutic properties such as anti-oxidant, anti-inflammatory and neuroprotective effects. In this context, the aim of this review was to provide an overview of preclinical and clinical Citation: Silvestro, S.; Sindona, C.; evidence aimed to illustrate the antioxidant effects of curcuminoids in neurodegenerative diseases. -

Prioritization of Metabolic Genes As Novel Therapeutic Targets in Estrogen-Receptor Negative Breast Tumors Using Multi-Omics Data and Text Mining
www.oncotarget.com Oncotarget, 2019, Vol. 10, (No. 39), pp: 3894-3909 Research Paper Prioritization of metabolic genes as novel therapeutic targets in estrogen-receptor negative breast tumors using multi-omics data and text mining Dinesh Kumar Barupal1,*, Bei Gao1,*, Jan Budczies2, Brett S. Phinney4, Bertrand Perroud4, Carsten Denkert2,3 and Oliver Fiehn1 1West Coast Metabolomics Center, University of California, Davis, CA, USA 2Institute of Pathology, Charité University Hospital, Berlin, Germany 3German Institute of Pathology, Philipps-University Marburg, Marburg, Germany 4UC Davis Genome Center, University of California, Davis, CA, USA *Co-first authors and contributed equally to this work Correspondence to: Oliver Fiehn, email: [email protected] Keywords: set-enrichment; ChemRICH; multi-omics; metabolic networks; candidate gene prioritization Received: March 12, 2019 Accepted: May 13, 2019 Published: June 11, 2019 Copyright: Barupal et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC BY 3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. ABSTRACT Estrogen-receptor negative (ERneg) breast cancer is an aggressive breast cancer subtype in the need for new therapeutic options. We have analyzed metabolomics, proteomics and transcriptomics data for a cohort of 276 breast tumors (MetaCancer study) and nine public transcriptomics datasets using univariate statistics, meta- analysis, Reactome pathway analysis, biochemical network mapping and text mining of metabolic genes. In the MetaCancer cohort, a total of 29% metabolites, 21% proteins and 33% transcripts were significantly different (raw p <0.05) between ERneg and ERpos breast tumors. -

Role of GSH and Iron-Sulfur Glutaredoxins in Iron Metabolism—Review
molecules Review Role of GSH and Iron-Sulfur Glutaredoxins in Iron Metabolism—Review 1, 1, 1 1 Trnka Daniel y , Hossain Md Faruq y , Jordt Laura Magdalena , Gellert Manuela and Lillig Christopher Horst 2,* 1 Institute for Medical Biochemistry and Molecular Biology, University Medicine, University of Greifswald, 17475 Greifswald, Germany; [email protected] (T.D.); [email protected] (H.M.F.); [email protected] (J.L.M.); [email protected] (G.M.) 2 Christopher Horst Lillig, Institute for Medical Biochemistry and Molecular Biology, University Medicine Greifswald, Ferdinand-Sauerbruch-Straße, 17475 Greifswald, Germany * Correspondence: [email protected]; Tel.: +49-3834-865407; Fax: +49-3834-865402 These authors contributed equally to this work. y Academic Editor: Pál Perjési Received: 29 July 2020; Accepted: 22 August 2020; Published: 25 August 2020 Abstract: Glutathione (GSH) was initially identified and characterized for its redox properties and later for its contributions to detoxification reactions. Over the past decade, however, the essential contributions of glutathione to cellular iron metabolism have come more and more into focus. GSH is indispensable in mitochondrial iron-sulfur (FeS) cluster biosynthesis, primarily by co-ligating FeS clusters as a cofactor of the CGFS-type (class II) glutaredoxins (Grxs). GSH is required for the export of the yet to be defined FeS precursor from the mitochondria to the cytosol. In the cytosol, it is an essential cofactor, again of the multi-domain CGFS-type Grxs, master players in cellular iron and FeS trafficking. In this review, we summarize the recent advances and progress in this field. The most urgent open questions are discussed, such as the role of GSH in the export of FeS precursors from mitochondria, the physiological roles of the CGFS-type Grx interactions with BolA-like proteins and the cluster transfer between Grxs and recipient proteins. -

The Microbiota-Produced N-Formyl Peptide Fmlf Promotes Obesity-Induced Glucose
Page 1 of 230 Diabetes Title: The microbiota-produced N-formyl peptide fMLF promotes obesity-induced glucose intolerance Joshua Wollam1, Matthew Riopel1, Yong-Jiang Xu1,2, Andrew M. F. Johnson1, Jachelle M. Ofrecio1, Wei Ying1, Dalila El Ouarrat1, Luisa S. Chan3, Andrew W. Han3, Nadir A. Mahmood3, Caitlin N. Ryan3, Yun Sok Lee1, Jeramie D. Watrous1,2, Mahendra D. Chordia4, Dongfeng Pan4, Mohit Jain1,2, Jerrold M. Olefsky1 * Affiliations: 1 Division of Endocrinology & Metabolism, Department of Medicine, University of California, San Diego, La Jolla, California, USA. 2 Department of Pharmacology, University of California, San Diego, La Jolla, California, USA. 3 Second Genome, Inc., South San Francisco, California, USA. 4 Department of Radiology and Medical Imaging, University of Virginia, Charlottesville, VA, USA. * Correspondence to: 858-534-2230, [email protected] Word Count: 4749 Figures: 6 Supplemental Figures: 11 Supplemental Tables: 5 1 Diabetes Publish Ahead of Print, published online April 22, 2019 Diabetes Page 2 of 230 ABSTRACT The composition of the gastrointestinal (GI) microbiota and associated metabolites changes dramatically with diet and the development of obesity. Although many correlations have been described, specific mechanistic links between these changes and glucose homeostasis remain to be defined. Here we show that blood and intestinal levels of the microbiota-produced N-formyl peptide, formyl-methionyl-leucyl-phenylalanine (fMLF), are elevated in high fat diet (HFD)- induced obese mice. Genetic or pharmacological inhibition of the N-formyl peptide receptor Fpr1 leads to increased insulin levels and improved glucose tolerance, dependent upon glucagon- like peptide-1 (GLP-1). Obese Fpr1-knockout (Fpr1-KO) mice also display an altered microbiome, exemplifying the dynamic relationship between host metabolism and microbiota. -

Differential Effects on Growth, Homocysteine, and Related
(CANCER RESEARCH 49, 324-330, January 15, 1989] Differential Effects on Growth, Homocysteine, and Related Compounds of Two Inhibitors of 5-AdenosyIhomocysteinc Catabolism, 3-Deazaadenosine, and 3-Deazaaristeromycin, in C3H/10T1/2 Cells1 Rune Djurhuus,2 Asbjorn M. Svardal,3 and Per M. Ueland Clinical Pharmacology Unit, Department of Pharmacology and Toxicology, University of Bergen, Norway ABSTRACT logues were shown to interfere with the function of AdoHcy hydrolase, and this enzyme was used as a target for design of The growth of nontransformed (Cl 8) and malignant (C) 16) C3H/ biological active compounds (3, 4). c3Ari is a result of such Kll 1/2 mouse embryo fibroblasts was inhibited by 3-deazaadenosine (c'Ailii) (I.I)«,= 195 MMfor Cl 8 and 30 MMfor CI 16 cells) and 3- rational design (5). deazaaristeromycin (c'Ari) (LDM about 36 MMfor Cl 8 and 9 MMfor Cl Adenosine analogues may function as competitive inhibitor, 16 cells). Both compounds inhibited in a dose-dependent manner .V- substrate, or inactivator (irreversible inhibitor) of AdoHcy hy adenosylhomocysteine (AdoHcy) catabolisnt and homocysteine produc drolase (2). In principle, the metabolic derangements induced tion, measured as homocysteine egress, and c'Ari was most potent in this by agents interacting with this enzyme can be divided into two respect. c'Ad»gave rise to its congener, 3-deazaadenosylhomocysteine types: (c3AdoHcy). Addition of homocysteine thiolactone (Hcy-tl) to the medium (a) Inhibition of AdoHcy catabolism results in accumulation enhanced AdoHcy (and c3AdoHcy) accumulation but did not affect the of large amount of this metabolite, and nucleosides serving as cell growth at concentrations of inhibitor less than 10 MM. -

Ab197002 Adenosylhomocysteinase (AHCY) Activity Assay Kit (Fluorometric)
ab197002 Adenosylhomocysteinase (AHCY) Activity Assay Kit (Fluorometric) Instructions for Use For the rapid, sensitive and accurate measurement of Adenosylhomocysteinase (AHCY) activity in various samples. This product is for research use only and is not intended for diagnostic use. Version 1 Last Updated 5 September 2017 Table of Contents INTRODUCTION 1. BACKGROUND 2 2. ASSAY SUMMARY 3 GENERAL INFORMATION 3. PRECAUTIONS 4 4. STORAGE AND STABILITY 4 5. MATERIALS SUPPLIED 5 6. MATERIALS REQUIRED, NOT SUPPLIED 5 7. LIMITATIONS 6 8. TECHNICAL HINTS 7 ASSAY PREPARATION 9. REAGENT PREPARATION 8 10. STANDARD PREPARATION 10 11. SAMPLE PREPARATION 11 ASSAY PROCEDURE and DETECTION 12. ASSAY PROCEDURE and DETECTION 13 DATA ANALYSIS 13. CALCULATIONS 15 14. TYPICAL DATA 17 RESOURCES 15. QUICK ASSAY PROCEDURE 19 16. TROUBLESHOOTING 20 17. FAQ 22 18. INTERFERENCES 23 19. NOTES 24 Discover more at www.abcam.com 1 INTRODUCTION 1. BACKGROUND Adenosylhomocysteinase Activity Assay Kit (ab197002) can kinetically measure AHCY activity by detecting adenosine generation resulting from the hydrolysis of SAH. Adenosine is detected via a multi-step reaction, resulting in the generation of an intermediate that reacts with the OxiRed Probe. The fluorescent product is measured at Ex/Em = 535/587 nm. Limit of quantification (L.O.Q) is 1 µU recombinant Human AHCY. Adenosylhomocysteinase (AHCY) (EC 3.3.1.1) or S- adenosylhomocysteine hydrolase (SAHH); is an enzyme that catalyzes the reversible hydrolysis of S-Adenosyl Homocysteine (SAH) to adenosine and homocysteine. AHCY regulates the intracellular SAH concentration which in turn regulates S-adenosyl methionine (SAM)- dependent methyltransferases. Down-regulation of AHCY has been associated with certain forms of cancer and Huntington’s disease, while in Wilson’s disease; the enzyme is inhibited by the accumulated copper. -

Proteomics Reveals the Preliminary Physiological States of the Spotted
www.nature.com/scientificreports OPEN Proteomics reveals the preliminary physiological states of the spotted seal (Phoca largha) pups Jiashen Tian1, Jing Du1, Jiabo Han1, Xiangbo Bao1, Xinran Song2 & Zhichuang Lu1* Spotted seal (Phoca largha) is a critically endangered pinniped in China and South Korea. The conventional method to protect and maintain the P. largha population is to keep them captive in artifcially controlled environments. However, little is known about the physiological diferences between wild and captive P. largha. To generate a preliminary protein expression profle for P. largha, whole blood from wild and captive pups were subjected to a label-free comparative proteomic analysis. According to the results, 972 proteins were identifed and predicted to perform functions related to various metabolic, immune, and cellular processes. Among the identifed proteins, the expression level of 51 were signifcantly diferent between wild and captive P. large pups. These diferentially expressed proteins were enriched in a wide range of cellular functions, including cytoskeleton, phagocytosis, proteolysis, the regulation of gene expression, and carbohydrate metabolism. The abundances of proteins involved in phagocytosis and ubiquitin-mediated proteolysis were signifcantly higher in the whole blood of wild P. largha pups than in captive individuals. In addition, heat shock protein 90-beta, were determined as the key protein associated with the diferences in the wild and captive P. largha pups due to the most interactions of it with various diferentially expressed proteins. Moreover, wild P. largha pups could be more nutritionally stressed and have more powerful immune capacities than captive pups. This study provides the frst data on the protein composition of P. -
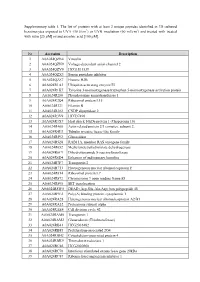
Supplementary Table 1. the List of Proteins with at Least 2 Unique
Supplementary table 1. The list of proteins with at least 2 unique peptides identified in 3D cultured keratinocytes exposed to UVA (30 J/cm2) or UVB irradiation (60 mJ/cm2) and treated with treated with rutin [25 µM] or/and ascorbic acid [100 µM]. Nr Accession Description 1 A0A024QZN4 Vinculin 2 A0A024QZN9 Voltage-dependent anion channel 2 3 A0A024QZV0 HCG1811539 4 A0A024QZX3 Serpin peptidase inhibitor 5 A0A024QZZ7 Histone H2B 6 A0A024R1A3 Ubiquitin-activating enzyme E1 7 A0A024R1K7 Tyrosine 3-monooxygenase/tryptophan 5-monooxygenase activation protein 8 A0A024R280 Phosphoserine aminotransferase 1 9 A0A024R2Q4 Ribosomal protein L15 10 A0A024R321 Filamin B 11 A0A024R382 CNDP dipeptidase 2 12 A0A024R3V9 HCG37498 13 A0A024R3X7 Heat shock 10kDa protein 1 (Chaperonin 10) 14 A0A024R408 Actin related protein 2/3 complex, subunit 2, 15 A0A024R4U3 Tubulin tyrosine ligase-like family 16 A0A024R592 Glucosidase 17 A0A024R5Z8 RAB11A, member RAS oncogene family 18 A0A024R652 Methylenetetrahydrofolate dehydrogenase 19 A0A024R6C9 Dihydrolipoamide S-succinyltransferase 20 A0A024R6D4 Enhancer of rudimentary homolog 21 A0A024R7F7 Transportin 2 22 A0A024R7T3 Heterogeneous nuclear ribonucleoprotein F 23 A0A024R814 Ribosomal protein L7 24 A0A024R872 Chromosome 9 open reading frame 88 25 A0A024R895 SET translocation 26 A0A024R8W0 DEAD (Asp-Glu-Ala-Asp) box polypeptide 48 27 A0A024R9E2 Poly(A) binding protein, cytoplasmic 1 28 A0A024RA28 Heterogeneous nuclear ribonucleoprotein A2/B1 29 A0A024RA52 Proteasome subunit alpha 30 A0A024RAE4 Cell division cycle 42 31