Analyse Comparative, Physiologique Et Moléculaire Des Effets De Trois
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Critical Role of Oxidative Stress in Estrogen-Induced Carcinogenesis
Critical role of oxidative stress in estrogen-induced carcinogenesis Hari K. Bhat*†, Gloria Calaf‡, Tom K. Hei*‡, Theresa Loya§, and Jaydutt V. Vadgama¶ *Department of Environmental Health Sciences, Mailman School of Public Health, 60 Haven Avenue-B1, Columbia University, New York, NY 10032; ‡Center for Radiological Research, Columbia University, New York, NY 10032; and Departments of §Pathology and ¶Medicine, Charles Drew University, Los Angeles, CA 90059 Communicated by Donald C. Malins, Pacific Northwest Research Institute, Seattle, WA, December 27, 2002 (received for review August 22, 2002) Mechanisms of estrogen-induced tumorigenesis in the target quinones generates oxidative stress and potentially harmful free organ are not well understood. It has been suggested that oxida- radicals that are postulated to be required for the carcinogenic tive stress resulting from metabolic activation of carcinogenic process, and analogous to the metabolic activation of hydrocar- estrogens plays a critical role in estrogen-induced carcinogenesis. bons and other nonsteroidal estrogen carcinogens (9, 19–22). We We tested this hypothesis by using an estrogen-induced hamster have investigated the role of oxidative stress in estrogen carci- renal tumor model, a well established animal model of hormonal nogenesis by using a well established hamster renal tumor model carcinogenesis. Hamsters were implanted with 17-estradiol (E2), that shares several characteristics with human breast and uterine 17␣-estradiol (␣E2), 17␣-ethinylestradiol (␣EE), menadione, a com- cancers, pointing to a common mechanistic origin (6, 9, 23). bination of ␣E2 and ␣EE, or a combination of ␣EE and menadione Different estrogens used in the present study differ in their for 7 months. -

Pp375-430-Annex 1.Qxd
ANNEX 1 CHEMICAL AND PHYSICAL DATA ON COMPOUNDS USED IN COMBINED ESTROGEN–PROGESTOGEN CONTRACEPTIVES AND HORMONAL MENOPAUSAL THERAPY Annex 1 describes the chemical and physical data, technical products, trends in produc- tion by region and uses of estrogens and progestogens in combined estrogen–progestogen contraceptives and hormonal menopausal therapy. Estrogens and progestogens are listed separately in alphabetical order. Trade names for these compounds alone and in combination are given in Annexes 2–4. Sales are listed according to the regions designated by WHO. These are: Africa: Algeria, Angola, Benin, Botswana, Burkina Faso, Burundi, Cameroon, Cape Verde, Central African Republic, Chad, Comoros, Congo, Côte d'Ivoire, Democratic Republic of the Congo, Equatorial Guinea, Eritrea, Ethiopia, Gabon, Gambia, Ghana, Guinea, Guinea-Bissau, Kenya, Lesotho, Liberia, Madagascar, Malawi, Mali, Mauritania, Mauritius, Mozambique, Namibia, Niger, Nigeria, Rwanda, Sao Tome and Principe, Senegal, Seychelles, Sierra Leone, South Africa, Swaziland, Togo, Uganda, United Republic of Tanzania, Zambia and Zimbabwe America (North): Canada, Central America (Antigua and Barbuda, Bahamas, Barbados, Belize, Costa Rica, Cuba, Dominica, El Salvador, Grenada, Guatemala, Haiti, Honduras, Jamaica, Mexico, Nicaragua, Panama, Puerto Rico, Saint Kitts and Nevis, Saint Lucia, Saint Vincent and the Grenadines, Suriname, Trinidad and Tobago), United States of America America (South): Argentina, Bolivia, Brazil, Chile, Colombia, Dominican Republic, Ecuador, Guyana, Paraguay, -

Dalton Pharma Catalogue Steroids 12-19-12
2012/2013 Version: December 31, 2012 Steroid Analytical Reference Standards and Drug Impurities Dalton Pharma Services: A Health Canada Approved Facility Level-8 Controlled Substances License on-site SCC Approved GLP Facility North American Manufactured and Shipped To Order Call: 1.800.567.5060 To Order Call: 416.661.2102 Email: [email protected] Email: [email protected] Page 2 Dalton Standards and Impurities Table of Contents Introduction/Company Profile 4 Custom Synthesis Overview 6 Contract Research/Medicinal Chemistry 7 Process Development Overview 8 Analytical Services 9 GMP API & Sterile Filling Services 11 Formulation Development Overview 12 Liposomes 13 Dalton Standards and Impurities 14 Establishment License 16 Certificate example 17 Steroids 18 Page 3 To Order Call: 1.800.567.5060 To Order Email: [email protected] Call: Company Profile About Dalton Pharma Services Dalton Pharma Services is a privately-held pharmaceutical services company that has been producing Fine Chemi- cals on a custom basis for research and chemical supply houses for over twenty-five years (Fluka, Aldrich, Sigma, Acros). We have identified and supplied many new re- agents for biological and pharmaceutical applications, this includes novel analogues and impurities of active pharma- ceuticals ingredients, as well as new linkers. We are lead- ers in process development, process improvement, and in the field of isolation of kilo quantities of biologically active molecules from natural sources. Dalton excels at advancing projects from R&D into GMP environments for its customers. With the ability to manufacture cGMP API's from bench scales to multi kilos we can meet most clinical and small scale commercial requirements. -

Porous Drug Matrices and Methods of Manufacture
(19) & (11) EP 1 180 020 B2 (12) NEW EUROPEAN PATENT SPECIFICATION After opposition procedure (45) Date of publication and mention (51) Int Cl.: of the opposition decision: A61K 9/16 (2006.01) 24.06.2009 Bulletin 2009/26 (86) International application number: (45) Mention of the grant of the patent: PCT/US2000/014578 14.12.2005 Bulletin 2005/50 (87) International publication number: (21) Application number: 00939365.3 WO 2000/072827 (07.12.2000 Gazette 2000/49) (22) Date of filing: 25.05.2000 (54) POROUS DRUG MATRICES AND METHODS OF MANUFACTURE THEREOF PORÖSE ARZNEISTOFFMATRIZEN UND DEREN HERSTELLUNGSVERFAHREN MATRICES MEDICAMENTEUSES POREUSES ET PROCEDES DE FABRICATION ASSOCIES (84) Designated Contracting States: • BERNSTEIN, Howard AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU Cambridge, MA 02138 (US) MC NL PT SE • CHICKERING, Donald, E. III Framingham, MA 01701 (US) (30) Priority: 27.05.1999 US 136323 P • KHATAK, Sarwat 08.10.1999 US 158659 P Hadley-Massachusetts 01035 (US) 04.11.1999 US 433486 • RANDALL, Greg 02.03.2000 US 186310 P Stoneham, MA 02180 (US) (43) Date of publication of application: (74) Representative: HOFFMANN EITLE 20.02.2002 Bulletin 2002/08 Patent- und Rechtsanwälte Arabellastraße 4 (60) Divisional application: 81925 München (DE) 05027194.9 / 1 642 572 (56) References cited: (73) Proprietor: Acusphere, Inc. EP-A- 0 655 237 WO-A-98/31346 Watertown, MA 02472 (US) WO-A-99/56731 WO-A1-98/31346 WO-A1-99/56731 DE-A- 3 713 326 (72) Inventors: GB-A- 1 265 615 US-A- 3 948 245 • STRAUB, Julie US-A- 4 687 660 Winchester, MA 01890 (US) EP 1 180 020 B2 Printed by Jouve, 75001 PARIS (FR) EP 1 180 020 B2 Description Background of the Invention 5 [0001] This invention generally relates to formulations of drugs, especially drugs having low solubility, and more particularly to methods of making formulations of such drugs to enhance their rate of dissolution. -

United States Patent Office Patented May 21, 1957 1
2,793,218 United States Patent Office Patented May 21, 1957 1. 2,793,218 9-HALO-11.KETO.17-ALKYTLTESTOSTERONES Milton E. Herr, Kalamazoo, Mich., assignor to The Up john Company, Kalamazoo, Mich., a corporation of Michigan No Drawing. Original application August 8, 1955, Serial No. 527,118. Divided and this application May 10, WII 1956, Serial No. 583,922 wherein R is a lower-alkyl group containing less than three carbon atoms, i. e., methyl or ethyl; R is hydrogen 3 Claims. (Cl. 260-397.45) or the acyl radical of a hydrocarbon carboxylic acid con taining from one to twelve carbon atoms, inclusive; X This invention relates to novel 17-alkyl-17-hydroxy 5 is a halogen having an atomic weight from 79 to 127, in steroids and esters thereof. clusive, i. e., bromine or iodine, X is a halogen having It is an object of this invention to provide novel 9a an atomic weight from 19 to 36, inclusive, i. e., fluorine halo - 116 - hydroxy - 17 - alkyltestosterones, 9a - halo or chlorine, and X' is a halogen having an atomic weight 11 - keto - 17 - alkyltestosterones, 17-esters thereof, and from 19 to 127, inclusive, i. e., fluorine, chlorine, bromine intermediates in the production thereof. Another object 20 or iodine. is the provision of processes for the production thereof. Following the series of reactions described hereinafter Other objects will be apparent to those skilled in the art for the conversion of 11o - hydroxy - 17 - methyltestos to which this invention pertains. terone (I) to 9a - hydroxy - 17 - methyltestosterone According to the present invention, the novel 9a-halo testosterone and esters thereof (VII), but substituting 10 116 - hydroxy - 17 - alkyltestosterones, 9a - halo - 11 25 normethyl - 11a - hydroxy - 17 - methyltestosterone keto - 17 - alkyltestosterones and 17 - esters thereof may (U. -

United States Patent (19) 11 Patent Number: 6,068,830 Diamandis Et Al
US00606883OA United States Patent (19) 11 Patent Number: 6,068,830 Diamandis et al. (45) Date of Patent: May 30, 2000 54) LOCALIZATION AND THERAPY OF FOREIGN PATENT DOCUMENTS NON-PROSTATIC ENDOCRINE CANCER 0217577 4/1987 European Pat. Off.. WITH AGENTS DIRECTED AGAINST 0453082 10/1991 European Pat. Off.. PROSTATE SPECIFIC ANTIGEN WO 92/O1936 2/1992 European Pat. Off.. WO 93/O1831 2/1993 European Pat. Off.. 75 Inventors: Eleftherios P. Diamandis, Toronto; Russell Redshaw, Nepean, both of OTHER PUBLICATIONS Canada Clinical BioChemistry vol. 27, No. 2, (Yu, He et al), pp. 73 Assignee: Nordion International Inc., Canada 75-79, dated Apr. 27, 1994. Database Biosis BioSciences Information Service, AN 21 Appl. No.: 08/569,206 94:393008 & Journal of Clinical Laboratory Analysis, vol. 8, No. 4, (Yu, He et al), pp. 251-253, dated 1994. 22 PCT Filed: Jul. 14, 1994 Bas. Appl. Histochem, Vol. 33, No. 1, (Papotti, M. et al), 86 PCT No.: PCT/CA94/00392 Pavia pp. 25–29 dated 1989. S371 Date: Apr. 11, 1996 Primary Examiner Yvonne Eyler S 102(e) Date: Apr. 11, 1996 Attorney, Agent, or Firm-Banner & Witcoff, Ltd. 87 PCT Pub. No.: WO95/02424 57 ABSTRACT It was discovered that prostate-specific antigen is produced PCT Pub. Date:Jan. 26, 1995 by non-proStatic endocrine cancers. It was further discov 30 Foreign Application Priority Data ered that non-prostatic endocrine cancers with Steroid recep tors can be stimulated with Steroids to cause them to produce Jul. 14, 1993 GB United Kingdom ................... 93.14623 PSA either initially or at increased levels. -

WO 2018/060501 A2 05 April 2018 (05.04.2018) W ! P O PCT
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2018/060501 A2 05 April 2018 (05.04.2018) W ! P O PCT (51) International Patent Classification: (71) Applicants: MYOVANT SCIENCES GMBH [CH/CH]; A61K 31/513 (2006.01) Viaduktstrasse 8, 405 1 Basel (CH). TAKEDA PHAR¬ MACEUTICAL COMPANY LIMITED [JP/JP]; 1-1, (21) International Application Number: Doshomachi 4-chome, Chuo-ku, Osaka-shi, Osaka, PCT/EP20 17/074907 541-0045 (JP). (22) International Filing Date: (72) Inventors: JOHNSON, Brendan Mark; 2017 Markham 29 September 2017 (29.09.2017) Drive, Chapel Hill, 275 14 (NC). SEELY, Lynn; 537 Occi (25) Filing Language: English dental Avenue, San Mateo, 94402 (US). MUDD, JR., Paul N.; 302 Beacon Falls Court, Cary, North Carolina 27519 (26) Publication Langi English (US). WOLLOWITZ, Susan; 32 Topper Court, Lafayette, (30) Priority Data: California 94549 (US). HIBBERD, Mark; The Old House, 62/402,034 30 September 2016 (30.09.2016) US Hawkley, Liss Hampshire GU33 6NQ (GB). TANIMO- 62/402,055 30 September 2016 (30.09.2016) US TO, Masataka; c/o Takeda Pharmaceutical Company Lim 62/402,150 30 September 2016 (30.09.2016) US ited, 1-1, Doshomachi 4-chome, Chuo-ku, Osaka-shi, O sa 62/492,839 0 1 May 2017 (01 .05.2017) US ka, 541-0045 (JP). RAJASEKHAR, Vijaykumar Reddy; 62/528,409 03 July 2017 (03.07.2017) US 20200 Quail Hollow Road, Apple Valley, California 92308 (54) Title: METHODS OF TREATING UTERINE FIBROIDS AND ENDOMETRIOSIS PBAC=0 Lumbar BMD CD n co CD P g CO O O CD Dose (mg) FIG. -

Pros and Cons Controversy on Molecular Imaging and Dynamic
Open Access Archives of Biotechnology and Biomedicine Research Article Pros and Cons Controversy on Molecular Imaging and Dynamics of Double- ISSN Standard DNA/RNA of Human Preserving 2639-6777 Stem Cells-Binding Nano Molecules with Androgens/Anabolic Steroids (AAS) or Testosterone Derivatives through Tracking of Helium-4 Nucleus (Alpha Particle) Using Synchrotron Radiation Alireza Heidari* Faculty of Chemistry, California South University, 14731 Comet St. Irvine, CA 92604, USA *Address for Correspondence: Dr. Alireza Abstract Heidari, Faculty of Chemistry, California South University, 14731 Comet St. Irvine, CA 92604, In the current study, we have investigated pros and cons controversy on molecular imaging and dynamics USA, Email: of double-standard DNA/RNA of human preserving stem cells-binding Nano molecules with Androgens/ [email protected]; Anabolic Steroids (AAS) or Testosterone derivatives through tracking of Helium-4 nucleus (Alpha particle) using [email protected] synchrotron radiation. In this regard, the enzymatic oxidation of double-standard DNA/RNA of human preserving Submitted: 31 October 2017 stem cells-binding Nano molecules by haem peroxidases (or heme peroxidases) such as Horseradish Peroxidase Approved: 13 November 2017 (HPR), Chloroperoxidase (CPO), Lactoperoxidase (LPO) and Lignin Peroxidase (LiP) is an important process from Published: 15 November 2017 both the synthetic and mechanistic point of view. Copyright: 2017 Heidari A. This is an open access article distributed under the Creative -

WHO Drug Information Vol
WHO Drug Information Vol. 29, No. 2, 2015 WHO Drug Information Contents Regulatory collaboration 153 Transparency WHO calls for disclosure of clinical trial 127 The African Vaccine Regulatory Forum results; Australia adopts new regulator (AVAREF): A platform for collaboration in a performance framework public health emergency 154 Databases Health Canada launches searchable inspection database; WHO launches WHO prequalification open access to its global medicines safety 133 Update on prequalification of diagnostics and database; EMA to record adverse events from medicines literature in EudraVigilance 155 Approved Cholic acid: for rare bile acid synthesis disorders; Eluxadoline : for irritable bowel disease; Empaglifozin & Norms and standards metformin : for diabetes; Evolocumab : to lower cholesterol; Isavuconazonium 138 Biotherapeutics and biosimilars sulfate: for certain invasive fungal infections; Atazanavir & cobicistat: for treatment of HIV-1 infection; Anthrax immunoglobulin (human); Dinutuximab : to prolong survival in children with high-risk neuroblastoma; Filgrastim-sndz :, first biosimilar in the U.S.; Tasimelteon : to regulate sleep patterns in blind adults; Safety news 157 Extensions of indications 142 Restrictions Moxifloxacin : for treatment of plague; Sirolimus : for very rare lung disease; Bromhexine : not to be used in children under six in New Zealand; Codeine for cough and cold : not to be used in children under 12; 158 Generic 142 Safety warnings Glatiramer acetate : Sitagliptin : thrombocytopenia; SGLT2 inhibitor diabetes -

Adulteration of Dietary Supplements by the Illegal Addition of Synthetic Drugs: a Review Tiago Rocha, Joana S
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Biblioteca Digital do IPB Adulteration of Dietary Supplements by the Illegal Addition of Synthetic Drugs: A Review Tiago Rocha, Joana S. Amaral, and Maria Beatriz P.P. Oliveira Abstract: In the last few years, the consumption of dietary supplements, especially those having plants as ingredients, has been increasing due to the common idea that they are natural products posing no risks to human health. In the European Union and the United States, dietary supplements are legally considered as foods/special category of foods, thus are not being submitted to any safety assessment prior to their commercialization. Among the issues that can affect safety, adulteration by the illegal addition of pharmaceutical substances or their analogs is of major concern since unscrupulous producers can falsify these products to provide for quick effects and to increase sales. This review discusses the various classes of synthetic drugs most frequently described as being illegally added to dietary supplements marketed for weight loss, muscle building/sport performance and sexual performance enhancement. Information regarding regulation and consumption is also presented. Finally, several conventional and advanced analytical techniques used to detect and identify different adulterants in dietary supplements and therefore also in foods, with particular emphasis on plant food supplements, are critically described. This review demonstrates that dietary supplement adulteration is an emerging food safety problem and that an effective control by food regulatory authorities is needed to safeguard consumers. Keywords: adulteration, analogs, dietary supplements, food safety, pharmaceutical drugs, plant food supplements Introduction and Education Act (DSHEA), respectively, thus not requiring any In the last few years, the consumption of dietary supplements, safety assessment prior to their commercialization. -
C:\Documents and Settings\Ms785a\Desktop\Web
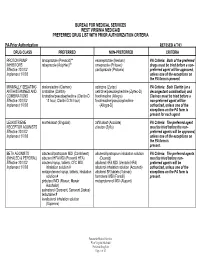
BUREAU FOR MEDICAL SERVICES WEST VIRGINIA MEDICAID PREFERRED DRUG LIST WITH PRIOR AUTHORIZATION CRITERIA PA-Prior Authorization REVISED 4/7/03 DRUG CLASS PREFERRED NON-PREFERRED CRITERIA PROTON PUMP lansoprazole (Prevacid)** esomeprazole (Nexium) PA Criteria: Both of the preferred INHIBITORS rabeprazole (AcipHex)** omeprazole (Prilosec) drugs must be tried before a non- Effective 10/1/02 pantoprazole (Protonix) preferred agent will be approved, Implement 1/7/03 unless one of the exceptions on the PA form is present. MINIMALLY SEDATING desloratadine (Clarinex) cetirizine (Zyrtec) PA Criteria: Both Claritin (or a ANTIHISTAMINES AND loratadine (Claritin) cetirizine/pseudoephedrine (Zyrtec-D) decongestant combination) and COMBINATIONS loratadine/pseudoephedrine (Claritin-D fexofenadine (Allegra) Clarinex must be tried before a Effective 10/1/02 12 hour, Claritin-D 24 hour) fexofenadine/pseudoephedrine non-preferred agent will be Implement 1/7/03 (Allegra-D) authorized, unless one of the exceptions on the PA form is present for each agent. LEUKOTRIENE montelukast (Singulair) zafirlukast (Accolate) PA Criteria: The preferred agent RECEPTOR AGONISTS zileuton (Zyflo) must be tried before the non- Effective 10/1/02 preferred agents will be approved, Implement 1/7/03 unless one of the exceptions on the PA form is present. BETA AGONISTS albuterol/ipratropium MDI (Combivent) albuterol/ipratropium inhalation solution PA Criteria: The preferred agents (INHALED & PERORAL) albuterol HFA MDI (Proventil HFA) (Duoneb) must be tried before non- Effective 10/1/02 albuterol syrup, tablets, CFC MDI, albuterol HFA MDI (Ventolin HFA) preferred agents will be Implement 1/7/03 inhalation solution # albuterol inhalation solution (Accuneb) authorized, unless one of the metaproterenol syrup, tablets, inhalation albuterol SR tablets (Volmax) exceptions on the PA form is solution # formoteral MDI (Foradil) present. -

Screening for Synthetic Steroids in Human Urine by LC-ESI-MS/MS
In: W Schänzer, H Geyer, A Gotzmann, U Mareck (eds.) Recent Advances In Doping Analysis (13). Sport und Buch Strauß - Köln 2005 Mario Thevis, Hans Geyer, Ute Mareck and Wilhelm Schänzer Screening for Synthetic Steroids in Human Urine by LC-ESI-MS/MS Institute of Biochemistry, German Sport University, Cologne, Germany Introduction The problem of unknown synthetic steroids in sports has become evident during the last few years. The anabolic steroid norbolethone (13-ethyl-17-hydroxy-18,19-dinor-17α-pregn-4-en- 3-one) was detected in a doping control urine sample in 2002 [1] although this compound has never been commercially available, and tetrahydrogestrinone (THG, 13-ethyl-17-hydroxy- 18,19-dinor-17α-pregn-4,9,11-trien-3-one) substantiated the fact that designer steroids are abused in the world of sport [2-5] in 2004. In February 2005, it was found that another steroid termed ‘madol’ (17α-methyl-5α-androst-2-ene-17β-ol, also known as desoxy- methyltestosterone (DMT)) [6, 7] may also have been used by athletes. The primary methods of drug screening are based on the mass spectrometric identification of known compounds. Conventional assays will not detect unknown compounds differing by as little as 1 or 2 Daltons from previously identified compounds, especially for MS/MS experiments where defined precursor ions are selected for collision-induced dissociation (CID). In this study a complementary screening procedure is reported allowing the detection of new or unknown steroids with distinct nuclei in human urine. Complementary urinary “steroid profiles” are generated from precursor ion scan experiments using LC-ESI-MS/MS, a strategy also commonly employed also in metabolite identification studies.