Mapping the Cell Symposium: RNA Platform
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Realizing the Allosteric Potential of the Tetrameric Protein Kinase a RIΑ Holoenzyme
Structure Article Realizing the Allosteric Potential of the Tetrameric Protein Kinase A RIa Holoenzyme Angela J. Boettcher,1,6 Jian Wu,1,6 Choel Kim,2 Jie Yang,1 Jessica Bruystens,1 Nikki Cheung,1 Juniper K. Pennypacker,1,3 Donald A. Blumenthal,4 Alexandr P. Kornev,3,5 and Susan S. Taylor1,3,5,* 1Department of Chemistry and Biochemistry, University of California at San Diego, La Jolla, CA 92093, USA 2Department of Pharmacology, Baylor College of Medicine, Houston, TX 77030, USA 3Department of Pharmacology, University of California at San Diego, La Jolla, CA 92093, USA 4Department of Pharmacology and Toxicology, University of Utah, Salt Lake City, UT 84112, USA 5Howard Hughes Medical Institute, University of California at San Diego, La Jolla, CA 92093, USA 6These authors contributed equally to this work *Correspondence: [email protected] DOI 10.1016/j.str.2010.12.005 SUMMARY the active site cleft in the C subunit in the inactive holoenzyme but is disordered in the dissociated free R subunits (Li et al., PKA holoenzymes containing two catalytic (C) 2000). The linker, as summarized in Figure 1, can be divided subunits and a regulatory (R) subunit dimer are acti- into three segments, the consensus inhibitor site (P-3 to P+1), vated cooperatively by cAMP. While cooperativity the N-linker that joins the inhibitor site to the D/D domain, and involves the two tandem cAMP binding domains in the C-linker that becomes ordered in the heterodimeric holoen- each R-subunit, additional cooperativity is associ- zyme complex. While much has been learned from the structures ated with the tetramer. -

TRP Channel Transient Receptor Potential Channels
TRP Channel Transient receptor potential channels TRP Channel (Transient receptor potential channel) is a group of ion channels located mostly on the plasma membrane of numerous human and animal cell types. There are about 28 TRP channels that share some structural similarity to each other. These are grouped into two broad groups: Group 1 includes TRPC ("C" for canonical), TRPV ("V" for vanilloid), TRPM ("M" for melastatin), TRPN, and TRPA. In group 2, there are TRPP ("P" for polycystic) and TRPML ("ML" for mucolipin). Many of these channels mediate a variety of sensations like the sensations of pain, hotness, warmth or coldness, different kinds of tastes, pressure, and vision. TRP channels are relatively non-selectively permeable to cations, including sodium, calcium and magnesium. TRP channels are initially discovered in trp-mutant strain of the fruit fly Drosophila. Later, TRP channels are found in vertebrates where they are ubiquitously expressed in many cell types and tissues. TRP channels are important for human health as mutations in at least four TRP channels underlie disease. www.MedChemExpress.com 1 TRP Channel Inhibitors, Antagonists, Agonists, Activators & Modulators (-)-Menthol (E)-Cardamonin Cat. No.: HY-75161 ((E)-Cardamomin; (E)-Alpinetin chalcone) Cat. No.: HY-N1378 (-)-Menthol is a key component of peppermint oil (E)-Cardamonin ((E)-Cardamomin) is a novel that binds and activates transient receptor antagonist of hTRPA1 cation channel with an IC50 potential melastatin 8 (TRPM8), a of 454 nM. Ca2+-permeable nonselective cation channel, to 2+ increase [Ca ]i. Antitumor activity. Purity: >98.0% Purity: 99.81% Clinical Data: Launched Clinical Data: No Development Reported Size: 10 mM × 1 mL, 500 mg, 1 g Size: 10 mM × 1 mL, 5 mg, 10 mg, 25 mg, 50 mg, 100 mg (Z)-Capsaicin 1,4-Cineole (Zucapsaicin; Civamide; cis-Capsaicin) Cat. -

A Perspective on Mechanisms of Protein Tetramer Formation
Biophysical Journal Volume 85 December 2003 3587–3599 3587 A Perspective on Mechanisms of Protein Tetramer Formation Evan T. Powers* and David L. Powersy *Department of Chemistry, The Scripps Research Institute, La Jolla, California 92037; and yDepartment of Mathematics and Computer Science, Clarkson University, Potsdam, New York 13699 ABSTRACT Homotetrameric proteins can assemble by several different pathways, but have only been observed to use one, in which two monomers associate to form a homodimer, and then two homodimers associate to form a homotetramer. To determine why this pathway should be so uniformly dominant, we have modeled the kinetics of tetramerization for the possible pathways as a function of the rate constants for each step. We have found that competition with the other pathways, in which homotetramers can be formed either by the association of two different types of homodimers or by the successive addition of monomers to homodimers and homotrimers, can cause substantial amounts of protein to be trapped as intermediates of the assembly pathway. We propose that this could lead to undesirable consequences for an organism, and that selective pressure may have caused homotetrameric proteins to evolve to assemble by a single pathway. INTRODUCTION Many proteins must be homotetrameric to be functional. ‘‘MDT’’ stands for monomer-dimer-tetramer and ‘‘a’’ and Prominent examples include transcription factors (e.g., p53) ‘‘b’’ indicate the type of homodimer formed. The pathways (Friedman et al., 1993), transport proteins (e.g., trans- that include monomers, homodimers, homotrimers, and thyretin) (Blake et al., 1974), potassium channels (Deutsch, homotetramers will be denoted MDRT, which stands for 2002; Miller, 2000), water channels (Fujiyoshi et al., 2002), monomer-dimer-trimer-tetramer. -

FEPS 2019 – BOLOGNA (ITALY) Abstracts of the Joint Meeting
www.actaphysiol.org September 2019 • Volume 227 • Supplement 718 OFFICIAL JOURNAL OF THE FEDERATION OF EUROPEAN PHYSIOLOGICAL SOCIETIES FEPS 2019 – BOLOGNA (ITALY) Joint Meeting of the Federation of European Physiological Societies (FEPS) and the Italian Physiological Society (SIF) Bologna (Italy), September 10th – 13th 2019 Abstracts of the Joint Meeting A Joint International Meeting celebrating the 70th Anniversary of the Italian Physiological Society PUBLICATION HISTORY Acta Physiologica 2006– Acta Physiologica Scandinavica 1940–2005 Skandinavisches Archiv für Physiologie 1889–1939 AAPHA_v227_s718_issueinfo.inddPHA_v227_s718_issueinfo.indd 1 88/28/2019/28/2019 9:07:319:07:31 AAMM Chief Editor INFORMATION FOR SUBSCRIBERS Pontus B. Persson, Berlin Acta Physiologica is published in 12 issues per year. Subscription prices for 2019 € Editors are: Institutional: 1059 (Europe), $1582 (The Americas), $1849 (Rest of World). Cardiovascular Physiology – Frantisek Kolar, Prague; Holger Nilsson, Gothenburg Prices are exclusive of tax. Australian GST, Canadian GST/HST and European and William E. Louch, Oslo VAT will be applied at the appropriate rates. For more information on current tax Cell Biology – Sari Lauri, Helsinki rates, please go to www.wileyonlinelibrary.com/tax-vat. The price includes online Chronobiology and Endocrinology – Charna Dibner, Geneva access to the current and all online back fi les to January 1st 2015, where available. Exercise Physiology – Jan Henriksson, Stockholm For other pricing options, including access information -

HOOK™ Maleimide Activated Streptavidin for Conjugation of Streptavidin to Sulfhydryl Groups Containing Proteins, Peptides and Ligands
G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name HOOK™ Maleimide Activated Streptavidin For conjugation of Streptavidin to sulfhydryl groups containing proteins, peptides and ligands (Cat. #786-1653, 786-1654) think proteins! think G-Biosciences www.GBiosciences.com INTRODUCTION ................................................................................................................. 3 ITEMS SUPPLIED ................................................................................................................ 4 STORAGE CONDITIONS ...................................................................................................... 4 ADDITIONAL ITEMS NEEDED .............................................................................................. 4 IMPORTANT INFORMATION .............................................................................................. 4 PROTOCOL ......................................................................................................................... 4 PREPARATION OF PROTEIN FOR CONJUGATION TO MALEIMIDE ACTIVATED PROTEIN 4 CONJUGATION REACTION ............................................................................................. 5 STORAGE OF CONJUGATED ANTIBODIES/PROTEINS ......................................................... 5 RELATED PRODUCTS .......................................................................................................... 5 Page 2 of 6 INTRODUCTION Streptavidin is a non-glycosylated -

Cryo-EM Structure of the Polycystic Kidney Disease-Like Channel PKD2L1
ARTICLE DOI: 10.1038/s41467-018-03606-0 OPEN Cryo-EM structure of the polycystic kidney disease-like channel PKD2L1 Qiang Su1,2,3, Feizhuo Hu1,3,4, Yuxia Liu4,5,6,7, Xiaofei Ge1,2, Changlin Mei8, Shengqiang Yu8, Aiwen Shen8, Qiang Zhou1,3,4,9, Chuangye Yan1,2,3,9, Jianlin Lei 1,2,3, Yanqing Zhang1,2,3,9, Xiaodong Liu2,4,5,6,7 & Tingliang Wang1,3,4,9 PKD2L1, also termed TRPP3 from the TRPP subfamily (polycystic TRP channels), is involved 1234567890():,; in the sour sensation and other pH-dependent processes. PKD2L1 is believed to be a non- selective cation channel that can be regulated by voltage, protons, and calcium. Despite its considerable importance, the molecular mechanisms underlying PKD2L1 regulations are largely unknown. Here, we determine the PKD2L1 atomic structure at 3.38 Å resolution by cryo-electron microscopy, whereby side chains of nearly all residues are assigned. Unlike its ortholog PKD2, the pore helix (PH) and transmembrane segment 6 (S6) of PKD2L1, which are involved in upper and lower-gate opening, adopt an open conformation. Structural comparisons of PKD2L1 with a PKD2-based homologous model indicate that the pore domain dilation is coupled to conformational changes of voltage-sensing domains (VSDs) via a series of π–π interactions, suggesting a potential PKD2L1 gating mechanism. 1 Ministry of Education Key Laboratory of Protein Science, Tsinghua University, Beijing 100084, China. 2 School of Life Sciences, Tsinghua University, Beijing 100084, China. 3 Beijing Advanced Innovation Center for Structural Biology, Tsinghua University, Beijing 100084, China. 4 School of Medicine, Tsinghua University, Beijing 100084, China. -

17Th ASIAN GAMES at Incheon/KOR, 27.09. – 03.10.2014
17th ASIAN GAMES at Incheon/KOR, 27.09. – 03.10.2014 -following are the complete results plus detailed additional information (medal & placing table, updated ASIAN GAMES Records etc.) from the Athletics competitions at the ASIAN GAMES- -with thanks to Mr Mirko Jalava and information from his excellent TILASTOPAJA-website – -athletes from 42 (out of the 45) ASIAN AA Member Federations participated (i.e. all except BAN, BRU & JOR) – the following statistical details are a proof for the outstanding standards in most events at these Games: -a comparison of the results for the winners to the Gold medalists from Guangzhou (2010 Games) shows better performances in 37 events (13 by Men & 14 by Women) against better performances at the 2010 Games in the other 19 events (10 by Men & 9 by Women); the remaining (47th) event, Men´s 50 km Walk, was not held 4 years ago; in addition another better time (one of the new GR´s) compared to the 2010 Games was achieved in a heat of Women´s 400 m hurdles – -see additional remarks concerning the outstanding standards at these Games on pages 28 & 29 - -the following abbreviations are used in the results section below (number of new records achieved in each category shown in bracketts); records broken twice (i.e. in a heat and again improved in a final) are not even counted for this survey: -AR = new Area Records (= 3); GR = new Games Records (= 18 – including 1 equalled GR) -NR = new National Records (= 44); these NR´s were achieved by athletes from 26 different federations with the most new records (each in -

Ion Channels 3 1
r r r Cell Signalling Biology Michael J. Berridge Module 3 Ion Channels 3 1 Module 3 Ion Channels Synopsis Ion channels have two main signalling functions: either they can generate second messengers or they can function as effectors by responding to such messengers. Their role in signal generation is mainly centred on the Ca2 + signalling pathway, which has a large number of Ca2+ entry channels and internal Ca2+ release channels, both of which contribute to the generation of Ca2 + signals. Ion channels are also important effectors in that they mediate the action of different intracellular signalling pathways. There are a large number of K+ channels and many of these function in different + aspects of cell signalling. The voltage-dependent K (KV) channels regulate membrane potential and + excitability. The inward rectifier K (Kir) channel family has a number of important groups of channels + + such as the G protein-gated inward rectifier K (GIRK) channels and the ATP-sensitive K (KATP) + + channels. The two-pore domain K (K2P) channels are responsible for the large background K current. Some of the actions of Ca2 + are carried out by Ca2+-sensitive K+ channels and Ca2+-sensitive Cl − channels. The latter are members of a large group of chloride channels and transporters with multiple functions. There is a large family of ATP-binding cassette (ABC) transporters some of which have a signalling role in that they extrude signalling components from the cell. One of the ABC transporters is the cystic − − fibrosis transmembrane conductance regulator (CFTR) that conducts anions (Cl and HCO3 )and contributes to the osmotic gradient for the parallel flow of water in various transporting epithelia. -
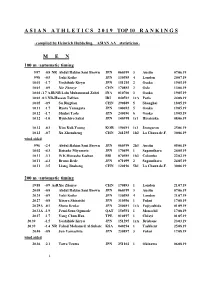
2019 TOP10-Rankings
A S I A N A T H L E T I C S 2 0 1 9 TOP 10 R A N K I N G S - compiled by Heinrich Hubbeling, ASIAN AA – statistician - M E N 100 m / automatic timing 9.97 +0.8 NR Abdul Hakim Sani Brown JPN 060399 3 Austin 07.06.19 9.98 +0.5 Yuki Koike JPN 130595 4 London 20.07.19 10.01 +1.7 Yoshihide Kiryu JPN 151295 2 Osaka 19.05.19 10.01 +0.9 Xie Zhenye CHN 170893 2 Oslo 13.06.19 10.03 +1.7 AJR/NR Lalu Muhammad Zohri INA 010700 3 Osaka 19.05.19 10.03 -0.3 NR=Hassan Taftian IRI 040593 1rA Paris 24.08.19 10.05 +0.9 Su Bingtian CHN 290889 5 Shanghai 18.05.19 10.11 +1.7 Ryota Yamagata JPN 100692 5 Osaka 19.05.19 10.12 +1.7 Shuhei Tada JPN 240696 6 Osaka 19.05.19 10.12 +1.0 Ryuichiro Sakai JPN 140398 1s1 Hiratsuka 08.06.19 10.12 -0.3 Kim Kuk-Young KOR 190491 1s1 Jeongseon 25.06.19 10.12 +0.7 Xu Zhouzheng CHN 261295 1h2 La Chaux-de-F. 30.06.19 wind-aided 9.96 +2.4 Abdul Hakim Sani Brown JPN 060399 2h3 Austin 05.06.19 10.02 +4.3 Daisuke Miyamoto JPN 170499 1 Sagamihara 24.05.19 10.11 +3.1 W.K.Himasha Eashan SRI 070595 1h3 Colombo 22.02.19 10.11 +4.3 Bruno Dede JPN 071099 2 Sagamihara 24.05.19 10.11 +3.5 Liang Jinsheng CHN 120196 5h1 La Chaux-de-F. -

Transcriptomic Profiling of Ca Transport Systems During
cells Article Transcriptomic Profiling of Ca2+ Transport Systems during the Formation of the Cerebral Cortex in Mice Alexandre Bouron Genetics and Chemogenomics Lab, Université Grenoble Alpes, CNRS, CEA, INSERM, Bâtiment C3, 17 rue des Martyrs, 38054 Grenoble, France; [email protected] Received: 29 June 2020; Accepted: 24 July 2020; Published: 29 July 2020 Abstract: Cytosolic calcium (Ca2+) transients control key neural processes, including neurogenesis, migration, the polarization and growth of neurons, and the establishment and maintenance of synaptic connections. They are thus involved in the development and formation of the neural system. In this study, a publicly available whole transcriptome sequencing (RNA-Seq) dataset was used to examine the expression of genes coding for putative plasma membrane and organellar Ca2+-transporting proteins (channels, pumps, exchangers, and transporters) during the formation of the cerebral cortex in mice. Four ages were considered: embryonic days 11 (E11), 13 (E13), and 17 (E17), and post-natal day 1 (PN1). This transcriptomic profiling was also combined with live-cell Ca2+ imaging recordings to assess the presence of functional Ca2+ transport systems in E13 neurons. The most important Ca2+ routes of the cortical wall at the onset of corticogenesis (E11–E13) were TACAN, GluK5, nAChR β2, Cav3.1, Orai3, transient receptor potential cation channel subfamily M member 7 (TRPM7) non-mitochondrial Na+/Ca2+ exchanger 2 (NCX2), and the connexins CX43/CX45/CX37. Hence, transient receptor potential cation channel mucolipin subfamily member 1 (TRPML1), transmembrane protein 165 (TMEM165), and Ca2+ “leak” channels are prominent intracellular Ca2+ pathways. The Ca2+ pumps sarco/endoplasmic reticulum Ca2+ ATPase 2 (SERCA2) and plasma membrane Ca2+ ATPase 1 (PMCA1) control the resting basal Ca2+ levels. -

Ion Channels
UC Davis UC Davis Previously Published Works Title THE CONCISE GUIDE TO PHARMACOLOGY 2019/20: Ion channels. Permalink https://escholarship.org/uc/item/1442g5hg Journal British journal of pharmacology, 176 Suppl 1(S1) ISSN 0007-1188 Authors Alexander, Stephen PH Mathie, Alistair Peters, John A et al. Publication Date 2019-12-01 DOI 10.1111/bph.14749 License https://creativecommons.org/licenses/by/4.0/ 4.0 Peer reviewed eScholarship.org Powered by the California Digital Library University of California S.P.H. Alexander et al. The Concise Guide to PHARMACOLOGY 2019/20: Ion channels. British Journal of Pharmacology (2019) 176, S142–S228 THE CONCISE GUIDE TO PHARMACOLOGY 2019/20: Ion channels Stephen PH Alexander1 , Alistair Mathie2 ,JohnAPeters3 , Emma L Veale2 , Jörg Striessnig4 , Eamonn Kelly5, Jane F Armstrong6 , Elena Faccenda6 ,SimonDHarding6 ,AdamJPawson6 , Joanna L Sharman6 , Christopher Southan6 , Jamie A Davies6 and CGTP Collaborators 1School of Life Sciences, University of Nottingham Medical School, Nottingham, NG7 2UH, UK 2Medway School of Pharmacy, The Universities of Greenwich and Kent at Medway, Anson Building, Central Avenue, Chatham Maritime, Chatham, Kent, ME4 4TB, UK 3Neuroscience Division, Medical Education Institute, Ninewells Hospital and Medical School, University of Dundee, Dundee, DD1 9SY, UK 4Pharmacology and Toxicology, Institute of Pharmacy, University of Innsbruck, A-6020 Innsbruck, Austria 5School of Physiology, Pharmacology and Neuroscience, University of Bristol, Bristol, BS8 1TD, UK 6Centre for Discovery Brain Science, University of Edinburgh, Edinburgh, EH8 9XD, UK Abstract The Concise Guide to PHARMACOLOGY 2019/20 is the fourth in this series of biennial publications. The Concise Guide provides concise overviews of the key properties of nearly 1800 human drug targets with an emphasis on selective pharmacology (where available), plus links to the open access knowledgebase source of drug targets and their ligands (www.guidetopharmacology.org), which provides more detailed views of target and ligand properties. -

Tradução Para Legendagem 7
UNIVERSIDADE DE LISBOA FACULDADE DE LETRAS Tradução audiovisual à luz da Teoria de Skopos: o caso da legendagem da série “As Imperatrizes no Palácio (Zhēn Huán zhuàn 甄嬛传)”. Jingrui Zhang Dissertação orientada pela Professora Doutora Elisabetta Colla Rosado Coelho David, especialmente elaborada para a obtenção do grau de Mestre em Tradução 2021 AGRADECIMENTOS Em primeiro lugar, à minha orientadora Elisabetta Colla Rosado Coelho David, pela simpatia com que me recebeu, pela sua orientação de trabalho e por todo o apoio prestado durante a frequência do meu mestrado. Aos meus pais, pelo vosso sacrifício e pela vossa educação desde que nasci. Aos meus primos e amigos, pela vossa amizade. Ao meu namorado, Ricardo Borges, pelo teu amor, carinho e companhia, por estares sempre presente, por me teres ajudado a ultrapassar os momentos mais difíceis e por todo o teu incentivo. Resumo Esta dissertação de mestrado propõe-se a estudar de que forma são traduzidos, nos textos audiovisuais, os elementos culturais que distinguem as línguas e culturas chinesa e portuguesa, tomando como objeto de estudo a legendagem da série chinesa “As Imperatrizes no Palácio (Zhēn Huán zhuàn 甄嬛传)” da Netflix. Esta série, cujo tema se desenvolve entre 1723 e 1735 durante o reinado da Dinastia Qing, relata a vida da personagem principal, Zhen Huan 甄嬛, no palácio imperial. Com a globalização, verificou-se um desenvolvimento abrangente e acelerado da comunicação política, económica e cultural entre a China e os restantes países. Ao longo dos últimos cem anos, as obras cinematográficas e televisivas revelaram-se formas de arte de alcance global com forte apelo popular, características que as estabeleceram como canais de comunicação de relevo e que possibilitaram a existência de intercâmbios culturais a um ritmo elevado, desencadeando assim um papel crucial na comunicação.