Hemoglobin Mediated Regulation of Platelet Functions
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplementary Information Changes in the Plasma Proteome At
Supplementary Information Changes in the plasma proteome at asymptomatic and symptomatic stages of autosomal dominant Alzheimer’s disease Julia Muenchhoff1, Anne Poljak1,2,3, Anbupalam Thalamuthu1, Veer B. Gupta4,5, Pratishtha Chatterjee4,5,6, Mark Raftery2, Colin L. Masters7, John C. Morris8,9,10, Randall J. Bateman8,9, Anne M. Fagan8,9, Ralph N. Martins4,5,6, Perminder S. Sachdev1,11,* Supplementary Figure S1. Ratios of proteins differentially abundant in asymptomatic carriers of PSEN1 and APP Dutch mutations. Mean ratios and standard deviations of plasma proteins from asymptomatic PSEN1 mutation carriers (PSEN1) and APP Dutch mutation carriers (APP) relative to reference masterpool as quantified by iTRAQ. Ratios that significantly differed are marked with asterisks (* p < 0.05; ** p < 0.01). C4A, complement C4-A; AZGP1, zinc-α-2-glycoprotein; HPX, hemopexin; PGLYPR2, N-acetylmuramoyl-L-alanine amidase isoform 2; α2AP, α-2-antiplasmin; APOL1, apolipoprotein L1; C1 inhibitor, plasma protease C1 inhibitor; ITIH2, inter-α-trypsin inhibitor heavy chain H2. 2 A) ADAD)CSF) ADAD)plasma) B) ADAD)CSF) ADAD)plasma) (Ringman)et)al)2015)) (current)study)) (Ringman)et)al)2015)) (current)study)) ATRN↓,%%AHSG↑% 32028% 49% %%%%%%%%HC2↑,%%ApoM↓% 24367% 31% 10083%% %%%%TBG↑,%%LUM↑% 24256% ApoC1↓↑% 16565% %%AMBP↑% 11738%%% SERPINA3↓↑% 24373% C6↓↑% ITIH2% 10574%% %%%%%%%CPN2↓%% ↓↑% %%%%%TTR↑% 11977% 10970% %SERPINF2↓↑% CFH↓% C5↑% CP↓↑% 16566% 11412%% 10127%% %%ITIH4↓↑% SerpinG1↓% 11967% %%ORM1↓↑% SerpinC1↓% 10612% %%%A1BG↑%%% %%%%FN1↓% 11461% %%%%ITIH1↑% C3↓↑% 11027% 19325% 10395%% %%%%%%HPR↓↑% HRG↓% %%% 13814%% 10338%% %%% %ApoA1 % %%%%%%%%%GSN↑% ↓↑ %%%%%%%%%%%%ApoD↓% 11385% C4BPA↓↑% 18976%% %%%%%%%%%%%%%%%%%ApoJ↓↑% 23266%%%% %%%%%%%%%%%%%%%%%%%%%%ApoA2↓↑% %%%%%%%%%%%%%%%%%%%%%%%%%%%%A2M↓↑% IGHM↑,%%GC↓↑,%%ApoB↓↑% 13769% % FGA↓↑,%%FGB↓↑,%%FGG↓↑% AFM↓↑,%%CFB↓↑,%% 19143%% ApoH↓↑,%%C4BPA↓↑% ApoA4↓↑%%% LOAD/MCI)plasma) LOAD/MCI)plasma) LOAD/MCI)plasma) LOAD/MCI)plasma) (Song)et)al)2014)) (Muenchhoff)et)al)2015)) (Song)et)al)2014)) (Muenchhoff)et)al)2015)) Supplementary Figure S2. -

The Role of Methemoglobin and Carboxyhemoglobin in COVID-19: a Review
Journal of Clinical Medicine Review The Role of Methemoglobin and Carboxyhemoglobin in COVID-19: A Review Felix Scholkmann 1,2,*, Tanja Restin 2, Marco Ferrari 3 and Valentina Quaresima 3 1 Biomedical Optics Research Laboratory, Department of Neonatology, University Hospital Zurich, University of Zurich, 8091 Zurich, Switzerland 2 Newborn Research Zurich, Department of Neonatology, University Hospital Zurich, University of Zurich, 8091 Zurich, Switzerland; [email protected] 3 Department of Life, Health and Environmental Sciences, University of L’Aquila, 67100 L’Aquila, Italy; [email protected] (M.F.); [email protected] (V.Q.) * Correspondence: [email protected]; Tel.: +41-4-4255-9326 Abstract: Following the outbreak of a novel coronavirus (SARS-CoV-2) associated with pneumonia in China (Corona Virus Disease 2019, COVID-19) at the end of 2019, the world is currently facing a global pandemic of infections with SARS-CoV-2 and cases of COVID-19. Since severely ill patients often show elevated methemoglobin (MetHb) and carboxyhemoglobin (COHb) concentrations in their blood as a marker of disease severity, we aimed to summarize the currently available published study results (case reports and cross-sectional studies) on MetHb and COHb concentrations in the blood of COVID-19 patients. To this end, a systematic literature research was performed. For the case of MetHb, seven publications were identified (five case reports and two cross-sectional studies), and for the case of COHb, three studies were found (two cross-sectional studies and one case report). The findings reported in the publications show that an increase in MetHb and COHb can happen in COVID-19 patients, especially in critically ill ones, and that MetHb and COHb can increase to dangerously high levels during the course of the disease in some patients. -

Section 8: Hematology CHAPTER 47: ANEMIA
Section 8: Hematology CHAPTER 47: ANEMIA Q.1. A 56-year-old man presents with symptoms of severe dyspnea on exertion and fatigue. His laboratory values are as follows: Hemoglobin 6.0 g/dL (normal: 12–15 g/dL) Hematocrit 18% (normal: 36%–46%) RBC count 2 million/L (normal: 4–5.2 million/L) Reticulocyte count 3% (normal: 0.5%–1.5%) Which of the following caused this man’s anemia? A. Decreased red cell production B. Increased red cell destruction C. Acute blood loss (hemorrhage) D. There is insufficient information to make a determination Answer: A. This man presents with anemia and an elevated reticulocyte count which seems to suggest a hemolytic process. His reticulocyte count, however, has not been corrected for the degree of anemia he displays. This can be done by calculating his corrected reticulocyte count ([3% × (18%/45%)] = 1.2%), which is less than 2 and thus suggestive of a hypoproliferative process (decreased red cell production). Q.2. A 25-year-old man with pancytopenia undergoes bone marrow aspiration and biopsy, which reveals profound hypocellularity and virtual absence of hematopoietic cells. Cytogenetic analysis of the bone marrow does not reveal any abnormalities. Despite red blood cell and platelet transfusions, his pancytopenia worsens. Histocompatibility testing of his only sister fails to reveal a match. What would be the most appropriate course of therapy? A. Antithymocyte globulin, cyclosporine, and prednisone B. Prednisone alone C. Supportive therapy with chronic blood and platelet transfusions only D. Methotrexate and prednisone E. Bone marrow transplant Answer: A. Although supportive care with transfusions is necessary for treating this patient with aplastic anemia, most cases are not self-limited. -
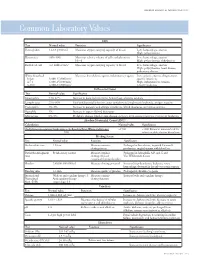
Common Laboratory Values
AmericAn AcAdemy of PediAtric dentistry Reference Manual 2006-2007 Resource Section 251 Common LaboratoryCommon Values Laboratory Values CBC Test Normal value Function Significance Hemoglobin 12-18 g/100 mL Measures oxygen carrying capacity of blood Low: hemorrhage, anemia High: polycythemia Hematocrit 35%-50% Measures relative volume of cells and plasma in Low: hemorrhage, anemia blood High: polycythemia, dehydration Red blood cell 4-6 million/mm3 Measures oxygen-carrying capacity of blood Low: hemorrhage, anemia High: polycythemia, heart disease, pulmonary disease White blood cell Measures host defense against inflammatory agents Low: aplastic anemia, drug toxicity, Infant 8,000-15,000/mm3 specific infections 4-7 y 6,000-15,000/mm3 High: inflammation, trauma, 8-18 y 4,500-13,500/mm3 toxicity, leukemia Differential Count Test Normal value Significance Neutrophils 54%-62% Increase in bacterial infections, hemorrhage, diabetic acidosis Lymphocytes 25%-30% Viral and bacterial infection, acute and chronic lymphocytic leukemia, antigen reaction Eosinophils 1%-3% Increase in parasitic and allergic conditions, blood dyscrasias, pernicious anemia Basophils 1% Increase in types of blood dyscrasias Monocytes 0%-9% Hodgkin’s disease, lipid storage disease, recovery from severe infections, monocytic leukemia Absolute Neutrophil Count (ANC) Calculation Normal value Significance (% Polymorphonuclear Leukocytes + % Bands)×Total White Cell Count >1500 <1000 Patient at increased risk for 100 infection; defer elective dental care Bleeding Screen Test -

Supporting Information for Proteomics DOI 10.1002/Pmic.200401292
Supporting Information for Proteomics DOI 10.1002/pmic.200401292 Robin Wait, Giulia Chiesa, Cinzia Parolini, Ingrid Miller, Shajna Begum, Daniela Brambilla, Lara Galluccio, Rossana Ballerio, Ivano Eberini, and Elisabetta Gianazza Reference maps of mouse serum acute-phase proteins. Changes with LPS-induced inflammation and apolipoprotein A-I and A-II transgenes ª 2005 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim www.proteomics-journal.de Reference maps of mouse serum acute-phase proteins. Changes with LPS-induced inflammation and apolipoprotein A-I and A-II transgenes Robin Wait1, Giulia Chiesa2, Cinzia Parolini2, Ingrid Miller5, Shajna Begum1, Daniela Brambilla2, Lara Galluccio2, Rossana Ballerio6, Ivano Eberini2,3,4, and Elisabetta Gianazza2,3,4 1Kennedy Institute of Rheumatology Division, Faculty of Medicine, Imperial College London, UK 2Dipartimento di Scienze Farmacologiche, Università degli Studi di Milano, Milan, Italy 3Gruppo di Studio per la Proteomica e la Struttura delle Proteine, Università degli Studi di Milano, Milan, Italy 4Centro di Eccellenza sulle Malattie del Sistema Nervoso Centrale e Periferico, Università degli Studi di Milano, Milan, Italy 5Institut für Medizinische Chemie, Department für Naturwissenschaften, Veterinärmedizinische Universität Wien, Vienna, Austria 6Centro Cardiologico Monzino, IRCCS, Milan, Italy Correspondence: Dr. Elisabetta Gianazza, Dipartimento di Scienze Farmacologiche, via G. Balzaretti 9, 20133 Milan, Italy E-mail: [email protected] Fax: +39-02-503-18284 Abbreviations: α1G, α1-acid -

Prolonged Partial Thromboplastin Time Without Bleeding History; Fletcher Factor Deficiency
Prolonged Partial Thromboplastin Time Without Bleeding History; Fletcher Factor Deficiency Celalettin ÜSTÜN*, Anand JILLELLA*, Linda HENDRIKS*, Mary JONAH**, Ferdane KUTLAR*, Russell BURGESS*, Abdullah KUTLAR* * Section of Hematology Oncology, Department of Medicine, Medical College of Georgia, ** Department of Pathology, Medical College of Georgia, Augusta, USA ABSTRACT A 67-year-old patient was admitted to the hospital to perform an esophagogastrectomy because a lesion at the lower esophagus was strongly suspicious for cancer. Her medical history and her family his- tory were negative for bleeding tendency or thrombosis. Her activated partial thromboplastin time (aPTT) was prolonged (44 s) whereas her prothrombin time (PT) was normal (11 s) presurgery. Mixing of her plasma with normal plasma corrected her prolonged aPTT (27.9 s). Prolonged incubation shortened the patient’s aPTT (36.3 s). Fletcher factor activity was found to be 50%. The patient underwent an esopha- gogastrectomy without bleeding complications under spinal anesthesia. Fletcher factor deficiency, a ra- re disorder, should be considered in patients who have no history of bleeding tendency with a prolonged aPTT. Surgical interventions are safe in these patients. Key Words: aPTT, Surgery, Fletcher factor, Prekallikrein. Turk J Haematol 2002;19(3): 417-419 Received: 20.06.2001 Accepted: 28.07.2001 INTRODUCTION unt[2]. Hematologists are frequently consulted for preoperative coagulation abnormalities. Preoperative evaluation of hemostasis is cru- cial to assess the risk of per-and peri-operative Prolonged aPTT is not an uncommon abnor- bleeding. The most effective screening method is mality encountered during preoperative evaluati- to obtain a thorough history of bleeding[1]. on. It may indicate the presence of antiphospholi- Preoperative screening tests mostly include acti- pid antibodies or a factor deficiency in the intrinsic vated partial thromboplastin time (aPTT), proth- and/or common pathways of blood coagulation. -

ELISA Kit for Hemopexin (HPX)
SEB986Ra 96 Tests Enzyme-linked Immunosorbent Assay Kit For Hemopexin (HPX) Organism Species: Rattus norvegicus (Rat) Instruction manual FOR IN VITRO AND RESEARCH USE ONLY NOT FOR USE IN CLINICAL DIAGNOSTIC PROCEDURES 11th Edition (Revised in July, 2013) [ INTENDED USE ] The kit is a sandwich enzyme immunoassay for in vitro quantitative measurement of hemopexin in rat serum, plasma and other biological fluids. [ REAGENTS AND MATERIALS PROVIDED ] Reagents Quantity Reagents Quantity Pre-coated, ready to use 96-well strip plate 1 Plate sealer for 96 wells 4 Standard 2 Standard Diluent 1×20mL Detection Reagent A 1×120μL Assay Diluent A 1×12mL Detection Reagent B 1×120μL Assay Diluent B 1×12mL TMB Substrate 1×9mL Stop Solution 1×6mL Wash Buffer (30 × concentrate) 1×20mL Instruction manual 1 [ MATERIALS REQUIRED BUT NOT SUPPLIED ] 1. Microplate reader with 450 ± 10nm filter. 2. Precision single or multi-channel pipettes and disposable tips. 3. Eppendorf Tubes for diluting samples. 4. Deionized or distilled water. 5. Absorbent paper for blotting the microtiter plate. 6. Container for Wash Solution [ STORAGE OF THE KITS ] 1. For unopened kit: All the reagents should be kept according to the labels on vials. The Standard, Detection Reagent A, Detection Reagent B and the 96-well strip plate should be stored at -20oC upon receipt while the others should be at 4 oC. 2. For opened kit: When the kit is opened, the remaining reagents still need to be stored according to the above storage condition. Besides, please return the unused wells to the foil pouch containing the desiccant pack, and reseal along entire edge of zip-seal. -

Haptoglobin and Its Related Protein, Zonulin—What Is Their Role in Spondyloarthropathy?
Journal of Clinical Medicine Review Haptoglobin and Its Related Protein, Zonulin—What Is Their Role in Spondyloarthropathy? Magdalena Chmieli ´nska 1,2,* , Marzena Olesi ´nska 2, Katarzyna Romanowska-Próchnicka 1,2 and Dariusz Szukiewicz 1 1 Department of Biophysics and Human Physiology, Medical University of Warsaw, Chałubi´nskiego5, 02-004 Warsaw, Poland; [email protected] (K.R.-P.); [email protected] (D.S.) 2 Department of Connective Tissue Diseases, National Institute of Geriatrics, Rheumatology and Rehabilitation, Sparta´nska1, 02-637 Warsaw, Poland; [email protected] * Correspondence: [email protected] Abstract: Haptoglobin (Hp) is an acute phase protein which supports the immune response and protects tissues from free radicals. Its concentration correlates with disease activity in spondy- loarthropathies (SpAs). The Hp polymorphism determines the functional differences between Hp1 and Hp2 protein products. The role of the Hp polymorphism has been demonstrated in many diseases. In particular, the Hp 2-2 phenotype has been associated with the unfavorable course of some inflammatory and autoimmune disorders. Its potential role in modulating the immune system in SpA is still unknown. This article contains pathophysiological considerations on the potential relationship between Hp, its polymorphism and SpA. Keywords: haptoglobin polymorphism; inflammation; pathogenesis; spondyloarthropathy; zonulin Citation: Chmieli´nska,M.; Olesi´nska, M.; Romanowska-Próchnicka, K.; Szukiewicz, D. Haptoglobin and Its 1. Introduction Related Protein, Zonulin—What Is Spondyloarthropathy is one of the most common rheumatic diseases whose prevalence Their Role in Spondyloarthropathy? J. varies between 0.4 and 1.9% in different countries [1]. The heterogeneity of SpA is the Clin. Med. 2021, 10, 1131. -

Downloaded from Bioscientifica.Com at 09/25/2021 07:25:24AM Via Free Access 812 M Andreassen and Others EUROPEAN JOURNAL of ENDOCRINOLOGY (2012) 166
European Journal of Endocrinology (2012) 166 811–819 ISSN 0804-4643 CLINICAL STUDY GH activity and markers of inflammation: a crossover study in healthy volunteers treated with GH and a GH receptor antagonist Mikkel Andreassen1, Jan Frystyk2,3, Jens Faber1,4 and Lars Østergaard Kristensen1 1Endocrine Unit, Laboratory of Endocrinology 54o4, Department of Internal Medicine O, Herlev Hospital, University of Copenhagen, Herlev Ringvej 75, DK-2730 Herlev, Denmark, 2Department of Endocrinology and Internal Medicine, Aarhus University Hospital, Aarhus, Denmark and 3Medical Research Laboratories, Faculty of Health Sciences, Institute of Clinical Medicine, Aarhus University, Aarhus, Denmark and 4Faculty of Health Science, Copenhagen University, Copenhagen, Denmark (Correspondence should be addressed to M Andreassen; Email: [email protected]) Abstract Introduction: The GH/IGF1 axis may modulate inflammatory processes. However, the relationship seems complicated as both pro- and anti-inflammatory effects have been demonstrated. Methods/design: Twelve healthy volunteers (mean age 36, range 27–49 years) were treated in random order with increasing doses of GH for 3 weeks (first week 0.01 mg/kg per day, second week 0.02 mg/kg per day, and third week 0.03 mg/kg per day) or a GH receptor antagonist (pegvisomant; first week 10 mg/day and last two weeks 15 mg/day), separated by 8 weeks of washout. Circulating levels of the pro-inflammatory cytokines tumor necrosis factor a (TNFa (TNFA)), interleukin 6 (IL6), and IL1b (IL1B) and the acute phase proteins (APPs) C-reactive protein (CRP), haptoglobin, orosomucoid, YKL40 (CHI3L1), and fibrinogen were measured. Results: During GH treatment, IGF1 (median 131 (Inter-quartile range (IQR) 112–166) vs 390 (322– 524) mg/l, PZ0.002) increased together with TNFa (0.87 (0.74–1.48) vs 1.27 (0.80–1.69) ng/l, PZ0.003), IL6 (1.00 (0.83–1.55) vs 1.35 (0.80–4.28) ng/l, PZ0.045), and fibrinogen (9.2 (8.8–9.6) vs 11.1 (9.4–12.4) mM, PZ0.002). -

Approach to Anemia
APPROACH TO ANEMIA Mahsa Mohebtash, MD Medstar Union Memorial Hospital Definition of Anemia • Reduced red blood mass • RBC measurements: RBC mass, Hgb, Hct or RBC count • Hgb, Hct and RBC count typically decrease in parallel except in severe microcytosis (like thalassemia) Normal Range of Hgb/Hct • NL range: many different values: • 2 SD below mean: < Hgb13.5 or Hct 41 in men and Hgb 12 or Hct of 36 in women • WHO: Hgb: <13 in men, <12 in women • Revised WHO/NCI: Hgb <14 in men, <12 in women • Scrpps-Kaiser based on race and age: based on 5th percentiles of the population in question • African-Americans: Hgb 0.5-1 lower than Caucasians Approach to Anemia • Setting: • Acute vs chronic • Isolated vs combined with leukopenia/thrombocytopenia • Pathophysiologic approach • Morphologic approach Reticulocytes • Reticulocytes life span: 3 days in bone marrow and 1 day in peripheral blood • Mature RBC life span: 110-120 days • 1% of RBCs are removed from circulation each day • Reticulocyte production index (RPI): Reticulocytes (percent) x (HCT ÷ 45) x (1 ÷ RMT): • <2 low Pathophysiologic approach • Decreased RBC production • Reduced effective production of red cells: low retic production index • Destruction of red cell precursors in marrow (ineffective erythropoiesis) • Increased RBC destruction • Blood loss Reduced RBC precursors • Low retic production index • Lack of nutrients (B12, Fe) • Bone marrow disorder => reduced RBC precursors (aplastic anemia, pure RBC aplasia, marrow infiltration) • Bone marrow suppression (drugs, chemotherapy, radiation) -

Haptoglobin 9D91-20 30-3966/R4
HAPTOGLOBIN 9D91-20 30-3966/R4 HAPTOGLOBIN This package insert contains information to run the Haptoglobin assay on the ARCHITECT c Systems™ and the AEROSET System. NOTE: Changes Highlighted NOTE: This package insert must be read carefully prior to product use. Package insert instructions must be followed accordingly. Reliability of assay results cannot be guaranteed if there are any deviations from the instructions in this package insert. Customer Support United States: 1-877-4ABBOTT Canada: 1-800-387-8378 (English speaking customers) 1-800-465-2675 (French speaking customers) International: Call your local Abbott representative Symbols in Product Labeling Calibrators 1 through 5 Catalog number/List number Concentration Serial number Authorized Representative in the Consult instructions for use European Community Ingredients Manufacturer In vitro diagnostic medical device Temperature limitation Batch code/Lot number Use by/Expiration date Reagent 1 Reagent 2 ABBOTT LABORATORIES ABBOTT Abbott Park, IL 60064, USA Max-Planck-Ring 2 65205 Wiesbaden Germany +49-6122-580 May 2007 ©2004, 2007 Abbott Laboratories 1 NAME REAGENT HANDLING AND STORAGE HAPTOGLOBIN Reagent Handling Remove air bubbles, if present in the reagent cartridge, with a new INTENDED USE applicator stick. Alternatively, allow the reagent to sit at the appropriate The Haptoglobin assay is used for the quantitation of haptoglobin in storage temperature to allow the bubbles to dissipate. To minimize human serum or plasma. volume depletion, do not use a transfer pipette to remove the bubbles. CAUTION: Reagent bubbles may interfere with proper detection of SUMMARY AND EXPLANATION OF TEST reagent level in the cartridge, causing insufficient reagent aspiration Haptoglobin is a protein synthesized in the liver that binds with the which could impact results. -

BAFF Expression Is Increased in Patients
g in Geno nin m i ic M s ta & a P Eilertsen, J Data Mining in Genom Proteomics 2012, 3:1 D r f o Journal of o t e l DOI: 10.4172/2153-0602.1000113 o a m n r i c u s o J ISSN: 2153-0602 Data Mining in Genomics & Proteomics Research Article Open Access BAFF Expression is Increased in Patients with Lupus Nephritis and Associated with Antinucleosome Antibodies, C1 Inhibitor, Α-1-Acid- Glycoprotein and Endothelial Activation Markers G. Ø. Eilertsen¹*, M. Van Ghelue² and J.C. Nossent¹ ¹Bone and Joint research group, Department of Health Science, Medical School, University of Tromsø, Norway ²Medical Genetics Department, University Hospital North Norway, Tromsø, Norway Abstract Objectives: B cell activating factor (BAFF) inhibitor therapy has recently been approved for non-renal Systemic Lupus Erythematosus (SLE). While BAFF plays a role in experimental lupus nephritis (LN), its role human LN is not well studied. Methods: Case control study in 102 SLE patients, 30 with LN (+LN) and 72 without LN (-LN) and 31 healthy controls. We analysed BAFF mRNA expression in PBMCs (BAFF-RQ) and serum BAFF (s-BAFF) levels and investigated their relation with clinical, histological- and additional acute phase proteins. Results: s-BAFF and BAFF-RQ were increased in +LN patients compared to controls, but their expression did not correlate with ISN/RPS class, Activity- or Chronicity index on biopsy. s-BAFF correlated with levels of anti-nucleosome antibodies, C1 inhibitor and α-1-acid-glycoprotein (AGP), while BAFF-RQ correlated inversely with Factor VIII.