How Many Neurons Do You Have? Some Dogmas of Quantitative Neuroscience Under Revision
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

In Vivo Imaging of Microglia-Mediated Axonal Pruning and Modulation By
Combined bioRxivsingle preprintmanuscript doi: https://doi.org/10.1101/2020.06.07.087221 file ; this version posted June 8, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1 In vivo imaging of microglia-mediated axonal pruning and modulation 2 by the complement system 3 Tony K.Y. Lim1 and Edward S. Ruthazer1,2,* 4 1. Department of Neurology & Neurosurgery, Montreal Neurological Institute-Hospital, McGill 5 University, Montreal, Quebec, H3A 2B4; Canada 6 2. Lead Contact 7 *Correspondence: [email protected] 8 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.06.07.087221; this version posted June 8, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 9 Summary 10 Partial phagocytosis – called trogocytosis – of axons by microglia has been documented in ex vivo 11 preparations but has yet to be observed in vivo. Fundamental questions regarding the mechanisms that 12 modulate axon trogocytosis as well as its function in neural circuit development remain unanswered. 13 Here we used 2-photon live imaging of the developing Xenopus laevis retinotectal circuit to observe 14 axon trogocytosis by microglia in vivo. Amphibian regulator of complement activation 3 (aRCA3) was 15 identified as a neuronally expressed, synapse-associated complement inhibitory molecule. 16 Overexpression of aRCA3 enhanced axonal arborization and inhibited trogocytosis, while expression of 17 VAMP2-C3, a complement-enhancing fusion protein tethered to the axon surface, reduced axonal 18 arborization. -

Oligodendrocytes in Development, Myelin Generation and Beyond
cells Review Oligodendrocytes in Development, Myelin Generation and Beyond Sarah Kuhn y, Laura Gritti y, Daniel Crooks and Yvonne Dombrowski * Wellcome-Wolfson Institute for Experimental Medicine, Queen’s University Belfast, Belfast BT9 7BL, UK; [email protected] (S.K.); [email protected] (L.G.); [email protected] (D.C.) * Correspondence: [email protected]; Tel.: +0044-28-9097-6127 These authors contributed equally. y Received: 15 October 2019; Accepted: 7 November 2019; Published: 12 November 2019 Abstract: Oligodendrocytes are the myelinating cells of the central nervous system (CNS) that are generated from oligodendrocyte progenitor cells (OPC). OPC are distributed throughout the CNS and represent a pool of migratory and proliferative adult progenitor cells that can differentiate into oligodendrocytes. The central function of oligodendrocytes is to generate myelin, which is an extended membrane from the cell that wraps tightly around axons. Due to this energy consuming process and the associated high metabolic turnover oligodendrocytes are vulnerable to cytotoxic and excitotoxic factors. Oligodendrocyte pathology is therefore evident in a range of disorders including multiple sclerosis, schizophrenia and Alzheimer’s disease. Deceased oligodendrocytes can be replenished from the adult OPC pool and lost myelin can be regenerated during remyelination, which can prevent axonal degeneration and can restore function. Cell population studies have recently identified novel immunomodulatory functions of oligodendrocytes, the implications of which, e.g., for diseases with primary oligodendrocyte pathology, are not yet clear. Here, we review the journey of oligodendrocytes from the embryonic stage to their role in homeostasis and their fate in disease. We will also discuss the most common models used to study oligodendrocytes and describe newly discovered functions of oligodendrocytes. -

Build a Neuron
Build a Neuron Objectives: 1. To understand what a neuron is and what it does 2. To understand the anatomy of a neuron in relation to function This activity is great for ALL ages-even college students!! Materials: pipe cleaners (2 full size, 1 cut into 3 for each student) pony beads (6/student Introduction: Little kids: ask them where their brain is (I point to my head and torso areas till they shake their head yes) Talk about legos being the building blocks for a tower and relate that to neurons being the building blocks for your brain and that neurons send messages to other parts of your brain and to and from all your body parts. Give examples: touch from body to brain, movement from brain to body. Neurons are the building blocks of the brain that send and receive messages. Neurons come in all different shapes. Experiment: 1. First build soma by twisting a pipe cleaner into a circle 2. Then put a 2nd pipe cleaner through the circle and bend it over and twist the two strands together to make it look like a lollipop (axon) 3. take 3 shorter pipe cleaners attach to cell body to make dendrites 4. add 6 beads on the axon making sure there is space between beads for the electricity to “jump” between them to send the signal super fast. (myelin sheath) 5. Twist the end of the axon to make it look like 2 feet for the axon terminal. 6. Make a brain by having all of the neurons “talk” to each other (have each student hold their neuron because they’ll just throw them on a table for you to do it.) messages come in through the dendrites and if its a strong enough electrical change, then the cell body sends the Build a Neuron message down it’s axon where a neurotransmitter is released. -

Microglia Control Glutamatergic Synapses in the Adult Mouse Hippocampus
bioRxiv preprint doi: https://doi.org/10.1101/2021.02.01.429096; this version posted February 2, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Microglia control glutamatergic synapses in the adult mouse hippocampus Short title: Microglia and glutamatergic synapses Bernadette Basilico1†*‡, Laura Ferrucci1‡, Patrizia Ratano2‡, Maria T. Golia1, Alfonso Grimaldi3, Maria Rosito3, Valentina Ferretti4, Ingrid Reverte1,5, Maria C. Marrone6, Maria Giubettini3,7, Valeria De Turris3, Debora Salerno3, Stefano Garofalo1, Marie-Kim St-Pierre8, Micael Carrier8, Massimiliano Renzi1, Francesca Pagani3, Marcello Raspa9, Ferdinando Scavizzi9, Cornelius T. Gross10, Silvia Marinelli5, Marie E. Tremblay8,11, Daniele Caprioli1,5, Laura Maggi1, Cristina Limatola1,2, Silvia Di Angelantonio1,3§, Davide Ragozzino1,5*§ 1Department of Physiology and Pharmacology, Sapienza University of Rome, Rome, Italy. 2IRCCS Neuromed, Via Atinese 18, 86077, Pozzilli, IS, Italy. 3Center for Life Nanoscience, Istituto Italiano di Tecnologia, Rome, Italy. 4Dipartimento di Biologia e Biotecnologie "Charles Darwin", Sapienza University of Rome, Rome, Italy. 5Santa Lucia Foundation (IRCCS Fondazione Santa Lucia), Rome, Italy. 6European Brain Research Institute-Rita Levi Montalcini, Rome, Italy. 7CrestOptics S.p.A., Via di Torre Rossa 66, 00165 Rome, Italy. 8Centre de Recherche du CHU de Québec, Axe Neurosciences Québec, QC, Canada; Département de médecine moléculaire, Université Laval Québec, QC, Canada. 9National Research Council, Institute of Biochemistry and Cell Biology (CNR- IBBC/EMMA/Infrafrontier/IMPC), International Campus “A. Buzzati-Traverso”, Monterotondo (Rome) Italy. -

Neuroglial Response to Neuron Injury. a Study Using Intraneural Injection of Ricinus Communis Agglutinin-60
J. Anat. (1989), 164, pp. 201-213 201 With 16 figures Printed in Great Britain Neuroglial response to neuron injury. A study using intraneural injection of ricinus communis agglutinin-60 E. A. LING, C. Y. WEN*, J. Y. SHIEH*, T. Y. YICK AND S. K. LEONG Department of Anatomy, Faculty of Medicine, National University of Singapore, Singapore 0511 and * Department of Anatomy, College of Medicine, National Taiwan University, Taipei, Taiwan 10018 (Accepted 27 September 1988) INTRODUCTION Several studies have shown that ricinus communis agglutinin (RCA), when injected into a nerve in minute amounts, is retrogradely transported by axons in the nerve, resulting in a selective destruction of the parental cell bodies. Thus, the administration of RCA into the vagus nerve would cause a selective destruction of the efferent neurons in the dorsal motor nucleus (Wiley, Blessing & Reis, 1982; Ling & Leong, 1987, 1988). Such a 'suicide transport' of the toxic lectin is also evident in sensory neurons (Yamamoto, Iwasaki & Konno, 1983, 1984; Johnson, Westrum, Henry & Canfield, 1985; Wiley & Oeltmann, 1986). Recently, the use of RCA has become increasingly important as a research tool for tracing the central projections of primary afferents of peripheral nerves (Yamamoto et al. 1983; Leong & Tan, 1987; Ling & Leong, 1987). While much is known about the consequent death of neurons following RCA application, little is known about the response of the non-neuronal cells either closely associated with, or in the vicinity of, the degenerating neurons. According to Yamamoto et al. (1984), the selective destruction of neurons in the trigeminal and dorsal root ganglia by RCA could possibly stimulate the capsule cells involved in the phagocytosis of the degenerating nerve cells. -

University International
INFORMATION TO USERS This was produced from a copy of a document sent to us for microfilming. While the most advanced technological means to photograph and reproduce this document have been used, the quality is heavily dependent upon the quality of the material submitted. The following explanation of techniques is provided to help you understand markings or notations which may appear on this reproduction. 1. The sign or “target” for pages apparently lacking from the document photographed is “Missing Page(s)”. If it was possible to obtain the missing page(s) or section, they are spliced into the film along with adjacent pages. This may have necessitated cutting through an image and duplicating adjacent pages to assure you of complete continuity. 2. When an image on the film is obliterated with a round black mark it is an indication that the film inspector noticed either blurred copy because of movement during exposure, or duplicate copy. Unless we meant to delete copyrighted materials that should not have been filmed, you will find a good image of the page in the adjacent frame. 3. When a map, drawing or chart, etc., is part of the material being photo graphed the photographer has followed a definite method in “sectioning” the material. It is customary to begin filming at the upper left hand corner of a large sheet and to continue from left to right in equal sections with small overlaps. If necescary, sectioning is continued again—beginning below the first row and continuing on until complete. 4. For any illustrations that cannot be reproduced satisfactorily by xerography, photographic prints can be purchased at additional cost and tipped into your xerographic copy. -

Neurons – Is a Basic Cell of the Nervous System. • Neurons Carry Nerve Messages, Or Impulses, from One Part of the Body to Another
Nervous System Nerves and Nerve Cells: Neurons – is a basic cell of the nervous system. • Neurons carry nerve messages, or impulses, from one part of the body to another. Structure of a Nerve Cell: A neuron has three basic parts: 1. Body – controls the cell’s growth 2. Axon – is a long thin fiber that carries impulses away from the cell body Myelin – is a fatty material that insulates the axon and increases the speed at which an impulse travels 3. Dendrites – are short, branching fibers that carry nerve impulses toward the cell body. • A nerve impulse begins when the dendrites are stimulated. The impulse travels along the dendrites to the cell body, and then away from the cell body on the axon. • The impulse must cross a synapse to a muscle or another neuron. Synapse – is the space between an axon and the structure with which the neuron communicates. Types of Nerve Cells: Sensory Neurons – pick up information about your external and internal environment from your sense organs and your body Motor Neurons – sends impulses to your muscles and glands, causing them to react Interneurons – are located only in the brain and spinal cord, pass impulses from one neuron to another The Central Nervous System: • The nervous system consists of two parts. Your brain and spinal cord make up one part, which is called the central nervous system. 1 • The peripheral nervous system, which is the other part, is made up of all the nerves that connect the brain and spinal cord to other parts of the body. The Brain: Brain ± a moist, spongy organ weighing about three pounds is made up of billions of neurons that control almost everything you do and experience. -

The Functional Organization of Descending Sensory-Motor 2 Pathways in Drosophila 3 4 5 Shigehiro Namiki,1 Michael H
bioRxiv preprint doi: https://doi.org/10.1101/231696; this version posted December 11, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1 The functional organization of descending sensory-motor 2 pathways in Drosophila 3 4 5 Shigehiro Namiki,1 Michael H. Dickinson,2 Allan M. Wong,1 Wyatt Korff,1 Gwyneth M. 6 Card,1,* 7 8 1Janelia Research Campus, Howard Hughes Medical Institute, Ashburn, VA 20147, USA 9 2Division of Biology and Bioengineering, California Institute of Technology, Pasadena, CA 91125, USA 10 11 12 This manuscript includes 55 pages of typescript, 15 figures, 0 tables, 24 supplemental figures, and 6 13 supplementary tables. 14 15 KEYWORDS: command neuron; descending neuron; motor control; neuron database; ventral 16 nervous system 17 18 *Correspondence should be addressed to [email protected] (G.M.C) 19 1 bioRxiv preprint doi: https://doi.org/10.1101/231696; this version posted December 11, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 20 SUMMARY 21 22 In most animals, the brain controls the body via a set of descending neurons (DNs) that traverse the neck 23 and terminate in post-cranial regions of the nervous system. This critical neural population is thought to 24 activate, maintain and modulate locomotion and other behaviors. Although individual members of this 25 cell class have been well-studied across species ranging from insects to primates, little is known about the 26 overall connectivity pattern of DNs as a population. -
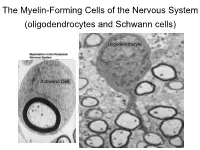
The Myelin-Forming Cells of the Nervous System (Oligodendrocytes and Schwann Cells)
The Myelin-Forming Cells of the Nervous System (oligodendrocytes and Schwann cells) Oligodendrocyte Schwann Cell Oligodendrocyte function Saltatory (jumping) nerve conduction Oligodendroglia PMD PMD Saltatory (jumping) nerve conduction Investigating the Myelinogenic Potential of Individual Oligodendrocytes In Vivo Sparse Labeling of Oligodendrocytes CNPase-GFP Variegated expression under the MBP-enhancer Cerebral Cortex Corpus Callosum Cerebral Cortex Corpus Callosum Cerebral Cortex Caudate Putamen Corpus Callosum Cerebral Cortex Caudate Putamen Corpus Callosum Corpus Callosum Cerebral Cortex Caudate Putamen Corpus Callosum Ant Commissure Corpus Callosum Cerebral Cortex Caudate Putamen Piriform Cortex Corpus Callosum Ant Commissure Characterization of Oligodendrocyte Morphology Cerebral Cortex Corpus Callosum Caudate Putamen Cerebellum Brain Stem Spinal Cord Oligodendrocytes in disease: Cerebral Palsy ! CP major cause of chronic neurological morbidity and mortality in children ! CP incidence now about 3/1000 live births compared to 1/1000 in 1980 when we started intervening for ELBW ! Of all ELBW {gestation 6 mo, Wt. 0.5kg} , 10-15% develop CP ! Prematurely born children prone to white matter injury {WMI}, principle reason for the increase in incidence of CP ! ! 12 Cerebral Palsy Spectrum of white matter injury ! ! Macro Cystic Micro Cystic Gliotic Khwaja and Volpe 2009 13 Rationale for Repair/Remyelination in Multiple Sclerosis Oligodendrocyte specification oligodendrocytes specified from the pMN after MNs - a ventral source of oligodendrocytes -

Structural and Developmental Principles of Neuropil Assembly in C
Article Structural and developmental principles of neuropil assembly in C. elegans https://doi.org/10.1038/s41586-020-03169-5 Mark W. Moyle1, Kristopher M. Barnes2, Manik Kuchroo3, Alex Gonopolskiy3, Leighton H. Duncan1, Titas Sengupta1, Lin Shao1, Min Guo4, Anthony Santella2, Received: 21 April 2020 Ryan Christensen4, Abhishek Kumar5, Yicong Wu4, Kevin R. Moon6, Guy Wolf7, Accepted: 12 November 2020 Smita Krishnaswamy3,10, Zhirong Bao2,10, Hari Shroff4,5,10, William A. Mohler8,10 & Daniel A. Colón-Ramos1,5,9,10 ✉ Published online: xx xx xxxx Check for updates Neuropil is a fundamental form of tissue organization within the brain1, in which densely packed neurons synaptically interconnect into precise circuit architecture2,3. However, the structural and developmental principles that govern this nanoscale precision remain largely unknown4,5. Here we use an iterative data coarse-graining algorithm termed ‘difusion condensation’6 to identify nested circuit structures within the Caenorhabditis elegans neuropil, which is known as the nerve ring. We show that the nerve ring neuropil is largely organized into four strata that are composed of related behavioural circuits. The stratifed architecture of the neuropil is a geometrical representation of the functional segregation of sensory information and motor outputs, with specifc sensory organs and muscle quadrants mapping onto particular neuropil strata. We identify groups of neurons with unique morphologies that integrate information across strata and that create neural structures that cage the strata within the nerve ring. We use high resolution light-sheet microscopy7,8 coupled with lineage-tracing and cell-tracking algorithms9,10 to resolve the developmental sequence and reveal principles of cell position, migration and outgrowth that guide stratifed neuropil organization. -
11 Introduction to the Nervous System and Nervous Tissue
11 Introduction to the Nervous System and Nervous Tissue ou can’t turn on the television or radio, much less go online, without seeing some- 11.1 Overview of the Nervous thing to remind you of the nervous system. From advertisements for medications System 381 Yto treat depression and other psychiatric conditions to stories about celebrities and 11.2 Nervous Tissue 384 their battles with illegal drugs, information about the nervous system is everywhere in 11.3 Electrophysiology our popular culture. And there is good reason for this—the nervous system controls our of Neurons 393 perception and experience of the world. In addition, it directs voluntary movement, and 11.4 Neuronal Synapses 406 is the seat of our consciousness, personality, and learning and memory. Along with the 11.5 Neurotransmitters 413 endocrine system, the nervous system regulates many aspects of homeostasis, including 11.6 Functional Groups respiratory rate, blood pressure, body temperature, the sleep/wake cycle, and blood pH. of Neurons 417 In this chapter we introduce the multitasking nervous system and its basic functions and divisions. We then examine the structure and physiology of the main tissue of the nervous system: nervous tissue. As you read, notice that many of the same principles you discovered in the muscle tissue chapter (see Chapter 10) apply here as well. MODULE 11.1 Overview of the Nervous System Learning Outcomes 1. Describe the major functions of the nervous system. 2. Describe the structures and basic functions of each organ of the central and peripheral nervous systems. 3. Explain the major differences between the two functional divisions of the peripheral nervous system. -

Heterogeneity in Ventricular Zone Neural Precursors Contributes to Neuronal Fate Diversity in the Postnatal Neocortex
7028 • The Journal of Neuroscience, May 19, 2010 • 30(20):7028–7036 Development/Plasticity/Repair Heterogeneity in Ventricular Zone Neural Precursors Contributes to Neuronal Fate Diversity in the Postnatal Neocortex Elizabeth K. Stancik,1 Ivan Navarro-Quiroga,1 Robert Sellke,2 and Tarik F. Haydar1 1Center for Neuroscience Research, Children’s National Medical Center, Washington, DC 20010, and 2University of Maryland School of Medicine, Baltimore, Maryland 21201 The recent discovery of short neural precursors (SNPs) in the murine neocortical ventricular zone (VZ) challenges the widely held view that radial glial cells (RGCs) are the sole occupants of this germinal compartment and suggests that precursor variety is an important factor of brain development. Here, we use in utero electroporation and genetic fate mapping to show that SNPs and RGCs cohabit the VZ but display different cell cycle kinetics and generate phenotypically different progeny. In addition, we find that RGC progeny undergo additional rounds of cell division as intermediate progenitor cells (IPCs), whereas SNP progeny generally produce postmitotic neurons directly from the VZ. By clearly defining SNPs as bona fide VZ residents, separate from both RGCs and IPCs, and uncovering their unique proliferativeandlineageproperties,theseresultsdemonstratehowindividualneuralprecursorgroupsintheembryonicrodentVZcreate diversity in the overlying neocortex. Introduction progenitors properly form the cerebral cortex as well as for elu- The ventricular zone (VZ) of the dorsal telencephalon contains cidating possible mechanisms of species-specific diversity. the progenitor cells that produce all of the various excitatory In addition to the neural precursors in the VZ, a separate class neurons of the mature neocortex. This process occurs during of neuronal progenitors has been described in the overlying sub- prenatal mammalian brain development through a precisely reg- ventricular zone (SVZ).