Detection and Characterization of Phosphorylation, Glycosylation, and Fatty Acid Bound to Fetuin a in Human Blood
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Fetuin (␣2-HS-Glycoprotein) Opsonizes Cationic Macrophage- Deactivating Molecules (CNI-1493͞P38 Mitogen-Activated Protein Kinase)
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 14429–14434, November 1998 Medical Sciences Fetuin (a2-HS-glycoprotein) opsonizes cationic macrophage- deactivating molecules (CNI-1493yp38 mitogen-activated protein kinase) HAICHAO WANG*†‡,MINGHUANG ZHANG†,MARINA BIANCHI†,BARBARA SHERRY†,ANDREW SAMA*, AND KEVIN J. TRACEY†§ Departments of *Emergency Medicine and §Surgery, North Shore University Hospital-New York University School of Medicine, and †The Picower Institute for Medical Research, Manhasset, NY 11030 Communicated by George J. Todaro, University of Washington, Seattle, WA, September 22, 1998 (received for review February 5, 1998) ABSTRACT Macrophages become activated by bacterial of circulating fetuin, but it also is expressed by mono- endotoxin (lipopolysaccharide) and other stimuli to release cyteymacrophages (26). The fetuin promoter region has several proinflammatory cytokines and NO. To prevent release of toxic potential interleukin 6-responsive elements (27), and its synthesis or potentially lethal quantities of these factors, the state of is down-regulated during injury and inflammation (20). Fetuin is macrophage activation is counter-regulated by anti-inflamma- an acidic glycoprotein with three N-linked and three O-linked tory mediators (e.g., glucocorticoid hormones, interleukin 10, oligosaccharide chains, whose terminal sugar residues are rich in b and transforming growth factor type ). Fetuin, a negative sialic acid (N-acetylneuraminic acid), contributing to its net acute-phase protein, recently was implicated as an anti- negative charge (28). A role for fetuin as a carrier of bioactive inflammatory mediator, because it is required for macrophage molecules has been proposed based on observations that it binds deactivation by spermine. In the present studies, we found that and carries Ca21 ion (22). -

A Systematic Review on the Implications of O-Linked Glycan Branching and Truncating Enzymes on Cancer Progression and Metastasis
cells Review A Systematic Review on the Implications of O-linked Glycan Branching and Truncating Enzymes on Cancer Progression and Metastasis 1, 1, 1 1,2,3, Rohitesh Gupta y, Frank Leon y, Sanchita Rauth , Surinder K. Batra * and Moorthy P. Ponnusamy 1,2,* 1 Department of Biochemistry and Molecular Biology, University of Nebraska Medical Center, Omaha, NE 68105, USA; [email protected] (R.G.); [email protected] (F.L.); [email protected] (S.R.) 2 Fred and Pamela Buffett Cancer Center, Eppley Institute for Research in Cancer and Allied Diseases, University of Nebraska Medical Center, Omaha, NE 681980-5900, USA 3 Department of Pathology and Microbiology, UNMC, Omaha, NE 68198-5900, USA * Correspondence: [email protected] (S.K.B.); [email protected] (M.P.P.); Tel.: +402-559-5455 (S.K.B.); +402-559-1170 (M.P.P.); Fax: +402-559-6650 (S.K.B. & M.P.P.) Equal contribution. y Received: 21 January 2020; Accepted: 12 February 2020; Published: 14 February 2020 Abstract: Glycosylation is the most commonly occurring post-translational modifications, and is believed to modify over 50% of all proteins. The process of glycan modification is directed by different glycosyltransferases, depending on the cell in which it is expressed. These small carbohydrate molecules consist of multiple glycan families that facilitate cell–cell interactions, protein interactions, and downstream signaling. An alteration of several types of O-glycan core structures have been implicated in multiple cancers, largely due to differential glycosyltransferase expression or activity. Consequently, aberrant O-linked glycosylation has been extensively demonstrated to affect biological function and protein integrity that directly result in cancer growth and progression of several diseases. -

Glycosylation: Rising Potential for Prostate Cancer Evaluation
cancers Review Glycosylation: Rising Potential for Prostate Cancer Evaluation Anna Kałuza˙ * , Justyna Szczykutowicz and Mirosława Ferens-Sieczkowska Department of Chemistry and Immunochemistry, Wroclaw Medical University, Sklodowskiej-Curie 48/50, 50-369 Wroclaw, Poland; [email protected] (J.S.); [email protected] (M.F.-S.) * Correspondence: [email protected]; Tel.: +48-71-770-30-66 Simple Summary: Aberrant protein glycosylation is a well-known hallmark of cancer and is as- sociated with differential expression of enzymes such as glycosyltransferases and glycosidases. The altered expression of the enzymes triggers cancer cells to produce glycoproteins with specific cancer-related aberrations in glycan structures. Increasing number of data indicate that glycosylation patterns of PSA and other prostate-originated proteins exert a potential to distinguish between benign prostate disease and cancer as well as among different stages of prostate cancer development and aggressiveness. This review summarizes the alterations in glycan sialylation, fucosylation, truncated O-glycans, and LacdiNAc groups outlining their potential applications in non-invasive diagnostic procedures of prostate diseases. Further research is desired to develop more general algorithms exploiting glycobiology data for the improvement of prostate diseases evaluation. Abstract: Prostate cancer is the second most commonly diagnosed cancer among men. Alterations in protein glycosylation are confirmed to be a reliable hallmark of cancer. Prostate-specific antigen is the biomarker that is used most frequently for prostate cancer detection, although its lack of sensitivity and specificity results in many unnecessary biopsies. A wide range of glycosylation alterations in Citation: Kałuza,˙ A.; Szczykutowicz, prostate cancer cells, including increased sialylation and fucosylation, can modify protein function J.; Ferens-Sieczkowska, M. -

New Metabolic and Genetic Biomarkers for Diagnosis of Ulcerative Colitis
Preprints (www.preprints.org) | NOT PEER-REVIEWED | Posted: 2 August 2021 doi:10.20944/preprints202108.0048.v1 Review New Metabolic and Genetic Biomarkers for Diagnosis of Ulcerative Colitis Elena-Emilia Tudoroiu1, Mihaela-Andreea Costache1 and Maria-Andreea Stancu 1,* and Anca Lucia Pop1 1 Department of Clinical Laboratory, Food Safety, "Carol Davila" University of Medicine and Pharmacy, 6 Traian Vuia Street, Bucharest 020945, Romania, [email protected], [email protected], [email protected], [email protected] * Correspondence: [email protected], (M.S.); [email protected] (A.L.P.) Tel.: +40745009910 Abstract: Ulcerative colitis (UC) is one of the two disorders known as inflammatory bowel diseases (IBD) along with Crohn’s disease (CD), with complex pathogenesis, requiring costly invasive investigations. Objective: to examine the most recent biomarkers proposed for UC diagnosis; to establish the strategy used to make the differential diagnosis between UC and CD relying on these biomarkers, also adding the benefit of finding new non-invasive tools in managing this condition. The search was performed in a single database (Web of Science) using the specific keywords „ulcerative colitis”, „biomarkers” and „diagnosis” for the last five years. Study eligibility criteria: clinical trials on adults and pediatric patients with ulcerative colitis compared with Crohn’s disease. Results: We selected 57 studies, randomized controlled trials (RCTs) and clinical case series (CCS), summarizing the latest most specific biomarkers in diagnosis of UC. Limitations: we considered RCTs and CCS from one database, limited to the search topics. Our findings indicate a important number of potential biomarkers with diagnostic value, which bring the advantage of a non-invasive method to approach this challenging disorder. -

Glycosylation of Immune Receptors in Cancer
cells Review Glycosylation of Immune Receptors in Cancer Ruoxuan Sun, Alyssa Min Jung Kim and Seung-Oe Lim * Department of Medicinal Chemistry and Molecular Pharmacology, Purdue Institute of Drug Discovery, Purdue Center for Cancer Research, Purdue University, West Lafayette, IN 47907, USA; [email protected] (R.S.); [email protected] (A.M.J.K.) * Correspondence: [email protected]; Tel.: +1-765-494-3531 Abstract: Evading host immune surveillance is one of the hallmarks of cancer. Immune checkpoint therapy, which aims to eliminate cancer progression by reprogramming the antitumor immune response, currently occupies a solid position in the rapidly expanding arsenal of cancer therapy. As most immune checkpoints are membrane glycoproteins, mounting attention is drawn to asking how protein glycosylation affects immune function. The answers to this fundamental question will stimulate the rational development of future cancer diagnostics and therapeutic strategies. Keywords: glycosylation; N-glycan; immune receptor; immune checkpoint therapy; cancer 1. Introduction The immune system can eliminate malignancy at an early stage by recognizing the antigenic peptide epitope presented by neoplastically transformed cells [1,2]. However, a subset of tumor cells may evolve to coexist with antitumor immunity by a process defined as immunoediting [3]. To combat the immune attack and maintain the balance with the host, cancer cells develop a series of approaches such as (1) downregulation of antigen Citation: Sun, R.; Kim, A.M.J.; Lim, presentation to make them invisible to immunity/the immune system, (2) production S.-O. Glycosylation of Immune of immunomodulatory cytokines to establish an immune suppressive environment, and Receptors in Cancer. Cells 2021, 10, (3) expression of surface immune checkpoint proteins such as programmed cell death 1 1100. -

Design of Glycosylation Sites by Rapid Synthesis and Analysis of Glycosyltransferases
ARTICLES https://doi.org/10.1038/s41589-018-0051-2 Design of glycosylation sites by rapid synthesis and analysis of glycosyltransferases Weston Kightlinger 1,2,7, Liang Lin2,3,7, Madisen Rosztoczy3, Wenhao Li3, Matthew P. DeLisa 4,5, Milan Mrksich 1,2,3,6* and Michael C. Jewett 1,2* Glycosylation is an abundant post-translational modification that is important in disease and biotechnology. Current methods to understand and engineer glycosylation cannot sufficiently explore the vast experimental landscapes required to accurately predict and design glycosylation sites modified by glycosyltransferases. Here we describe a systematic platform for glycosyl- ation sequence characterization and optimization by rapid expression and screening (GlycoSCORES), which combines cell-free protein synthesis and mass spectrometry of self-assembled monolayers. We produced six N- and O-linked polypeptide-modify- ing glycosyltransferases from bacteria and humans in vitro and rigorously determined their substrate specificities using 3,480 unique peptides and 13,903 unique reaction conditions. We then used GlycoSCORES to optimize and design small glycosylation sequence motifs that directed efficient N-linked glycosylation in vitro and in the Escherichia coli cytoplasm for three heterolo- gous proteins, including the human immunoglobulin Fc domain. We find that GlycoSCORES is a broadly applicable method to facilitate fundamental understanding of glycosyltransferases and engineer synthetic glycoproteins. rotein glycosylation is the post-translational attachment of O-linked polypeptide N-acetylgalactosaminyltransferase (ppGalN- oligosaccharides (glycans), most commonly at asparagine AcT), O-linked N-acetylglucosamine transferase (OGT), and oligo- (N-linked) or serine and threonine (O-linked) amino acid side saccharyltransferase (OST) enzyme families. Such enzymes are of P 1,2 chains . -

2HS-Glycoprotein, an Antagonist of Transforming Growth Factor in Vivo
[CANCER RESEARCH 64, 6402–6409, September 15, 2004] ␣2HS-glycoprotein, an Antagonist of Transforming Growth Factor  In vivo, Inhibits Intestinal Tumor Progression Carol J. Swallow,1,2 Emily A. Partridge,1 Jennifer C. Macmillan,1 Tania Tajirian,1 Gianni M. DiGuglielmo,1 Kazy Hay,1 Melanie Szweras,1 Willi Jahnen-Dechent,3 Jeff L. Wrana,1,2 Mark Redston,1 Steven Gallinger,1,2 and James W. Dennis1,2 1Samuel Lunenfeld Research Institute, Mount Sinai Hospital, Toronto, Ontario, Canada; 2Departments of Molecular and Medical Genetics and Surgery, University of Toronto, Toronto, Ontario, Canada; and 3Interdisziplina¨ren Zentrums fu¨r Klinische Forschung BIOMAT, Klinikum der Rheinisch-Westfa¨lischen Technischen Hoschschule, Aachen, Aachen, Germany ABSTRACT 1-induced epithelial-mesenchymal transition is characterized by down-regulation of E-cadherin expression, loss of tight junctions, and  Transforming growth factor (TGF)- 1 is associated with tumor pro- increased cell motility (1, 10) and is dependent on phosphatidylinosi- gression and resistance to chemotherapy in established cancers, as well as tol 3Ј-kinase/Akt activation (11), a key pathway promoting tumor cell host immune suppression. Here, we show that the serum glycoprotein ␣2-HS-glycoprotein (AHSG) blocks TGF-1 binding to cell surface re- invasiveness (12). Epithelial-mesenchymal transition appears to be a ceptors, suppresses TGF- signal transduction, and inhibits TGF-- close in vitro correlate of metastatic capacity, and both require coop- induced epithelial-mesenchymal transition, suggesting that AHSG may erative signaling of TGF- and Ras/mitogen-activated protein kinase play a role in tumor progression. In 66 consecutive sporadic human pathways (13, 14). Tumor-derived TGF-1 also has paracrine effects colorectal cancer specimens, we observed a 3-fold depletion of ASHG in on host cells that promote angiogenesis, alter matrix turnover, and tumor compared with normal tissue, whereas levels of other abundant suppress immune cell functions (1, 7, 15, 16). -

Native Glycosylation Is Important for Biological Function and Activity of Recombinant Human Proteins
Native Glycosylation Is Important For Biological Function and Activity of Recombinant Human Proteins INTRODUCTION Mark Azam, Ph.D. VP - R&D and Manufacturing Patricia Ahrweiler, Ph.D. Sr. Marketing Consultant SUMMARY Although recombinant human cytokines are commercially available from bacterial, eukaryotic, and mammalian cell expression systems (E. coli, SF9, CHO), human cell- expressed (HEK293) glycoproteins offer the distinct advantage of native human glycosylation. In this Whitepaper, several examples illustrating the importance of correct glycosylation in cytokine activity, stability and half-life, are provided for cytokines widely used in stem cell research. the in to to in the in the in the tion to N to the to the threonine or the to in vivo in E. coli to there in the no or the in Erythropoietin Erythropoietin the in in the other to the EPO (Humankine) glycostructures and the amount of sialic acid in the glycan trees (Croset, 2012). 1 a release of the glycan structures, Calf serum fetuin containing sialylated N and O used as a control for endoglycosidase results that a large of neutral glycans, of ) is mast endothelial acids, sites for glycosylation and cysteine residues forming three 12 and 20 glycosylation. a man cell line, migrates as a of due a a an molecular mass of a due to glycosylation at one glycosylation site and its of is for efficient secretion, the role of glycosylation is unclear. the The bioactivity of VEGF165 was determined by its ability to induce proliferation of human umbilical vein endothelial cells. This supports the importance of correct glycosylation in the biological properties of VEGF165 in cell culture. -

Development of a Whole Cell Fermentation Process for Glycosylation of Small Molecule Flavonoids
Development of a Whole Cell Fermentation Process for Glycosylation of Small Molecule Flavonoids Dissertation Zur Erlangung der Würde des Doktors der Naturwissenschaften des Fachbereichs Biologie, der Fakultät für Mathematik, Informatik und Natur- wissenschaften, der Universität Hamburg vorgelegt von Tanja Vanessa Heyer aus Norderstedt Hamburg 2019 Vollständiger Abdruck der von der Fakultät für Mathematik, Informatik und Natur- wissenschaften der Universität Hamburg zur Erlangung des akademischen Grades eines Doktors der Naturwissenschaften genehmigten Dissertation. Gutachter: Prof. Dr. Wolfgang R. Streit PD Dr. Andreas Pommerening-Röser Tag der Disputation: 08.11.2019 Eidesstattliche Versicherung Hiermit erkläre ich an Eides statt, dass ich die vorliegende Dissertationsschrift selbst verfasst und keine anderen als die angegebenen Quellen und Hilfsmittel benutzt habe. ____________________________________ (Tanja Heyer) Funding This work was part of the project “GO-Bio 6: GLYCONIC - Selektiv glykosylierte Polyphenole” funded by the GO-Bio initiative from the German Federal Ministry of Education and Research (BMBF, funding number: 031A376). A b s t r a c t | I Abstract Flavonoids are known to have multiple beneficial effects on human health. These make them desired compounds as bioactives, e.g. in cosmeceutical products. The glycosylation of flavonoids has positive impact on their solubility, bioavailability, and stability and is furthermore able to improve their physiological effects. In this study, flavonoid glycosides were produced in a whole-cell biocatalytic process in Escherichia coli by directed transfer of activated sugar residues to hydroxy groups of the polyphenolic scaffold within an enzymatic reaction by use of glycosyltransferases. The goal of this work was the development and optimization of the production process for glycosylated flavonoids in gram-scale, comprising the upstream (USP) and downstream (DSP) processing. -

The Serum Glycoprotein Fetuin-A Promotes Lewis Lung Carcinoma Tumorigenesis Via Adhesive-Dependent and Adhesive-Independent Mechanisms
Research Article The Serum Glycoprotein Fetuin-A Promotes Lewis Lung Carcinoma Tumorigenesis via Adhesive-Dependent and Adhesive-Independent Mechanisms Madappa N. Kundranda,1 Melodie Henderson,3 Kathy J. Carter,3 Lee Gorden,3 Awadh Binhazim,2 Sanhita Ray,1 Trevor Baptiste,1 Masih Shokrani,1 Maria L. Leite-Browning,3 Willi Jahnen-Dechent,4 Lynn M. Matrisian,3 and Josiah Ochieng1,3 Departments of 1Biochemistry and 2Pathology, Meharry Medical College and 3Department of Cancer Biology, Vanderbilt University, Nashville, Tennessee, and 4IZKF BIOMAT, University Clinics, Aachen, Germany Abstract serum concentration drops after trauma, infection, and certain Fetuin-A is a serum glycoprotein in the cystatin family malignancies. Recently, a second protein of the fetuin family called associated with the regulation of soft tissue calcification. We fetuin-B was described in humans and rodents (1). Based on domain tested the role of systemic fetuin in tumor cell growth and homology, overall conservation of cysteine residues and chromo- metastasis by injecting Lewis lung carcinoma (LLC) cells into somal assignments of the corresponding genes in these species, fetuin-B was described as a paralogue of fetuin-A. fetuin-A null and their wild-type (WT) littermate control C57BL/6 mice via the tail vein, s.c., and intrasplenic routes. Fetuin-A has been linked to a number of cellular functions such In the experimental metastasis assay, the lungs of the WT as brain development (2), bone remodeling, and immune function (5, 6). It can also mimic and antagonize the anti-mitogenic effects mice were filled with metastatic nodules, whereas the lungs h of the fetuin-A null mutant mice were virtually free of of transforming growth factor- in test tube assays, in cell culture, colonies at the end of 2 weeks. -

Fetuin a Concentration in the Second Trimester Amniotic Fluid of Fetuses with Trisomy 21 Appears to Be Lower: Phenotypic Considerations
Hindawi Publishing Corporation Mediators of Inflammation Volume 2012, Article ID 138971, 4 pages doi:10.1155/2012/138971 Research Article Fetuin A Concentration in the Second Trimester Amniotic Fluid of Fetuses with Trisomy 21 Appears to Be Lower: Phenotypic Considerations S. Iliodromiti,1, 2 N. Vrachnis,1 Evangelia Samoli,3 Z. Iliodromiti,1 C. Pangalos,4 N. Drakoulis,5 G. Creatsas,1 and D. Botsis1 1 2nd Department of Obstetrics and Gynecology, University of Athens Medical School, Aretaieion Hospital, 124B Vas. Sofias Avenue, 115 26 Athens, Greece 2 West of Scotland Deanery, Obstetrics and Gynaecology, Glasgow, G3 8BW, UK 3 Department of Hygiene, Epidemiology and Medical Statistics, University of Athens Medical School, 115 27 Athens, Greece 4 Diagnostic Genetic Centre, 115 28 Athens, Greece 5 Department of Pharmaceutical Technology, School of Pharmacy, University of Athens, 157 71 Athens, Greece Correspondence should be addressed to N. Vrachnis, [email protected] Received 23 July 2011; Revised 21 November 2011; Accepted 21 November 2011 Academic Editor: Teresa Zelante Copyright © 2012 S. Iliodromiti et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Objective. We investigated whether the concentration of the glycoprotein fetuin A is altered in the second trimester amniotic fluid of trisomy 21 pregnancies compared with euploid pregnancies. Methods. 25 pregnancies with an extra chromosome 21 were matched for maternal and gestational age with 25 pregnancies with normal karyotype. Levels of fetuin A in amniotic fluid were measured by a commercially available enzyme-linked immunosorbent assay (ELISA) kit. -
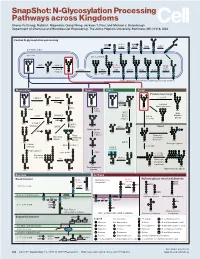
N-Glycosylation Processing Pathways Across Kingdoms Cheng-Yu Chung, Natalia I
SnapShot: N-Glycosylation Processing Pathways across Kingdoms Cheng-Yu Chung, Natalia I. Majewska, Qiong Wang, Jackson T. Paul, and Michael J. Betenbaugh Department of Chemical and Biomolecular Engineering, The Johns Hopkins University, Baltimore, MD 21218, USA Central N-glycosylation processing UDP ALG13 GDP ALG7 ALG14 ALG1 ALG2 ALG11 P P P P P CYTOPLASM P P P P P P GOLGI ER LUMEN GNTI α -Man IA En Bloc ALG3 Transfer ALG9 α-Man IB ER Man I α -Glc I OST ALG10 ALG8 ALG6 ALG12 Asn α -Man IC Asn Asn Asn α -Glc II P P P P P Asn P P P P P ALGs: asparagine-linked N-glycosylation processing enzymes Mammalian Insect Plant Fungi Filamentous fungi α-Man II FUT8 α-1,2-MT XYLT GDP Terminal Asn Asn Asn Asn UDP Asn Galactofuranose GNTII Asn Asn FUT8 f ± α-Gal Asn FUTC3 GNTII High FUT11 α -GALT B14GALTI Och1p Mannose FUT12 glycan α-Man II α-1,2-MT UDP Asn Asn ST3GAL4 Asn Asn GNTIV ST6GAL1 GNTIII GNTV Asn GNase Yeast Hybrid glycan P CMP GNase Asn Asn Tetra- α-Man II Mannosyl- Bisecting antennary phosphate glycan glycan transferase Asn Asn Asn Asn GALT1 Asn ST8SIA2 Core type glycan GNTs B14GALTI α-1,6-MT ST8SIA4 Asn Lewis A structure Polysialylation α-Man PolyLacNAc α-1,2-MT Alternative α-1,3-MT P sialic acid FUT13 ] [ STs Mannosyl- n phosphate Asn Asn Asn transferase Asn Asn Paucimannose Hypermannose glycan Asn Asn Asn glycans Bacteria Archaea Archaea glycan structural diversity Block transfer En Bloc Methanococcus En Bloc Transfer Transfer maripaludis OCH3 Thr OCH AgIB Thr OCH3 3 PERIPLASM PglB B B Asn P - P Asn OSO3 P Asn Asn INNER MEMBRANE P Asn P