A Guide to the Biology and Use of Forest Tree Seeds
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Plant Physiology
PLANT PHYSIOLOGY Vince Ördög Created by XMLmind XSL-FO Converter. PLANT PHYSIOLOGY Vince Ördög Publication date 2011 Created by XMLmind XSL-FO Converter. Table of Contents Cover .................................................................................................................................................. v 1. Preface ............................................................................................................................................ 1 2. Water and nutrients in plant ............................................................................................................ 2 1. Water balance of plant .......................................................................................................... 2 1.1. Water potential ......................................................................................................... 3 1.2. Absorption by roots .................................................................................................. 6 1.3. Transport through the xylem .................................................................................... 8 1.4. Transpiration ............................................................................................................. 9 1.5. Plant water status .................................................................................................... 11 1.6. Influence of extreme water supply .......................................................................... 12 2. Nutrient supply of plant ..................................................................................................... -

What Are Soybeans?
candy, cakes, cheeses, peanut butter, animal feeds, candles, paint, body lotions, biodiesel, furniture soybeans USES: What are soybeans? Soybeans are small round seeds, each with a tiny hilum (small brown spot). They are made up of three basic parts. Each soybean has a seed coat (outside cover that protects the seed), VOCABULARY cotyledon (the first leaf or pair of leaves within the embryo that stores food), and the embryo (part of a seed that develops into Cultivar: a variety of plant that has been created or a new plant, including the stem, leaves and roots). Soybeans, selected intentionally and maintained through cultivation. like most legumes, perform nitrogen fixation. Modern soybean Embryo: part of a seed that develops into a new plant, cultivars generally reach a height of around 1 m (3.3 ft), and including the stem, leaves and roots. take 80–120 days from sowing to harvesting. Exports: products or items that the U.S. sells and sends to other countries. Exports include raw products like whole soybeans or processed products like soybean oil or Leaflets soybean meal. Fertilizer: any substance used to fertilize the soil, especially a commercial or chemical manure. Hilum: the scar on a seed marking the point of attachment to its seed vessel (the brown spot). Leaflets: sub-part of leaf blade. All but the first node of soybean plants produce leaves with three leaflets. Legume: plants that perform nitrogen fixation and whose fruit is a seed pod. Beans, peas, clover and alfalfa are all legumes. Nitrogen Fixation: the conversion of atmospheric nitrogen Leaf into a nitrogen compound by certain bacteria, such as Stem rhizobium in the root nodules of legumes. -

International Union for the Protection of New Varieties of Plants
E TG/81/7(proj.1) ORIGINAL: English DATE: 2018-04-05 INTERNATIONAL UNION FOR THE PROTECTION OF NEW VARIETIES OF PLANTS Geneva DRAFT * COMMON SUNFLOWER UPOV Code(s): HLNTS_ANN Helianthus annuus L. GUIDELINES FOR THE CONDUCT OF TESTS FOR DISTINCTNESS, UNIFORMITY AND STABILITY prepared by experts from Hungary to be considered by the Technical Working Party for Agricultural Crops at its forty-seventh session, to be held in Naivasha, Kenya, from 2018-05-21 to 2018-05-25 Disclaimer: this document does not represent UPOV policies or guidance Alternative names:* Botanical name English French German Spanish Helianthus annuus L. Common Sunflower Soleil, Tournesol Sonnenblume Girasol The purpose of these guidelines (“Test Guidelines”) is to elaborate the principles contained in the General Introduction (document TG/1/3), and its associated TGP documents, into detailed practical guidance for the harmonized examination of distinctness, uniformity and stability (DUS) and, in particular, to identify appropriate characteristics for the examination of DUS and production of harmonized variety descriptions. ASSOCIATED DOCUMENTS These Test Guidelines should be read in conjunction with the General Introduction and its associated TGP documents. * These names were correct at the time of the introduction of these Test Guidelines but may be revised or updated. [Readers are advised to consult the UPOV Code, which can be found on the UPOV Website (www.upov.int), for the latest information.] TG/81/7(proj.1) Common Sunflower, 2018-04-05 2 TABLE OF CONTENTS PAGE 1. -

Effects of Root Pruning on Germinated Pecan Seedlings
PROPAGATION AND TISSUE CULTURE HORTSCIENCE 50(10):1549–1552. 2015. if pruning the roots shortly after germination stimulates lateral root formation. Similarly, little work has been attempted to consider Effects of Root Pruning on Germinated the effects of taproot pruning in young pecan seedlings back to different lengths on root Pecan Seedlings regeneration. The objective of this study 1 was to determine if root pruning at different Rui Zhang, Fang-Ren Peng , Pan Yan, Fan Cao, periods after seed germination affects pecan and Zhuang-Zhuang Liu seedling root and shoot growth and to evaluate College of Forestry, Nanjing Forestry University, 159 Longpan Road, different degrees of taproot pruning on root Nanjing 210037, China initiation. Dong-Liang Le Materials and Methods College of Forestry, Nanjing Forestry University, 159 Longpan Road, Nanjing 210037, China; and Nanjing Green Universe Pecan Science and The test was established at Lvzhou Pecan Research Station, Nanjing, China (lat. Technology Co. Ltd., 38 Muxuyuan Street, Nanjing 210007, China 32.05°N, long. 118.77°E). Open-pollinated Peng-Peng Tan seeds of the Chinese selection ‘Shaoxing’ were used as seed stock to produce seedlings. College of Forestry, Nanjing Forestry University, 159 Longpan Road, ‘Shaoxing’ had a small nut of very good Nanjing 210037, China quality with 50% percentage fill. The seeds averaged 30.4 mm in length, 20.9 mm in Additional index words. Carya illinoinensis, taproot, lateral root, shoot growth width, and 5.4 g in weight. Seeds were Abstract. Root systems of pecan trees are usually dominated by a single taproot with few collected on 7 Nov. -

Botany for Gardeners Offers a Clear Explanation of How Plants Grow
BotGar_Cover (5-8-2004) 11/8/04 11:18 AM Page 1 $19.95/ £14.99 GARDENING & HORTICULTURE/Reference Botany for Gardeners offers a clear explanation of how plants grow. • What happens inside a seed after it is planted? Botany for Gardeners Botany • How are plants structured? • How do plants adapt to their environment? • How is water transported from soil to leaves? • Why are minerals, air, and light important for healthy plant growth? • How do plants reproduce? The answers to these and other questions about complex plant processes, written in everyday language, allow gardeners and horticulturists to understand plants “from the plant’s point of view.” A bestseller since its debut in 1990, Botany for Gardeners has now been expanded and updated, and includes an appendix on plant taxonomy and a comprehensive index. Twodozen new photos and illustrations Botany for Gardeners make this new edition even more attractive than its predecessor. REVISED EDITION Brian Capon received a ph.d. in botany Brian Capon from the University of Chicago and was for thirty years professor of botany at California State University, Los Angeles. He is the author of Plant Survival: Adapting to a Hostile Brian World, also published by Timber Press. Author photo by Dan Terwilliger. Capon For details on other Timber Press books or to receive our catalog, please visit our Web site, www.timberpress.com. In the United States and Canada you may also reach us at 1-800-327-5680, and in the United Kingdom at [email protected]. ISBN 0-88192-655-8 ISBN 0-88192-655-8 90000 TIMBER PRESS 0 08819 26558 0 9 780881 926552 UPC EAN 001-033_Botany 11/8/04 11:20 AM Page 1 Botany for Gardeners 001-033_Botany 11/8/04 11:21 AM Page 2 001-033_Botany 11/8/04 11:21 AM Page 3 Botany for Gardeners Revised Edition Written and Illustrated by BRIAN CAPON TIMBER PRESS Portland * Cambridge 001-033_Botany 11/8/04 11:21 AM Page 4 Cover photographs by the author. -
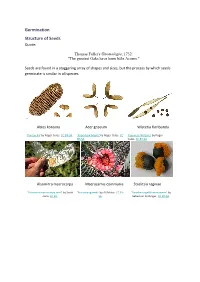
Germination Structure of Seeds Quote
Germination Structure of Seeds Quote: Thomas Fuller's Gnomologia, 1732: "The greatest Oaks have been little Acorns." Seeds are found in a staggering array of shapes and sizes, but the process by which seeds germinate is similar in all species. Abies koreana Acer griseum Wisteria floribunda 'Korean Fir' by Roger Culos. CC BY-SA. 'Paperbark Maple' by Roger Culos. CC 'Japanese Wisteria' by Roger BY-SA. Culos. CC BY-SA. Alsomitra macrocarpa Macrozamia communis Strelitzia reginae 'Alsomitra macrocarpa seed' by Scott 'BurrawangSeeds' by AYArktos. CC BY- 'Paradiesvogelblumensamen' by Zona. CC BY. SA. Sebastian Stabinger. CC BY-SA. Taraxicum officinale Stephanotis floribunda Phleum pratense 'Achane of Taraxacum sect. 'Stephanotis seed' by L. Marie"/Lenore 'Timoteegras vruchten Phleum Ruderalia' by Didier Edman, Sunnyvale, CA. CC BY. pratense' by Rasbak. CC BY-SA. Descouens. CC BY-SA. Dicotyledon seeds testa epicotyl plumule hypocotyl cotyledon radicle 'Aesculus hippocastanum seed section' by Boronian. CC BY. plumule epicotyl hypocotyl testa hilum radicle cotyledon micropyle endosperm Monocotyledon seeds endosperm epicotyl testa hypocotyl cotyledon radicle Parts of a seed Testa The seed coat. A protective layer which is tough and hard and it protects the seed from attack by insects, fungi and bacteria. Cotyledon Dicotyledons have 2 cotyledons Monocotyledons have 1 cotyledon A cotyledon is an embryonic leaf. It is the first leaf to appear when a seedling grows. They often contain reserves of food which the developing seedling can use to grow. Epicotyl The section of stem between the cotyledon(s) and the plumule. In a seedling it is the section of stem between the cotyledons and the first true leaves. -

Tree Seedling Availability, Planting, and Initial Care
F-5024 Tree Seedling Availability, Planting, and Initial Care William G. Ross Assistant Professor Oklahoma Cooperative Extension Fact Sheets Extension Forestry Specialistst are also available on our website at: http://www.osuextra.com Introduction This is the second in a series of three publications addressing the topic of forest stand establishment. If one is Bareroot not sure of what seedlings to plant or of what forest product to Most nurseries that sell seedlings produce some form of produce, it may be beneficial to read the first publication in this bareroot stock. Bareroot seedlings, as the name describes, is series, titled “Tree Planting Objectives and the Seedling a seedling with only the stem and the root supplied to the Selection Process.” landowner for planting. The major advantages of bareroot There are four points toconsider to identify management stock are the relative ease of production in the nurseries, the objectives. These are listed in brief form below. relative low cost to landowner, the relative ease of planting, and the ability to mechanize many of the operations in the 1. Identify the growing region where land is located. seedling production method. 2. Identify the soil types and moisture regimes located on the Shortcomings of bareroot seedlings include the vulner- land. ability of the scedling to the uncontrollable nursery environ- 3. Identify those objectives feasible and within the bounds of ment, exposure of the root system to harsh environmental the above environmental constraints. conditions after lifting, and the requirement of a large, sea- 4. Identify those tree species that, when managed correctly, sonal labor force to plant the seedlings. -

Life Cycles of Plants
Life Cycles of Plants Flowering plants produce flowers. The flowers later become fruit. Inside the fruit, we can find seeds. The fruit protect the seeds. Seeds can develop into new plants. The life cycle of flowering plants follows the three stages of seed-seedling-adult. Seed Seedling (Young plant) Stage 1: The plant begins Stage 2: The young plant that grows is known as a its life as a seed. With seedling. enough air, food, water and the right temperature, At first, the seedling obtains its food from the seed the seed will begin to leaves. germinate or grow. When the true leaves start to grow, the plant is ready to make its own food by the process of photosynthesis. The seed leaves will shrivel and fall off. Roots grow deep down into the ground to obtain water and mineral salts needed for the plant. The plant will grow towards the sunlight. The shoot grows upwards to obtain maximum sunlight. When the true leaves appear, the plant is ready to make its own food. Seed leaves provide the plant with food before the true leaves develop. Roots grow downwards to absorb water and mineral salts from the ground. They hold the plant firmly in the soil. Adult plant Stage 3: As the plant grows, it develops flowers which later become fruit. There are seeds inside the fruit. The seeds will fall to the ground and develop into new plants. The life cycle then repeats itself. Life cycle of a flowering plant Adapted: PSLE Science Partner A Complete Guide to L&U Block © Singapore Asia Publishers Pte Ltd. -

Genetic Suppression of Plant Development and Chloroplast Biogenesis Via the Snowy Cotyledon 3 and Phytochrome B Pathways
CSIRO PUBLISHING Functional Plant Biology, 2015, 42, 676–686 http://dx.doi.org/10.1071/FP15026 Genetic suppression of plant development and chloroplast biogenesis via the Snowy Cotyledon 3 and Phytochrome B pathways Diep Ganguly A, Peter Crisp A, Klaus Harter B, Barry J. Pogson A and Verónica Albrecht-Borth A,C AARC (Australian Research Council) Centre of Excellence in Plant Energy Biology, Research School of Biology, Australian National University Canberra, Acton, ACT 0200, Australia. BZentrum für Molekularbiologie der Pflanzen, Plant Physiology, University of Tübingen, 72076 Tübingen, Germany. CCorresponding author. Email: [email protected] Abstract. Plant development is regulated by external and internal factors such as light and chloroplast development. A revertant of the Arabidopsis thaliana (L.) Heyhn. chloroplast biogenesis mutant snowy cotyledon 3 (sco3–1) was isolated partially recovering the impaired chloroplast phenotype. The mutation was identified in the Phytochrome B (PhyB) gene and is a result of an amino acid change within the PAS repeat domain required for light-induced nuclear localisation. An independent phyB-9 mutation was crossed into sco3–1 mutants, resulting in the same partial reversion of sco3–1. Further analysis demonstrated that SCO3 and PhyB influence the greening process of seedlings and rosette leaves, embryogenesis, rosette formation and flowering. Interestingly, the functions of these proteins are interwoven in various ways, suggesting a complex genetic interaction. Whole-transcriptome profiling of sco3–1phyB-9 indicated that a completely distinct set of genes was differentially regulated in the double mutant compared with the single sco3–1 or phyB-9 mutants. Thus, we hypothesise that PhyB and SCO3 genetically suppress each other in plant and chloroplast development. -

New Insights on the Main Factors That Guide Seed Dormancy and Germination
G C A T T A C G G C A T genes Review From the Outside to the Inside: New Insights on the Main Factors That Guide Seed Dormancy and Germination Chiara Longo †, Soyanni Holness †, Veronica De Angelis, Andrea Lepri, Sara Occhigrossi, Veronica Ruta and Paola Vittorioso * Department Biology and Biotechnology C. Darwin, Sapienza Università di Roma, P.le Aldo Moro 5, 00185 Rome, Italy; [email protected] (C.L.); [email protected] (S.H.); [email protected] (V.D.A.); [email protected] (A.L.); [email protected] (S.O.); [email protected] (V.R.) * Correspondence: [email protected]; Tel.: +39-064-991-2265 † These authors contributed equally to this work. Abstract: The transition from a dormant to a germinating seed represents a crucial developmental switch in the life cycle of a plant. Subsequent transition from a germinating seed to an autotrophic organism also requires a robust and multi-layered control. Seed germination and seedling growth are multistep processes, involving both internal and external signals, which lead to a fine-tuning control network. In recent years, numerous studies have contributed to elucidate the molecular mechanisms underlying these processes: from light signaling and light-hormone crosstalk to the effects of abiotic stresses, from epigenetic regulation to translational control. However, there are still many open questions and molecular elements to be identified. This review will focus on the different aspects of the molecular control of seed dormancy and germination, pointing out new molecular elements and how these integrate in the signaling pathways already known. -

The Effect of Seed Weight on Photosynthetic Area Development and Weight of the Sainfoin (Onebrychis Spp
The effect of seed weight on photosynthetic area development and weight of the sainfoin (Onebrychis spp. Scop.) seedling by Stephen Carl Fransen A thesis submitted in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE in Agronomy Montana State University © Copyright by Stephen Carl Fransen (1975) Abstract: The hypothesis that cotyledon area to seed weight ratio might vary among sainfoin accessions was tested by comparing regression coefficients of area on weight. In addition, effects of seed weight upon areas and weights of cotyledons, first and second leaves and total seedlings were studied. Ten accessions of sainfoin representing three different species were studied. After 17 days of growth in a growth chamber, seedlings of all accessions were sampled for the characteristics to be studied. The ratios of cotyledon area to seed weight were similar for most accessions. Seedlings from heavier seeds emerged and developed more rapidly than seedlings from lighter seeds. Embryo axis length and width and leaf primordia length were all highly correlated with seed weight. Areas and weights of cotyledons, first and second leaves were correlated with seed weight for most accessions. Total seedling area and weight, at 17 days of age, were also correlated with initial seed weight. Trifoliolate seedlings had greater first leaf area but less second leaf area than unifoliolate seedlings. As a result, trifoliolate seedlings had only 3.5 percent more total leaf area than unifoliolate seedlings at 17 days. These results help explain some of the variability among previous tests comparing unifoliolate and trifoliolate seedlings for seedling vigor. STATEMENT OF PERMISSION TO COPY Iu presenting this thesis in partial fulfillment of the requirements for an advanced degree at Montana State University, I agree that the Library shall make it freely .available for inspection„ I further agree that permission for extensive copying of this thesis for scholarly purposes- may be granted by my major professor, or, in his absence, by the Director of Libraries. -

BIO 102 General Biology Lecture Outline Plantae I. Introduction A. Taxonomy Domain
BIO 102 General Biology Lecture Outline Plantae I. Introduction A. Taxonomy Domain: Eukarya Kingdom: Plantae B. General characteristics Multicellular Autotrophic / Photosynthetic Cellulose cell wall Reproduction – alternation of generations C. Life cycle Alternation of generations Haploid = gametophyte, which produces gametes via mitosis Diploid = sporophyte, which produces spores via meiosis Meiosis = cell division from 2n to n Mitosis = cell division either from 2n to 2n, OR from n to n Fertilization = combine gametes: n + n = 2n Homospory vs. heterospory II. Major Groups of Plants A. Characterized by 4 criteria in branching manner Vascular vs. non-vascular Seed production or lack thereof Types of seeds produced Development & morphology B. Non-tracheophytes (nonvascular plants) General characteristics No vascular tissue Gametophyte (haploid) generation dominant Requires water for reproduction Examples Liverworts Hornworts Mosses Moss reproductive cycle C. Tracheophytes General characteristics Vascular tissue: Xylem and Phloem Specialized tissues: Leaves Roots Stems Cuticles Stomata Sporophyte (diploid) generation dominant Divisions based on seed-bearing vs. non-seed bearing (Seed vs. spore) D. Seedless plants BIO 102 General Biology Lecture Outline General characteristics Have vascular tissue but lack seeds Examples Ferns Whisk ferns Club mosses Horsetails E. Seed-bearing plants General characteristics Have vascular tissue and seeds Divisions based on whether seeds are “covered” or not F. Gymnosperms General characteristics Vascular plants with “naked” seeds = i.e., no fruit Examples Conifers Gingkos Cycads Gnetophytes Gymnosperm life cycle G. Angiosperms General characteristics Vascular plants with “covered” seeds = i.e., seeds within fruits Fruits are derived from flowers Angiosperm = flowering plant Divisions are based on development and plant morphology (i.e., structures) Angiosperm life cycle H.