Sin/Sio2 Passivation Stack of N-Type Silicon Surface
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Evaluation of Passivation Process for Stainless Steel Hypotubes Used in Coronary Angioplasty Technique
coatings Article Evaluation of Passivation Process for Stainless Steel Hypotubes Used in Coronary Angioplasty Technique Lucien Reclaru 1,2 and Lavinia Cosmina Ardelean 2,3,* 1 Scientific Independent Consultant Biomaterials and Medical Devices, 103 Paul-Vouga, 2074 Marin-Neuchâtel, Switzerland; [email protected] 2 Multidisciplinary Center for Research, Evaluation, Diagnosis and Therapies in Oral Medicine, “Victor Babes” University of Medicine and Pharmacy Timisoara, 2 Eftimie Murgu sq, 300041 Timisoara, Romania 3 Department of Technology of Materials and Devices in Dental Medicine, “Victor Babes” University of Medicine and Pharmacy Timisoara, 2 Eftimie Murgu sq, 300041 Timisoara, Romania * Correspondence: [email protected] Abstract: In the manufacturing of hypotubes for coronary applications, austenitic steels of types 304, 304, or 316 L are being used. The manufacturing process involves bending steel strips into tubes and the continuous longitudinal welding of the tubes. Manufacturing also includes heat treatments and stretching operations to achieve an external/internal diameter of 0.35/0.23 mm, with a tolerance of +/− 0.01 mm. Austenitic steels are sensitive to localized corrosion (pitting, crevice, and intergranular) that results from the welding process and various heat treatments. An extremely important step is the cleaning and the internal and external passivation of the hypotube surface. During patient interventions, there is a high risk of metal cations being released in contact with human blood. The aim of this study was to evaluate the state of passivation and corrosion resistance by using electrochemical methods and specific intergranular corrosion tests (the Strauss test). There were difficulties in passivating the hypotubes and assessing the corrosion phenomena in the interior of the tubes. -

Si Passivation and Chemical Vapor Deposition of Silicon Nitride: Final Technical Report, March 18, 2007
A national laboratory of the U.S. Department of Energy Office of Energy Efficiency & Renewable Energy National Renewable Energy Laboratory Innovation for Our Energy Future Si Passivation and Chemical Subcontract Report NREL/SR-520-42325 Vapor Deposition of Silicon Nitride November 2007 Final Technical Report March 18, 2007 H.A. Atwater California Institute of Technology Pasadena, California NREL is operated by Midwest Research Institute ● Battelle Contract No. DE-AC36-99-GO10337 Si Passivation and Chemical Subcontract Report NREL/SR-520-42325 Vapor Deposition of Silicon Nitride November 2007 Final Technical Report March 18, 2007 H.A. Atwater California Institute of Technology Pasadena, California NREL Technical Monitor: R. Matson/F. Posey-Eddy Prepared under Subcontract No. AAT-2-31605-01 National Renewable Energy Laboratory 1617 Cole Boulevard, Golden, Colorado 80401-3393 303-275-3000 • www.nrel.gov Operated for the U.S. Department of Energy Office of Energy Efficiency and Renewable Energy by Midwest Research Institute • Battelle Contract No. DE-AC36-99-GO10337 This publication was reproduced from the best available copy Submitted by the subcontractor and received no editorial review at NREL NOTICE This report was prepared as an account of work sponsored by an agency of the United States government. Neither the United States government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product, or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States government or any agency thereof. -

What Is the Value of Water Contact Angle on Silicon?
materials Article What Is the Value of Water Contact Angle on Silicon? Paweł Bryk 1, Emil Korczeniewski 2, Grzegorz S. Szyma ´nski 2, Piotr Kowalczyk 3, Konrad Terpiłowski 4 and Artur P. Terzyk 2,* 1 Department of Chemistry, Chair of Theoretical Chemistry, Maria Curie-Skłodowska University, 20-031 Lublin, Poland; [email protected] 2 Faculty of Chemistry, Physicochemistry of Carbon Materials Research Group, Nicolaus Copernicus University in Toru´n,Gagarin Street 7, 87-100 Toru´n,Poland; [email protected] (E.K.); [email protected] (G.S.S.) 3 College of Science, Health, Engineering and Education, Murdoch University, Murdoch WA 6150, Australia; [email protected] 4 Department of Chemistry, Chair of Physical Chemistry of Interfacial Phenomena, Maria Curie-Skłodowska University, 20-031 Lublin, Poland; [email protected] * Correspondence: [email protected]; Tel.: +48-56-61-14-371 Received: 4 March 2020; Accepted: 26 March 2020; Published: 27 March 2020 Abstract: Silicon is a widely applied material and the wetting of silicon surface is an important phenomenon. However, contradictions in the literature appear considering the value of the water contact angle (WCA). The purpose of this study is to present a holistic experimental and theoretical approach to the WCA determination. To do this, we checked the chemical composition of the silicon (1,0,0) surface by using the X-ray photoelectron spectroscopy (XPS) method, and next this surface was purified using different cleaning methods. As it was proved that airborne hydrocarbons change a solid wetting properties the WCA values were measured in hydrocarbons atmosphere. Next, molecular dynamics (MD) simulations were performed to determine the mechanism of wetting in this atmosphere and to propose the force field parameters for silica wetting simulation. -

Analysis of the Silicon Dioxide Passivation and Forming Gas
&RQIHUHQFH3URFHHGLQJV 6RODU:RUOG&RQJUHVV Daegu, Korea, 08 – 12 November 2015 Analysis of the Silicon Dioxide Passivation and Forming Gas Annealing in Silicon Solar Cells Izete Zanesco and Adriano Moehlecke Solar Energy Technology Nucleus (NT-Solar), Faculty of Physics, Pontifical Catholic University of Rio Grande do Sul (PUCRS), Porto Alegre (Brazil) Abstract The passivation of silicon solar cells by the deposition of SiNx anti-reflection coating is usual in the industry. However, materials such as SiO2, TiO2 and Al2O3 may be a cost-effective alternative and its analysis is mainly reported in silicon wafers. The goal of this paper is to present the development and analysis of silicon solar cells passivated with a thin layer of SiO2 as well as the evaluation of the effectiveness of the annealing step in forming gas. The dry oxidation was performed before the TiO2 anti-reflection coating deposition and the annealing in forming gas was performed in the same furnace. The temperature and time of the oxidation and the annealing step were experimentally optimized. The efficiency of 15.9 %was achieved. The highest average efficiency was found in the oxidation temperature range from 750 ºC to 800 ºC, during 7 minutes, caused by the increasing of open circuit voltage and fill factor. At short wavelengths, the internal quantum efficiency decreases slightly with the increasing of the oxidation temperature. The minority carrier diffusion length (LD) of the solar cells processed with the oxidation temperature of 800 °C was around 1890 Pm. The open circuit voltage shows a slight trend of increasing with the oxidation time. The annealing step in forming gas did not improve the average efficiency of the solar cells. -

H2O2 Hydrogen Peroxide Passivation Procedure
Solvaytechnical Chemicals PUBLICATION H2O2 Passivation Procedure Introduction Hydrogen peroxide is a strong chemical oxidant which decomposes into water and oxygen in the presence of a catalytic quantity of any transition metal (e.g., iron, copper, nickel, etc.). The primary concern with decomposition is the buildup of pressure which can lead to pressure Prepare for passivation by roping off the work area bursts. To prevent this from occurring, any metal surface that comes in and posting warning signs. All open lights and tools contact with hydrogen peroxide must be degreased, pickled and which may spark must be removed from the passivated, even if only used once. The degreasing and pickling steps passivation area. Smoking is prohibited within the chemically clean the metal surfaces. The passivating step oxidizes the passivation area. Prior to preparation of the chemical metal surface. The thin oxide coating, which forms on the metal surface solutions, determine how to dispose of the spent during passivation, renders the surface nonreactive to hydrogen peroxide chemicals. These chemicals must be disposed of in and prevents the metal from decomposing the peroxide. a safe and environmentally sound manner that is consistent with all applicable federal, state, and The passivation procedure consists of: local regulations. 1. Grinding to remove weld spatter and smooth out scratches. 2. Degreasing to remove oil and grease films. Application methods 3. Pickling to chemically clean the surface. The chemical solutions may be applied to the metal surfaces by the four different methods listed below. 4. Passivating with nitric acid to form an oxide film. • The metal surfaces may be sprayed with the 5. -

MAGNESIUM SILICATE, Synthetic
MAGNESIUM SILICATE, synthetic Prepared at the 74th JECFA (2011) and published in FAO Monographs 11 (2011) superseding specifications prepared at the 61st JECFA (2003), published in the Combined Compendium of Food Additive Specifications, FAO JECFA Monographs 1 (2005). An ADI ‘not specified’ was established at the 25th JECFA (1981). SYNONYMS INS No. 553(i) DEFINITION Magnesium silicate (synthetic) is manufactured by the precipitation reaction between sodium silicate and a soluble magnesium salt. The aqueous suspension of the precipitate is filtered and the collected solid washed, dried, classified for particle size and packaged. The finest material is intended for use as an anticaking agent and the coarser particles are for use as a filtering aid. The moisture content of the material meant for use as an anticaking agent is kept to less than 15%. Although magnesium silicate is of variable composition, the molar ratio of MgO to SiO2 is approximately 2:5. Chemical name Magnesium silicate C.A.S. number 1343-88-0 Assay Not less than 15% of MgO and not less than 67% of SiO2, calculated on the ignited basis. DESCRIPTION Very fine, white, odourless powder, free from grittiness FUNCTIONAL USES Anticaking agent, filtering aid CHARACTERISTICS IDENTIFICATION Solubility (Vol. 4) Insoluble in water pH (Vol. 4) 7.0-11.0 (1 in 10 slurry) Magnesium (Vol. 4) Mix about 0.5 g of the sample with 10 ml of dilute hydrochloric acid TS, filter, and neutralize the filtrate to litmus paper with ammonia TS. The neutralized filtrate gives a positive test for magnesium. Silicate Prepare a bead by fusing a few crystals of sodium ammonium phosphate on a platinum loop in the flame of a Bunsen burner. -

Effect of the Electrochemical Passivation on the Corrosion Behaviour of Austenitic Stainless Steel
Effect of the electrochemical passivation on the corrosion behaviour of austenitic stainless steel A. Barbucci, M.Delucchi, M. Panizza, G, Farné, G. Cerisola DICheP, University of Genova, P.le Kennedy 1, 16129 Genova, Italy tel: +390103536030, e-mail: [email protected] Abstract: Cold rolled SS is also fruitfully used in deep drawing however the presence of scales or oxides on the surface reduces the life of the tools and emphasises creep phenomena of the material. Then a cleaning of the SS surface from these impurities is necessary. Oxides can be formed during the hot rolling preceding the cold one, or during the annealing performed between the several steps of thickness reduction. The annealing helps to decrease the work hardening occurring during the process. Normally this heat treatment is performed in reducing atmosphere of pure hydrogen (bright annealing), but even in this conditions oxides are formed on the SS surface. To avoid this uncontrolled oxide growth one method recently applied is an electrochemical cleaning performed in an electrolytic solution containing chrome, generally called electrochemical passivation. The electrochemical passivation allows the dissolution of the contaminating hard particles on the strips. Few scientific contributions are available in literature, which explain in detail the process mechanism. The aim of this work is to investigate if the electrochemical passivated surface acts in a different way with regard to corrosion phenomena with respect to conventional SS. Electrochemical measurements like polarisations, chronoamperometries and surface analysis were used to investigate the corrosion behaviour of electrochemically passivated AISI 304L and AISI 305. The effect of some process parameters were considered, too. -

Why Passivate Stainless Steel and What Happens If You Don't
Technical Article Page 1 of 4 Why Passivate Stainless Steel and What Happens If You Don’t By: Patrick H. Banes, Astro Pak Corporation To insure having the maximum corrosion resistance that austenitic stainless steel has to offer, the stainless steel surface must be in a passive state. The passive condition on the surface of the stainless steel is obtained by formation of a chromium oxide film on the metal‘s surface. This is accomplished by passivating the metal. Chemical passivation procedures using alkalines, wetting agents, chelants, and organic or inorganic acids are used to clean the metal surface thoroughly and in conjunction with an oxygen rich atmosphere, a uniform chromium oxide film forms. The key is to have a uniform chromium oxide film with an acceptable chrome to iron ratio (typically>1) and thickness (20-50) angstroms. Passivation is required to restore or enhance the chromium oxide film on the stainless steel surface when it has been manipulated by surface finishing, welding, grinding, external contamination, etc. Mechanical finishing of stainless steel surfaces allows for a very smooth surface and is a prerequisite for sub- sequent electro-polishing. The mechanical action disrupts the existing passive surface and results in a surface with a low chrome to iron ratio. The possibility of surface contamination getting pushed or smeared into the metal surface or sub-surface also exists. Welding stainless steel is one of the primary reasons for passivating. The metal surface is altered and free iron is released from the alloy. The chrome to iron ratio is also lowered in the heat affected zone of the weld. -
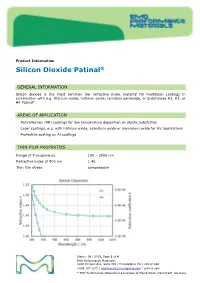
Sio2,Silicon Dioxide Patinal®Us
Product Information Silicon Dioxide Patinal ® GENERAL INFORMATION Silicon dioxide is the most common low refractive index material for multilayer coatings in combination with e.g. titanium oxide, hafnium oxide, tantalum pentoxide, or Substances H1, H2, or H4 Patinal . AREAS OF APPLICATION • Antireflection (AR) coatings for low temperature deposition on plastic substrates • Laser coatings, e.g. with hafnium oxide, scandium oxide or aluminium oxide for UV applications • Protective coating on Al-coatings THIN FILM PROPERTIES Range of Transparency 190 – 2000 nm Refractive index at 500 nm 1.46 Thin film stress compressive Status: 08 / 2019, Page 1 of 4 EMD Performance Materials* 1200 Intrepid Ave, Suite 300 / Philadelphia, PA / 19112 USA +888 367 3275 / [email protected] / patinal.com * EMD Performance Materials is a business of Merck KGaA, Darmstadt, Germany wavl / nm 200 250 350 500 650 800 1000 1250 n 1.551 1.531 1.487 1.470 1.463 1.461 1.458 1.457 k 7.1-04 2.6E-04 < 1E-4 < 1E-4 < 1E-4 < 1E-4 < 1E-4 < 1E-4 The resulting optical properties of the thin film are dependent on process conditions like deposition rate and substrate temperature. SiO 2 thin films exhibit an amorphous structure. High packing densities can be achieved by using IAD or a high substrate temperature. NOTES FOR EVAPORATION Evaporator source Electron beam evaporator Crucible / Boat Copper crucible Evaporation temperature 1800 – 2200 °C Deposition rate 0.4 – 1.2 nm/s Oxygen partial pressure none to ~5·10 -5 mbar IAD settings (Leybold APS) 130 – 150 V Bias Substrate temperature conventional RT to 350 °C IAD @ RT QCR-settings Density 2.648 g/cm³, z-ratio 1.0 The evaporation characteristics and the properties of the coating depend on the focusing of the electron beam. -

Brief Review of Surface Passivation on III-V Semiconductor
crystals Review Brief Review of Surface Passivation on III-V Semiconductor Lu Zhou 1, Baoxue Bo 2, Xingzhen Yan 1, Chao Wang 1, Yaodan Chi 1 and Xiaotian Yang 1,* 1 Jilin Provincial Key Laboratory of Architectural Electricity & Comprehensive Energy Saving, School of Electrical Engineering and Computer, Jilin Jianzhu University, Changchun 130118, China; [email protected] (L.Z.); [email protected] (X.Y.); [email protected] (C.W.); [email protected] (Y.C.) 2 National Key Laboratory on High Power Semiconductor Lasers, Changchun University of Science and Technology, Changchun 130022, China; [email protected] * Correspondence: [email protected] Received: 30 March 2018; Accepted: 9 May 2018; Published: 18 May 2018 Abstract: The III-V compound semiconductor, which has the advantage of wide bandgap and high electron mobility, has attracted increasing interest in the optoelectronics and microelectronics field. The poor electronic properties of III-V semiconductor surfaces resulting from a high density of surface/interface states limit III-V device technology development. Various techniques have been applied to improve the surface and interface quality, which cover sulfur-passivation, plasmas-passivation, ultrathin film deposition, and so on. In this paper, recent research of the surface passivation on III-V semiconductors was reviewed and compared. It was shown that several passivation methods can lead to a perfectly clean surface, but only a few methods can be considered for actual device integration due to their effectiveness and simplicity. Keywords: III-V semiconductor; passivation; MOCAP; ALD; XPS 1. Introduction III-V semiconductor materials, such as GaAs, InGaAs, GaSb, InP, InAs, InSb, have high surface state density (>1013 cm−2) lacking a high quality intrinsic oxide passivation layer, which lead to Fermi level pinning, surface recombination accelerating, and therefore deteriorate the device performance in microelectronic and optoelectronic fields. -

(Quartz) Silica, Sio2, Is a White Or Colorless Crystalline Compound
Silica (quartz) Silica, SiO2, is a white or colorless crystalline compound found mainly as quartz, sand, flint, and many other minerals. Silica is an important ingredient to manufacture a wide variety of materials. Quartz; Quartz is the most abundant silica mineral. Pure Quartz is colorless and transparent. It occurs in most igneous and practically all metamorphic and sedimentary rocks. It is used as a component of numerous industrial materials. Silicon (Si) has the atomic number 14 and is closely related to carbon. It is a relatively inert metalloid. Silicon is often used for microchips, glass, cement, and pottery. Silica is the most abundant mineral found in the crust of the earth. One of the most common uses of silica quarts is the manufacturer of glass. Silica is the fourteenth element on the periodic table. It can sometimes be found as the substance, quartz which is usually used in jewelry, test tubes, and when placed under pressure, generates an electrical charge. Quartz is the second most abundant mineral in the Earth's crust. It is a clear, glossy mineral with a hardness of 7 on the MOHS scale. Silica, Sa,is a component of glass and concrete. A form of Silica commonly known as quartz, Silica tetrahedra, is the second most common mineral in the earth's crust, it comes in many different forms. Silica is a compound of silicon and oxygen. Earth's outer crust contains 59% of this material. It has three major rock forms, which are quartz, tridymite, and cristobalite. Silica, commonly known in the form of quartz, is the dioxide form of silicon, SiO2. -

Wettability of Silicon, Silicon Dioxide, and Organosilicate Glass
WETTABILITY OF SILICON, SILICON DIOXIDE, AND ORGANOSILICATE GLASS Nelson Martinez, B.S. Thesis Prepared for the Degree of MASTER OF SCIENCE UNIVERSITY OF NORTH TEXAS December 2009 APPROVED: Richard F. Reidy, Major Professor and Interim Chair of the Department of Materials Science and Engineering Witold Brostow, Committee Member Jincheng Du, Committee Member Srinivasan G. Srivilliputhur, Committee Member Costas Tsatsoulis, Dean of the College of Engineering Michael Monticino, Dean of the Robert B. Toulouse School of Graduate Studies Martinez, Nelson. Wettability of Silicon, Silicon Dioxide, and Organosilicate Glass. Master of Science (Materials Science and Engineering), December 2009, 106 pp., 26 tables, 48 illustrations, references 88 titles. Wetting of a substance has been widely investigated since it has many applications to many different fields. Wetting principles can be applied to better select cleans for front end of line (FEOL) and back end of line (BEOL) cleaning processes. These principles can also be used to help determine processes that best repel water from a semiconductor device. It is known that the value of the dielectric constant in an insulator increases when water is absorbed. These contact angle experiments will determine which processes can eliminate water absorption. Wetting is measured by the contact angle between a solid and a liquid. It is known that roughness plays a crucial role on the wetting of a substance. Different surface groups also affect the wetting of a surface. In this work, it was investigated how wetting was affected by different solid surfaces with different chemistries and different roughness. Four different materials were used: silicon; thermally grown silicon dioxide on silicon; chemically vapor deposited (CVD) silicon dioxide on silicon made from tetraethyl orthosilicate (TEOS); and organosilicate glass (OSG) on silicon.