Olecular Complexes of Picric Acid Master of Science
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Alcohols Combined 1405
ALCOHOLS COMBINED 1405 Formulas: Table 1 MW: Table 1 CAS: Table 2 RTECS: Table 2 METHOD: 1405, Issue 1 EVALUATION: PARTIAL Issue 1: 15 March 2003 OSHA : Table 2 PROPERTIES: Table 1 NIOSH: Table 2 ACGIH: Table 2 COMPOUNDS: (1) n-butyl alcohol (4) n-propyl alcohol (7) cyclohexanol (2) sec-butyl alcohol (5) allyl alcohol (8) isoamyl alcohol (3) isobutyl alcohol (6) diacetone alcohol (9) methyl isobutyl carbinol SYNONYMS: See Table 3. SAMPLING MEASUREMENT SAMPLER: SOLID SORBENT TUBE TECHNIQUE: GAS CHROMATOGRAPHY, FID (Coconut shell charcoal, 100 mg/50 mg) ANALYTE: Compounds above FLOW RATE: 0.01 to 0.2 L/min DESORPTION: 1 mL 5% 2-propanol in CS2 Compounds: (1-3 ) (4-9) VOL-MIN: 2 L 1 L INJECTION -MAX: 10 L 10 L VOLUME: 1 µL SHIPMENT: Routine TEMPERATURE -INJECTION: 220 °C SAMPLE -DETECTOR: 250 - 300 °C STABILITY: See Evaluation of Method. -COLUMN: 35 °C (7 minutes), to 60 °C at 5 °C/minute, hold 5 minutes, up to BLANKS: 2 to 10 field blanks per set 120 °C at 10 °C /minute, hold 3 minutes. CARRIER GAS: He, 4 mL/min ACCURACY COLUMN: Capillary, fused silica, 30 m x 0.32-mm RANGE STUDIED: Not studied [1, 2]. ID; 0.5 µm film polyethylene glycol, DB- wax or equivalent BIAS: Not determined CALIBRATION: Solutions of analyte in eluent (internal OVERALL standard optional) PRECISION (Ö ): Not determined rT RANGE: See EVALUATION OF METHOD. ACCURACY: Not determined ESTIMATED LOD: 1 µg each analyte per sample PRECISION: See EVALUATION OF METHOD. APPLICABILITY: This method may be used to determine two or more of the specified analytes simultaneously. -

Acid-Base Behavior in Aprotic Organic Solvents
UNITED STATES DEPARTMENT OF COMMERCE • C. R. Smith, Secretary NATIONAL BUREAU OF STANDARDS • A. V. Astin, Director Acid-Base Behavior in Aprotic Organic Solvents Marion Maclean Davis Institute for Materials Research National Bureau of Standards Washington, D.C. 20234 , 105 . U^S^ National Bureau of Standards.Monograph r » » Issued August 1968 For sale by the Superinlendent <if Documents. U.S. Government Printing Office Washington, D.C. 20402 - Price $2.25 NOV 2 9 1368 1^4 2 5 '46 Library of Congress Catalog Card No. 67-62078 Foreword During the past 50 years the American chemical industry has made available for common uses a great variety of organic solvents, in some of which acids and bases behave very differently than in water. For example, the order of relative strengths of a series of acids or bases may be altered by a change of solvent. This is especially evident when acid-base behavior in water is compared with that in hydrocarbons and halogenated hydrocarbons. To the latter two groups, which are often called "inert" or "aprotic" solvents, belong important liquids like benzene, toluene, cyclo- hexane, and carbon tetrachloride. Industrially important materials such as drycleaning solvents, lubricants, motor fuels, refrigerants, and transformer oils are additional examples. From 1941 to the end of 1965 the National Bureau of Standards maintained, in response to requests from industry and other Government agencies, a research program designed to ascertain and explain acid-base behavior in aprotic organic solvents, as well as to develop methods and reference materials for determining total acid and base content and relative strengths of acids and bases in such media. -

Synthesis and Properties of Lead Picrates
Science and Technology of Energetic Materials, Vol.65, No.1, 2004 7 Article Synthesis and properties of lead picrates Makoto Matsukawa*, Takehiro Matsunaga**, Masatake Yoshida**, and Shuzo Fujiwara** *Showa Kinzoku Kogyo Co., Ltd. Iwase 2120, Iwase, Nishi-Ibaraki Gun, Ibaraki 309-1211, JAPAN e-mail: [email protected] **National Institute of Advanced Industrial Science and Technology, 1-1 Higashi, Tsukuba, Ibaraki 305-8565, JAPAN e-mail: [email protected] Received: November 10, 2003 Accepted: January 23, 2004 Abstract Picric acid is known to react with metals to form highly unstable metallic picrates, which have been implicated in a num- ber of serious explosive accidents. In this study, lead picrates were synthesized by several methods, and the thermody- namic and explosive properties such as sensitivity were examined. Differential scanning calorimetry (DSC) results revealed that lead picrates had several heat decomposition patterns depending on the synthesis method, and they had lower temperature of start of exothermic reaction than sodium picrate had. The heat of decomposition of lead picrates was found to be lower than that of picric acid. Lead picrate from lead acetate and picric acid had lower activation energy of thermal decomposition than the other lead picrates. Thermogravimetry analysis and Karl Fischer analysis confirmed that lead picrates contained crystalline H2O, which dehydrated at above 375 K. And, lead picrates did not have property of fusion. Drop hammer test results showed that lead picrates had high strike sensitivity, whereas the lead picrates had low friction sensitivity in friction tests, attributable to the presence of crystalline H2O. The ignition temperatures of lead picrates were found to be in the range 543.9–600.3 K. -
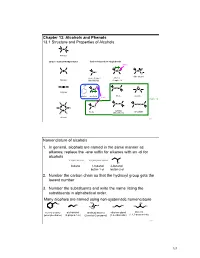
Chapter 13.Pptx
Chapter 13: Alcohols and Phenols 13.1 Structure and Properties of Alcohols C C Alkanes Carbon - Carbon Multiple Bonds Carbon-heteroatom single bonds basic O C C C N C N C X O nitro alkane X= F, Cl, Br, I amines Alkenes Alkyl Halide Chapter 23 OH C C H O C O C C O C C Alkynes phenol alcohols ethers epoxide acidic Chapter 14 H H H C S C C C C S S C C S C C H C C sulfides thiols disulfide H H (thioethers) Arenes 253 Nomenclature of alcohols 1. In general, alcohols are named in the same manner as alkanes; replace the -ane suffix for alkanes with an -ol for alcohols CH3CH2CH2CH3 CH3CH2CH2CH2OH OH butane 1-butanol 2-butanol butan-1-ol butan-2-ol 2. Number the carbon chain so that the hydroxyl group gets the lowest number 3. Number the substituents and write the name listing the substituents in alphabetical order. Many alcohols are named using non-systematic nomenclature H C OH 3 OH OH C OH OH HO OH H3C HO H3C benzyl alcohol allyl alcohol tert-butyl alcohol ethylene glycol glycerol (phenylmethanol) (2-propen-1-ol) (2-methyl-2-propanol) (1,2-ethanediol) (1,2,3-propanetriol) 254 127 Alcohols are classified according to the H R C OH C OH H H degree of substitution of the carbon bearing H H 1° carbon the -OH group methanol primary alcohol primary (1°) : one alkyl substituent R R C OH C OH R R secondary (2°) : two alkyl substituents H R 2° carbon 3° carbon tertiary (3°) : three alkyl substituents secondary alcohol tertiary alcohol Physical properties of alcohols – the C-OH bond of alcohols has a significant dipole moment. -

A New Approach to Prepare Polyethylene Glycol Allyl Glycidyl Ether
E3S Web of Conferences 267, 02004 (2021) https://doi.org/10.1051/e3sconf/202126702004 ICESCE 2021 A new approach to prepare Polyethylene Glycol Allyl Glycidyl Ether Huizhen Wang1*, Ruiyang Xie1, Mingjun Chen1*, Weihao Deng1, Kaixin Zhang2, Jiaqin Liu1 1School of Science, Xihua University, Chengdu 610039, China; 2Chengdu Jingyiqiang Environmental Protection Technology Co., Ltd. Abstract. The polyethylene glycol allyl glycidyl ether (PGAGE) is an important intermediate for preparing silicone softener that can be synthesized from allyl alcohol polyoxyethylene ether and epichlorohydrin (ECH). The performance parameters including the concentration of ECH, initial boron trifluoride diethyl etherate (BFEE) as well as CaCl2 quality were investigated respectively. The optimum process parameters which can get high capping and low by-product rate are as follows: the ECH concentration is 2.0 M, the initial BFEE concentration is 1.65mM, and the CaCl2 dosage is 1.65g/L. Under these conditions, the maximal yield can be improved to 91.36%, the percent of capping rate is higher than 98.16%, the residual concentration of F- is only 0.63 mg/L. concentrated basic solution, in which the total yield was between 90%~91% by Matsuoka et al. [10] also use the 1 Introduction two-step reaction to synthesize AGE based on the reaction Polyethylene glycol allyl glycidyl ether (PGAGE) and the of allyl alcohol with ECH using BFEE as the catalyst. allyl polyoxyethylene ether (APEG), tethering with both Their results demonstrated that the yield reaches 82% alkene and epoxy groups, are widely used as fabric under the following condition: n (ECH) : n (allyl alcohol): finishing agent [1-2] , reactive diluent [3] , cross-linking (catalysis) = 1: (1~3) : (0.01~0.002). -

Picric Acid Picric Acid (CAS No
Picric Acid Picric acid (CAS No. 88-89-1,2,4,6-Trinitrophenol, picronitric acid) is a pale yellow, odorless crystal that is slightly soluble in water. It is primarily used as a staining reagent and in synthesis reactions. When hydrated, it is typically harmless but when dry can be a powerful explosive. Picric acid is highly sensitive to heat, shock and friction and, additionally, is a toxic substance by all modes of entry (i.e., inhalation, ingestion, dermal contact). Picric acid is highly reactive with a wide variety of materials (e.g., concrete, plaster, amines, bases, and metals such as lead, zinc, copper, and mercury) to form picrate salts, which are more reactive and shock sensitive than the acid itself. Purchasing • Purchase of picric acid should be restricted to the smallest practicable quantity. • If possible, eliminate it from your inventory by purchasing premixed stains or a 1% solution for using in stain preparation. Storage • Label containers with date received and date opened. • Store in original container in a cool, dry, wellventilated area away from sources of heat. • Keep wet - material should be a wet paste and greater than 10% water by volume. • Check for evidence of dried crystals (see handling section) and rehydrate contents every 6 months with DI water as needed and document on bottle. • Dispose after 2 years of storage. • Store separately from oxidizers, reducing agents, inorganic salts, metals (copper, lead, zinc, aluminum + water), ammonia, concrete, plaster, salts, gelatin, alkaloids and albumin. Handling • Do not use metal spatulas to remove picric acid. • Clean the bottleneck, cap and threads with a wet cloth before resealing. -

Trinitrobenzene
doi: 10.5028/jatm.2011.03010411 Gilson da Silva* National Industrial Property Institute Synthesis of 2,4,6-triamino-1,3,5- Rio de Janeiro – Brazil [email protected] trinitrobenzene Elizabeth da Costa Mattos Abstract: The 2,4,6-triamino-1,3,5-trinitrobenzene (TATB) is perhaps the Institute of Aeronautics and Space most thermostable and insensitive explosive known. Its low sensibility to São José dos Campos – Brazil shock makes it suitable for military and civil applications. TATB application [email protected] is done either alone or in combination with another high energetic material. This study aimed at reporting the review about many processes to produce *author for correspondence TATB and the problems associated with them, as well as suggest techniques like Fourier Transform Infrared Spectroscopy (FT-IR) and Differential Scanning Calorimetry (DSC), which can be useful in the characterization of this energetic compound. Keywords: TATB, Fourier Transform Infrared Spectroscopy, Differential Scanning Calorimetry, Plastic-bonded explosive. LIST OF SYMBOLS impact hazards is important. Other potential applications include the use of TATB as the booster or main charge TATB 2,4,6-triamino-1,3,5-trinitrobenzene explosives for down-hole oil perforation at elevated HE high explosive temperature surroundings (Lee, 1996). PBX plastic-bonded explosive HMX octogen TATB is a high explosive (HE) that can be combined with plastic binder to produce a plastic-bonded explosive RDX hexogen (PBX) composition, which is heat-resistant and highly TCB 1,3,5-trichlorobenzene insensitive. It is insoluble in organic solvents and has a TCTNB 1,3,5-trichloro-2,4,6-trinitrobenzene melting point above 400oC. -

Title Selective Reduction of Acrolein to Allyl Alcohol by a Vapor Phase
Selective Reduction of Acrolein to Allyl Alcohol by a Vapor Title Phase Hydrogen Transfer Reaction Author(s) Kunichika, Sango; Oka, Shinzaburo; Sakai, Mutsuji Bulletin of the Institute for Chemical Research, Kyoto Citation University (1966), 44(3): 215-220 Issue Date 1966-10-31 URL http://hdl.handle.net/2433/76125 Right Type Departmental Bulletin Paper Textversion publisher Kyoto University Selective Reduction of Acrolein to Allyl Alcohol by a Vapor Phase Hydrogen Transfer Reaction Sango KUNICHIKA,Shinzaburo OKA and Mutsuji SAKAI* (KunichikaLaboratory) (ReceivedJune 15, 1966) Selectivereduction of acroleinto ally! alcoholhas been achievedby a vapor phase hydrogen transferreaction using isopropyl alcohol as a hydrogendonor. It was foundthat the preferredcatalyst was 50 weight% zinc oxide-magnesiumoxide catalyst prepared by calcininga mixtureof zincnitrate and magnesiumoxide at 500°C. The optimumconditions were found to be as follows;reaction tem- perature,near 200°C,total feed rate, 45 moles/1.of catalyst/hr.,feed ratio of isopropylalcohol per acrolein,10 moles. Under theseconditions, the yieldsof allyl alcoholof 80-65% at 80-90% conversion of acroleinwere obtained. INTRODUCTION Acrolein has two reactive functional groups, one is carbonyl group and the other is carbon-carbon double bond. Selective reduction of acrolein to allyl alcohol has been studied by several investigators. The reaction was generally carried out by the following two methods: (1) the Meerwein-Ponndorf-Verley reduction1-3), (2) vapor phase hydrogen transfer reaction with various metal oxide catalysts using an alcohol as a hydrogen donor4-11> The liquid phase reaction has some technical difficulties owing to the polymeri- zation of acrolein especially at high conversion. Therefore, in the present work, the more promising vapor phase reaction has been studied to find an effective catalyst and favorable reaction conditions. -

Conversion of Glycerol Into Allyl Alcohol Over Potassium-Supported Zirconia-Iron Oxide Catalyst
Title Conversion of glycerol into allyl alcohol over potassium-supported zirconia-iron oxide catalyst Author(s) Konaka, Aya; Tago, Teruoki; Yoshikawa, Takuya; Nakamura, Ayaka; Masuda, Takao Applied catalysis b-environmental, 146, 267-273 Citation https://doi.org/10.1016/j.apcatb.2013.03.007 Issue Date 2014-03 Doc URL http://hdl.handle.net/2115/54715 Type article (author version) File Information Manuscript_Glycerol to Allyl Alcohol .pdf Instructions for use Hokkaido University Collection of Scholarly and Academic Papers : HUSCAP Conversion of Glycerol into Allyl Alcohol over Potassium-Supported Zirconia-Iron Oxide Catalyst Aya Konaka, Teruoki Tago*, Takuya Yoshikawa, Ayaka Nakamura, and Takao Masuda Research Group of Chemical Engineering, Division of Chemical Process Engineering, Faculty of Engineering, Hokkaido University, N13W8, Kita-ku, Sapporo, Hokkaido, Japan 060-8628 * Corresponding author should be addressed E-mail: [email protected] Tel: +81-11-706-6551 Fax: +81-11-706-6552 1 Abstract The catalytic conversion of glycerol was performed with iron oxide-based catalysts for production of allyl alcohol using a fixed-bed flow reactor at 623 K under atmospheric pressure. The glycerol dehydration proceeds on acid sites of catalysts while the allyl alcohol production is assumed to be catalyzed by non-acidic sites of catalysts through a hydrogen transfer mechanism. Different alkali metals, including Na, K, Rb, and Cs were supported on ZrO2-FeOX and all of them gave impressively higher allyl alcohol yield and suppressed glycerol dehydration due to the reduced catalyst acidic property. K-supported ZrO2-FeOX (K/ZrO2-FeOX) was chosen for further studies, and allyl alcohol yield remarkably increased up to 27 mol%-C at the K content of 3-5 mol%. -

Nited States Patent Office. Arthur George Green, of Leeds, England
NITED STATES PATENT OFFICE. ARTHUR GEORGE GREEN, OF LEEDS, ENGLAND. ANUFACURE OF PICRIC ACD. 1,299,171. specification of Letters Patent, Patented Apr. 1, 1919, NoDrawing. Application fled January 29, 1916. Serial No. 75,023, To all whom it may concern: generated in the mixture from sodium nitrate. Beit known that I, ARTHUR GEORGE GREEN, The dinitrophenol itself is most conveniently B.Sc., F. R. S., F. I. C., subject of the King produced by chlorinating benzene to mono of Great Britain and Ireland, residing in the chlorobenzene, nitrating the latter with a University of Leeds, Leeds, in the county of mixture of nitric acid and surfuric acid to 60 York, England, professor of applied chem: dinitrochlorobenzene, and conversion of this istry, have invented certain new and useful into dinitrophenol by boiling with caustic Improvements in the Manufacture of Picric alkali. The entire chain of reaetions, start Acid, of which the following is a specifica ing from benzene, is therefore the follow 0. Ing:- . tion.The ordinary--- method of manufacturingO 35 picric acid from phenol is subject to several CHCHCCNO)Cl. disadvantages. In the first place it is de of Nö),0fc.H.(NO),OH pendent upon the available supplies of phe As the conditions for carrying out the first nol, which if much in demand is liable to rise three steps are well known, it is only neces to a high price. In the second place the ni tration of phenol, as usually carried out, effectingsy to describe the last instep detail in the the conversion best way i. -

United States Patent (19) (11) Patent Number: 4,968,451 Scheibel Et Al
United States Patent (19) (11) Patent Number: 4,968,451 Scheibel et al. 45) Date of Patent: Nov. 6, 1990 (54) SOIL RELEASE AGENTS HAVING ALLYL-DERVED SULFONATED END CAPS FOREIGN PATENT DOCUMENTS 155710 9/1985 European Pat. Off. (75) Inventors: Jeffrey J. Scheibel; Eugene P. 180356 5/1986 European Pat. Off. Gosselink, both of Cincinnati, Ohio 1475798 6/1977 United Kingdom . (73) Assignee: The Procter & Gamble Company, OTHER PUBLICATIONS Cincinnati, Ohio S. C. Bright, C. E. Stubbs and L. Thompson, J. Appl. (21) Appl. No.: 474,709 Chem. Biotechnol, 1975, vol. 25, pp. 901-912. 22) Filed: Jan. 29, 1990 Norton et al., J. Org. Chem, vol. 33, No. 11, pp. 4158-4165 (1967). Related U.S. Application Data Primary Examiner-A. Lionel Clingman (63) Continuation of Ser. No. 237,598, Aug. 26, 1988, aban Attorney, Agent, or Firm-Leonard W. Lewis; Jerry J. doned. Yetter (51 Int, C. .................... CO7C 309/05; C08G 63/68; 57 ABSTRACT C11D 3/37 (52) U.S. C. ............................... 252/549; 252/174.19; The present invention relates to novel soil release 252/539; 252/558; 252/DIG. 15 agents, which are particular sulfonated linear tere (58) Field of Search ......................................... 252/549 phthalate ester oligomers (S.T.E. oligomers). The S.T.E. oligomers are especially suitable for formulation (56 References Cited into laundry products such as laundry detergents or U.S. PATENT DOCUMENTS fabric conditioners. Thus formulated, they provide ef. 3,821,169 6/1974 Duddey et al. ..................... 528/293 fective soil release treatments for fabrics laundered in 4,156,073 5/1979 Login ............. -

Complexation
FACULTY OF PHARMACEUTICAL SCIENCES, RAMAUNIVERSITY, KANPUR B.PHARM 3rd SEM PHYSICAL PHARMACEUTICS-I BP302T MR. PEEYUSH Assistant professor Rama university, kanpur Complexation Overview Classification Introduction Metal ion complexes Organic Complexes Inclusion Complexes Methods of Analysis Method of Continuous Variation PH Titration Distribution Method Solubility Method Spectroscopy Learning Objectives 1. Define the three classes of complexes with pharmaceutically relevant examples. 2. Describe chelates, their physically properties, and what differentiates them from organic molecular complexes. 3. Describe the types of forces that hold together organic molecular complexes with examples. 4. Describe the forces in polymer–drug complexes used for drug delivery. 5. Discuss the pharmaceutical applications of cyclodextrins. 6. Describe the methods of analysis of complexes and determine their stoichiometric ratios and stability constants. Classification Introduction Metal ion complexes Organic Complexes Inclusion Complexes INTRODUCTION Complexes are compounds that result from donor–acceptor mechanisms between two or more chemical species. Complexes can be divided broadly into three classes depending the type of the acceptor substance: 1. Metal ion complexes 2. Organic molecular complexes 3. Inclusion complexes Intermolecular forces involved in the formation of complexes: 1. Van der Waals forces. 2. Hydrogen bonds (important in molecular complexes). 3. Coordinate covalence (important in metal complexes). 4. Charge transfer. 5. Hydrophobic interaction. Introduction Types of Complexes Metal Ion Complexes A. Inorganic type B. Chelates C. Olefin type D. Aromatic type II. Organic Molecular Complexes A. Quinhydrone type B. Picric acid type C. Caffeine and other drug complexes D. Polymer type III. Inclusion Compounds A. Channel lattice type B. Layer type C. Clathrates D. Monomolecular type E.