Unmasking Genes in a Type 1 Diabetes–Resistant Mouse Strain That Enhances Pathogenic CD8 T-Cell Responses John P
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

(Dominance) Effects of Genetic Variants Associated with Refractive Error And
Molecular Genetics and Genomics https://doi.org/10.1007/s00438-020-01666-w ORIGINAL ARTICLE Non‑additive (dominance) efects of genetic variants associated with refractive error and myopia Alfred Pozarickij1 · Cathy Williams2 · Jeremy A. Guggenheim1 · and the UK Biobank Eye and Vision Consortium Received: 26 November 2019 / Accepted: 16 March 2020 © The Author(s) 2020 Abstract Genome-wide association studies (GWAS) have revealed that the genetic contribution to certain complex diseases is well- described by Fisher’s infnitesimal model in which a vast number of polymorphisms each confer a small efect. Under Fisher’s model, variants have additive efects both across loci and within loci. However, the latter assumption is at odds with the com- mon observation of dominant or recessive rare alleles responsible for monogenic disorders. Here, we searched for evidence of non-additive (dominant or recessive) efects for GWAS variants known to confer susceptibility to the highly heritable quantitative trait, refractive error. Of 146 GWAS variants examined in a discovery sample of 228,423 individuals whose refractive error phenotype was inferred from their age-of-onset of spectacle wear, only 8 had even nominal evidence (p < 0.05) of non-additive efects. In a replication sample of 73,577 individuals who underwent direct assessment of refractive error, 1 of these 8 variants had robust independent evidence of non-additive efects (rs7829127 within ZMAT4, p = 4.76E−05) while a further 2 had suggestive evidence (rs35337422 in RD3L, p = 7.21E−03 and rs12193446 in LAMA2, p = 2.57E−02). Account- ing for non-additive efects had minimal impact on the accuracy of a polygenic risk score for refractive error (R2 = 6.04% vs. -

Structural Insights Into Ubiquitin Recognition and Ufd1 Interaction of Npl4
ARTICLE https://doi.org/10.1038/s41467-019-13697-y OPEN Structural insights into ubiquitin recognition and Ufd1 interaction of Npl4 Yusuke Sato1,2,3,5,7, Hikaru Tsuchiya4,7, Atsushi Yamagata1,2,3,6, Kei Okatsu1,2, Keiji Tanaka4, Yasushi Saeki 4* & Shuya Fukai 1,2,3* Npl4 is likely to be the most upstream factor recognizing Lys48-linked polyubiquitylated substrates in the proteasomal degradation pathway in yeast. Along with Ufd1, Npl4 forms a 1234567890():,; heterodimer (UN), and functions as a cofactor for the Cdc48 ATPase. Here, we report the crystal structures of yeast Npl4 in complex with Lys48-linked diubiquitin and with the Npl4- binding motif of Ufd1. The distal and proximal ubiquitin moieties of Lys48-linked diubiquitin primarily interact with the C-terminal helix and N-terminal loop of the Npl4 C-terminal domain (CTD), respectively. Mutational analysis suggests that the CTD contributes to linkage selectivity and initial binding of ubiquitin chains. Ufd1 occupies a hydrophobic groove of the Mpr1/Pad1 N-terminal (MPN) domain of Npl4, which corresponds to the catalytic groove of the MPN domain of JAB1/MPN/Mov34 metalloenzyme (JAMM)-family deubiquitylating enzyme. This study provides important structural insights into the polyubiquitin chain recognition by the Cdc48–UN complex and its assembly. 1 Institute for Quantitative Biosciences, The University of Tokyo, Tokyo 113-0032, Japan. 2 Synchrotron Radiation Research Organization, The University of Tokyo, Tokyo 113-0032, Japan. 3 Department of Computational Biology and Medical Sciences, Graduate School of Frontier Sciences, The University of Tokyo, Chiba 277-8562, Japan. 4 Laboratory of Protein Metabolism, Tokyo Metropolitan Institute of Medical Science, Tokyo 156-8506, Japan. -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -

Functional Receptor Molecules Cd300lf and Cd300ld Within the CD300 Family Enable Murine Noroviruses to Infect Cells
Functional receptor molecules CD300lf and CD300ld within the CD300 family enable murine noroviruses to infect cells Kei Hagaa,1, Akira Fujimotoa,1, Reiko Takai-Todakaa, Motohiro Mikia,b, Yen Hai Doana, Kosuke Murakamia, Masaru Yokoyamac, Kazuyoshi Muratad, Akira Nakanishie,2, and Kazuhiko Katayamaa,2 aDepartment of Virology II, National Institute of Infectious Diseases, Tokyo 208-0011, Japan; bNew Business Planning Division, Innovative Diagnostics Research Center, Denka Innovation Center, Denka Company Limited, Tokyo 194-8560, Japan; cPathogen Genomics Center, National Institute of Infectious Diseases, Tokyo 208-0011, Japan; dNational Institute for Physiological Sciences, Aichi 444-8585, Japan; and eSection of Gene Therapy, Department of Aging Intervention, National Center for Geriatrics and Gerontology, Aichi 474-8511, Japan Edited by Mary K. Estes, Baylor College of Medicine, Houston, Texas, and approved August 11, 2016 (received for review April 9, 2016) Norovirus is the leading cause of acute gastroenteritis worldwide. mice that were deficient in STAT1 and RAG2 (11). MNV repli- Since the discovery of human norovirus (HuNoV), an efficient and cates within the murine macrophage cell line RAW264.7 cells. reproducible norovirus replication system has not been established in MNV strains MNV-1, WU11, and S99 bind macrophages ex vivo cultured cells. Although limited amounts of virus particles can be through a terminal sialic acid on the ganglioside GD1a, which produced when the HuNoV genome is directly transfected into cells, has been suggested as a binding receptor (12). Another MNV the HuNoV cycle of infection has not been successfully reproduced in strain, CR3, binds a different glycan, and the difference in glycan any currently available cell-culture system. -

Functional Receptor Molecules Cd300lf and Cd300ld Within the CD300 Family Enable Murine Noroviruses to Infect Cells
Functional receptor molecules CD300lf and CD300ld within the CD300 family enable murine noroviruses to infect cells Kei Hagaa,1, Akira Fujimotoa,1, Reiko Takai-Todakaa, Motohiro Mikia,b, Yen Hai Doana, Kosuke Murakamia, Masaru Yokoyamac, Kazuyoshi Muratad, Akira Nakanishie,2, and Kazuhiko Katayamaa,2 aDepartment of Virology II, National Institute of Infectious Diseases, Tokyo 208-0011, Japan; bNew Business Planning Division, Innovative Diagnostics Research Center, Denka Innovation Center, Denka Company Limited, Tokyo 194-8560, Japan; cPathogen Genomics Center, National Institute of Infectious Diseases, Tokyo 208-0011, Japan; dNational Institute for Physiological Sciences, Aichi 444-8585, Japan; and eSection of Gene Therapy, Department of Aging Intervention, National Center for Geriatrics and Gerontology, Aichi 474-8511, Japan Edited by Mary K. Estes, Baylor College of Medicine, Houston, Texas, and approved August 11, 2016 (received for review April 9, 2016) Norovirus is the leading cause of acute gastroenteritis worldwide. mice that were deficient in STAT1 and RAG2 (11). MNV repli- Since the discovery of human norovirus (HuNoV), an efficient and cates within the murine macrophage cell line RAW264.7 cells. reproducible norovirus replication system has not been established in MNV strains MNV-1, WU11, and S99 bind macrophages ex vivo cultured cells. Although limited amounts of virus particles can be through a terminal sialic acid on the ganglioside GD1a, which produced when the HuNoV genome is directly transfected into cells, has been suggested as a binding receptor (12). Another MNV the HuNoV cycle of infection has not been successfully reproduced in strain, CR3, binds a different glycan, and the difference in glycan any currently available cell-culture system. -

Supplementary Materials
Supplementary materials Supplementary Table S1: MGNC compound library Ingredien Molecule Caco- Mol ID MW AlogP OB (%) BBB DL FASA- HL t Name Name 2 shengdi MOL012254 campesterol 400.8 7.63 37.58 1.34 0.98 0.7 0.21 20.2 shengdi MOL000519 coniferin 314.4 3.16 31.11 0.42 -0.2 0.3 0.27 74.6 beta- shengdi MOL000359 414.8 8.08 36.91 1.32 0.99 0.8 0.23 20.2 sitosterol pachymic shengdi MOL000289 528.9 6.54 33.63 0.1 -0.6 0.8 0 9.27 acid Poricoic acid shengdi MOL000291 484.7 5.64 30.52 -0.08 -0.9 0.8 0 8.67 B Chrysanthem shengdi MOL004492 585 8.24 38.72 0.51 -1 0.6 0.3 17.5 axanthin 20- shengdi MOL011455 Hexadecano 418.6 1.91 32.7 -0.24 -0.4 0.7 0.29 104 ylingenol huanglian MOL001454 berberine 336.4 3.45 36.86 1.24 0.57 0.8 0.19 6.57 huanglian MOL013352 Obacunone 454.6 2.68 43.29 0.01 -0.4 0.8 0.31 -13 huanglian MOL002894 berberrubine 322.4 3.2 35.74 1.07 0.17 0.7 0.24 6.46 huanglian MOL002897 epiberberine 336.4 3.45 43.09 1.17 0.4 0.8 0.19 6.1 huanglian MOL002903 (R)-Canadine 339.4 3.4 55.37 1.04 0.57 0.8 0.2 6.41 huanglian MOL002904 Berlambine 351.4 2.49 36.68 0.97 0.17 0.8 0.28 7.33 Corchorosid huanglian MOL002907 404.6 1.34 105 -0.91 -1.3 0.8 0.29 6.68 e A_qt Magnogrand huanglian MOL000622 266.4 1.18 63.71 0.02 -0.2 0.2 0.3 3.17 iolide huanglian MOL000762 Palmidin A 510.5 4.52 35.36 -0.38 -1.5 0.7 0.39 33.2 huanglian MOL000785 palmatine 352.4 3.65 64.6 1.33 0.37 0.7 0.13 2.25 huanglian MOL000098 quercetin 302.3 1.5 46.43 0.05 -0.8 0.3 0.38 14.4 huanglian MOL001458 coptisine 320.3 3.25 30.67 1.21 0.32 0.9 0.26 9.33 huanglian MOL002668 Worenine -
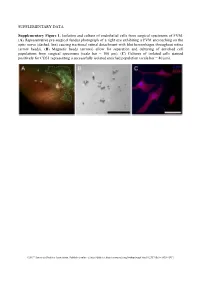
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Brain Region-Dependent Gene Networks Associated with Selective Breeding for Increased Voluntary Wheel-Running Behavior
UC Riverside UC Riverside Previously Published Works Title Brain region-dependent gene networks associated with selective breeding for increased voluntary wheel-running behavior. Permalink https://escholarship.org/uc/item/8c49g8fd Journal PloS one, 13(8) ISSN 1932-6203 Authors Zhang, Pan Rhodes, Justin S Garland, Theodore et al. Publication Date 2018 DOI 10.1371/journal.pone.0201773 Peer reviewed eScholarship.org Powered by the California Digital Library University of California RESEARCH ARTICLE Brain region-dependent gene networks associated with selective breeding for increased voluntary wheel-running behavior Pan Zhang1,2, Justin S. Rhodes3,4, Theodore Garland, Jr.5, Sam D. Perez3, Bruce R. Southey2, Sandra L. Rodriguez-Zas2,6,7* 1 Illinois Informatics Institute, University of Illinois at Urbana-Champaign, Urbana, IL, United States of America, 2 Department of Animal Sciences, University of Illinois at Urbana-Champaign, Urbana, IL, United a1111111111 States of America, 3 Beckman Institute for Advanced Science and Technology, Urbana, IL, United States of a1111111111 America, 4 Center for Nutrition, Learning and Memory, University of Illinois at Urbana-Champaign, Urbana, a1111111111 IL, United States of America, 5 Department of Evolution, Ecology, and Organismal Biology, University of a1111111111 California, Riverside, CA, United States of America, 6 Department of Statistics, University of Illinois at Urbana-Champaign, Urbana, IL, United States of America, 7 Carle Woese Institute for Genomic Biology, a1111111111 University of Illinois at Urbana-Champaign, Urbana, IL, United States of America * [email protected] OPEN ACCESS Abstract Citation: Zhang P, Rhodes JS, Garland T, Jr., Perez SD, Southey BR, Rodriguez-Zas SL (2018) Brain Mouse lines selectively bred for high voluntary wheel-running behavior are helpful models region-dependent gene networks associated with for uncovering gene networks associated with increased motivation for physical activity and selective breeding for increased voluntary wheel- other reward-dependent behaviors. -

Allele-Specific Alternative Splicing and Its Functional Genetic Variants in Human Tissues
Downloaded from genome.cshlp.org on September 30, 2021 - Published by Cold Spring Harbor Laboratory Press Allele-specific alternative splicing and its functional genetic variants in human tissues Kofi Amoah1,†, Yun-Hua Esther Hsiao2,†, Jae Hoon Bahn3†, Yiwei Sun3, Christina Burghard1, Boon Xin Tan3, Ei-Wen Yang3, Xinshu Xiao1,2,3,4,5,* 1Bioinformatics Interdepartmental Program, 2Department of Bioengineering, 3Department of Integrative Biology and Physiology, 4Molecular Biology Institute, 5Institute for Quantitative and Computational Biosciences, University of California, Los Angeles, CA 90095, USA. † These authors contributed equally. *Correspondence: [email protected] 1 Downloaded from genome.cshlp.org on September 30, 2021 - Published by Cold Spring Harbor Laboratory Press Abstract Alternative splicing is an RNA processing mechanism that affects most genes in human, contributing to disease mechanisms and phenotypic diversity. The regulation of splicing involves an intricate network of cis-regulatory elements and trans-acting factors. Due to their high sequence specificity, cis-regulation of splicing can be altered by genetic variants, significantly affecting splicing outcomes. Recently, multiple methods have been applied to understanding the regulatory effects of genetic variants on splicing. However, it is still challenging to go beyond apparent association to pinpoint functional variants. To fill in this gap, we utilized large-scale datasets of the Genotype-Tissue Expression (GTEx) project to study genetically-modulated alternative splicing (GMAS) via identification of allele-specific splicing events. We demonstrate that GMAS events are shared across tissues and individuals more often than expected by chance, consistent with their genetically driven nature. Moreover, although the allelic bias of GMAS exons varies across samples, the degree of variation is similar across tissues vs. -

Identi Cation of Potential Biomarkers and Pathways
Identication of Potential Biomarkers and Pathways in Neonatal Hypoxic-Ischemic Brain Injury: Based on Bioinformatics Technology Shangbin Li The First Hospital of Hebei Medical University Shuangshuang Li Chengdu University of Traditional Chinese Medicine Qian Zhao The First Hospital of Hebei Medical University Jiayu Huang The First Hospital of Hebei Medical University Jinfeng Meng The First Hospital of Hebei Medical University Weichen Yan The First Hospital of Hebei Medical University Jie Wang The First Hospital of Hebei Medical University Changjun Ren ( [email protected] ) The First Hospital of Hebei Medical University Ling Hao The First Hospital of Hebei Medical University Research Article Keywords: neonatal hypoxic-ischemic brain injury, inammatory response, immune response, differentially expressed genes, bioinformatics analysis Posted Date: August 5th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-777916/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/22 Abstract Background Neonatal hypoxic-ischemic brain damage (HIBD) is one of the most common serious diseases in newborns, with a high mortality and disability rate. This study aims to use the bioinformatics analysis to identify potential hematologic/immune systems tissue-specic genes and related signaling pathways neonatal HIBD. Methods Microarray datasets in HIBD were downloaded from the Gene Expression Omnibus database, and DEGs were identied by R software.Enrichment analyses were performed and protein–protein interaction networks were constructed to understand the functions and enriched pathways of DEGs and to identify central genes and key modules. Results In the cerebral cortex tissue with HIBD, 2598 DEGs were identied, including 2362 up-regulated and 236 down-regulated DEGs. -

A Non-Synonymous SNP Within Membrane Metalloendopeptidase-Like 1 (MMEL1) Is Associated with Multiple Sclerosis
Genes and Immunity (2010) 11, 660–664 & 2010 Macmillan Publishers Limited All rights reserved 1466-4879/10 www.nature.com/gene ORIGINAL ARTICLE A non-synonymous SNP within membrane metalloendopeptidase-like 1 (MMEL1) is associated with multiple sclerosis M Ban1,15, JL McCauley2,15, R Zuvich3,15, A Baker1, L Bergamaschi4, M Cox5, A Kemppinen1, S D’Alfonso4, FR Guerini6, J Lechner-Scott5, F Dudbridge7, J Wason7, NP Robertson8, PL De Jager9,10, DA Hafler11, LF Barcellos12, AJ Ivinson13, D Sexton3, JR Oksenberg14, SL Hauser14, MA Pericak-Vance2, J Haines3, A Compston1 and S Sawcer1 1Department of Clinical Neuroscience, University of Cambridge, Addenbrooke’s Hospital, Cambridge, UK; 2Miami Institute for Human Genomics, University of Miami Miller School of Medicine, Miami, FL, USA; 3Center for Human Genetics Research, Vanderbilt University Medical Center, Nashville, TN, USA; 4Department of Medical Sciences and Interdisciplinary Research Center of Autoimmune Diseases, University of Eastern Piedmont, Novara, Italy; 5University of Newcastle, John Hunter Hospital, Newcastle, New South Wales, Australia; 6Laboratory of Molecular Medicine and Biotechnology, Don C Gnocchi Foundation IRCCS, S Maria Nascente, Milan, Italy; 7MRC Biostatistics Unit, Cambridge, UK; 8Department of Neurology, University Hospital of Wales, Heath Park, Cardiff, UK; 9Program in Translational NeuroPsychiatric Genomics, Center for Neurologic Diseases, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA, USA; 10Broad Institute, Cambridge, MA, USA; 11Department of Neurology, Yale University School of Medicine, New Haven, CT, USA; 12Division of Epidemiology, School of Public Health, University of California at Berkeley, Berkeley, CA, USA; 13Harvard NeuroDiscovery Center, Harvard Medical School, Boston, MA, USA and 14Department of Neurology, University of California San Francisco, San Francisco, CA, USA Several single-nucleotide polymorphism (SNP) genome-wide association studies (GWASs) have been completed in multiple sclerosis (MS). -

Nº Ref Uniprot Proteína Péptidos Identificados Por MS/MS 1 P01024
Document downloaded from http://www.elsevier.es, day 26/09/2021. This copy is for personal use. Any transmission of this document by any media or format is strictly prohibited. Nº Ref Uniprot Proteína Péptidos identificados 1 P01024 CO3_HUMAN Complement C3 OS=Homo sapiens GN=C3 PE=1 SV=2 por 162MS/MS 2 P02751 FINC_HUMAN Fibronectin OS=Homo sapiens GN=FN1 PE=1 SV=4 131 3 P01023 A2MG_HUMAN Alpha-2-macroglobulin OS=Homo sapiens GN=A2M PE=1 SV=3 128 4 P0C0L4 CO4A_HUMAN Complement C4-A OS=Homo sapiens GN=C4A PE=1 SV=1 95 5 P04275 VWF_HUMAN von Willebrand factor OS=Homo sapiens GN=VWF PE=1 SV=4 81 6 P02675 FIBB_HUMAN Fibrinogen beta chain OS=Homo sapiens GN=FGB PE=1 SV=2 78 7 P01031 CO5_HUMAN Complement C5 OS=Homo sapiens GN=C5 PE=1 SV=4 66 8 P02768 ALBU_HUMAN Serum albumin OS=Homo sapiens GN=ALB PE=1 SV=2 66 9 P00450 CERU_HUMAN Ceruloplasmin OS=Homo sapiens GN=CP PE=1 SV=1 64 10 P02671 FIBA_HUMAN Fibrinogen alpha chain OS=Homo sapiens GN=FGA PE=1 SV=2 58 11 P08603 CFAH_HUMAN Complement factor H OS=Homo sapiens GN=CFH PE=1 SV=4 56 12 P02787 TRFE_HUMAN Serotransferrin OS=Homo sapiens GN=TF PE=1 SV=3 54 13 P00747 PLMN_HUMAN Plasminogen OS=Homo sapiens GN=PLG PE=1 SV=2 48 14 P02679 FIBG_HUMAN Fibrinogen gamma chain OS=Homo sapiens GN=FGG PE=1 SV=3 47 15 P01871 IGHM_HUMAN Ig mu chain C region OS=Homo sapiens GN=IGHM PE=1 SV=3 41 16 P04003 C4BPA_HUMAN C4b-binding protein alpha chain OS=Homo sapiens GN=C4BPA PE=1 SV=2 37 17 Q9Y6R7 FCGBP_HUMAN IgGFc-binding protein OS=Homo sapiens GN=FCGBP PE=1 SV=3 30 18 O43866 CD5L_HUMAN CD5 antigen-like OS=Homo