Toxicological Profile for Naphthalene, 1-Methylnaphthalene, and 2-Methylnaphthalene
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Reactivity and Functionalization of Naphthalene and Anthracene Complexes of {Tpw(NO)(Pme3)}
Reactivity and Functionalization of Naphthalene and Anthracene Complexes of {TpW(NO)(PMe3)} Laura Jessica Strausberg Baltimore, Maryland B.A., Hollins University, 2008 A Dissertation presented to the Graduate Faculty of the University of Virginia in Candidacy for the Degree of Doctor of Philosophy Department of Chemistry University of Virginia July, 2013 ii Abstract Chapter 1 introduces the organic chemistry of aromatic hydrocarbons, with attention paid to regiochemical outcomes of organic reactions. The binding of naphthalene and anthracene to metal complexes is discussed, along with organic transformations they undergo as a result of their complexation. The previous work on osmium and rhenium complexes of naphthalene from the Harman group is explored. Finally, some spectroscopic techniques for exploring the chemistry of {TpW(NO)(PMe3)} complexes of naphthalene and anthracene are introduced. Chapter 2 discusses the highly distorted allyl complexes formed from {TpW(NO)(PMe3)} and the exploration of their origin. Attempts at stereoselectively deprotonating these cationic complexes is also discussed. 2 Chapter 3 describes our study of TpW(NO)(PMe3)(3,4-η -naphthalene)’s ability to undergo a Diels-Alder reaction with N-methylmaleimide. A solvent study suggested that this reaction proceeds by a concerted mechanism. To probe the mechanism further, we synthesized a series of methylated and methoxylated naphthalene complexes and measured their rates of reaction with N-methylmaleimide compared to the parent complex. We found that 1- substitution on the naphthalene increased the rate of cycloaddition, even if the substituent was in the unbound ring, while 2-substitution slowed the reaction rate when in the bound ring. This information is consistent with a concerted mechanism, as a 2-substituted product would be less able to isomerize to form the active isomer for the cycloaddition to occur. -

Supplementary Information for “Oligomeric Models for Estimation of Polydimethylsiloxane
Electronic Supplementary Material (ESI) for Environmental Science: Processes & Impacts. This journal is © The Royal Society of Chemistry 2016 Supplementary Information for “Oligomeric models for estimation of polydimethylsiloxane- water partition ratios with COSMO-RS theory: Impact of the combinatorial term on absolute error” by J. Mark Parnis and Donald Mackay. Contents: 1) The complete set of chemicals, with the published values of log K(PDMS-w), and the abbreviated reference, corresponding to references given in the principle text. 1) The complete set of chemicals, with the published values of log KPDMS-w. Hsieh 2011 CHAO 2014 PCB18 4.91 PCB203 7.09 PCB16 5.12 PCB195 6.89 PCB32 5.12 PCB194 6.79 PCB28,31 5.17 Benzyl alcohol -0.35 PCB33,53 5.18 4-Fluorophenol -0.28 PCB22 5.30 m-Cresol -0.03 PCB52 5.48 Phenethyl alcohol 0.12 PCB47,48 5.49 3-Methylbenzyl alcohol 0.17 PCB44 5.44 3-Chlorophenol 0.31 PCB71 5.49 3,5-Dimethylphenol 0.42 PCB41 5.49 3-Bromophenol 0.46 PCB70 5.79 4-Ethylphenol 0.60 PCB66 5.70 4-Chloroaniline 0.84 PCB95 5.77 Phenyl acetate 0.86 PCB56,60 5.86 Benzonitrile 1.04 PCB101 6.01 Acetophenone 1.04 PCB99 6.17 4-Chloroacetophenone 1.64 PCB83 6.02 Methyl benzoate 1.65 PCB97 6.05 Benzene 1.76 PCB87 6.19 Ethylbenzoate 2.12 PCB85 6.38 Toluene 2.28 PCB110 6.03 4-Chloroanisole 2.37 PCB151 6.31 Chlorobenzene 2.40 PCB135 6.48 Bromobenzene 2.51 PCB149 6.42 o-Xylene 2.69 PCB118 6.23 m-Xylene 2.73 PCB146 6.66 Iodobenzene 2.73 PCB153 6.62 p-Xylene 2.75 PCB105,132 6.40 Ethylbenzene 2.75 PCB179 6.73 Phenol -0.18 PCB141 6.73 Naphthalene 2.83 PCB138 6.61 Cyclopentane 2.85 PCB163 6.56 4-Chlorotoluene 2.87 PCB158 6.83 Methylcyclopentane 3.13 PCB187 6.96 Propylbenzene 3.14 PCB182 6.96 Isopropylbenzene 3.15 PCB183 6.26 1,3,5-Trimethylbenzene 3.16 PCB128 6.61 1-Methyl-4-Ethylbenzene 3.20 PCB185 6.86 Cyclohexane 3.20 PCB174 7.04 1-Methylnaphthalene 3.26 PCB177 7.02 1,2,4-Trimethylbenzene 3.34 PCB171,202 6.78 tert-Butylbenzene 3.34 PCB180 6.89 Biphenyl 3.37 PCB170 6.82 2,3-Dimethylbutane 3.37 PCB201 7.06 2-Methylpentane 3.51 CHAO 2014 Kang et al. -

Pesticides EPA 738-R-07-010 Environmental Protection and Toxic Substances September 12, 2008 Agency (7508P)
United States Prevention, Pesticides EPA 738-R-07-010 Environmental Protection and Toxic Substances September 12, 2008 Agency (7508P) Reregistration Eligibility Decision for Naphthalene ii TABLE OF CONTENTS I. Introduction .............................................................................................................................. 8 II. Chemical Overview................................................................................................................. 8 A. Regulatory History............................................................................................................ 8 B. Chemical Identification....................................................................................................... 9 C. Use Profile............................................................................................................................ 9 D. Estimated Usage of Pesticide............................................................................................ 10 III. Summary of Naphthalene Risk Assessments........................................................................ 10 A. Human Health Risk Assessment......................................................................................... 10 1. Toxicity of Naphthalene ................................................................................................ 11 2. Carcinogenicity of Naphthalene ...................................................................................... 16 3. Metabolites and Degradates......................................................................................... -

Minutes of the IUPAC Chemical Nomenclature and Structure Representation Division (VIII) Committee Meeting Boston, MA, USA, August 18, 2002
Minutes of the IUPAC Chemical Nomenclature and Structure Representation Division (VIII) Committee Meeting Boston, MA, USA, August 18, 2002 Members Present: Dr Stephen Heller, Prof Herbert Kaesz, Prof Dr Alexander Lawson, Prof G. Jeffrey Leigh, Dr Alan McNaught (President), Dr. Gerard Moss, Prof Bruce Novak, Dr Warren Powell (Secretary), Dr William Town, Dr Antony Williams Members Absent: Dr. Michael Dennis, Prof Michael Hess National representatives Present: Prof Roberto de Barros Faria (Brazil) The second meeting of the Division Committee of the IUPAC Division of Chemical Nomenclature and Structure Representation held in the Great Republic Room of the Westin Hotel in Boston, Massachusetts, USA was convened by President Alan McNaught at 9:00 a.m. on Sunday, August 18, 2002. 1.0 President McNaught welcomed the members to this meeting in Boston and offered a special welcome to the National Representative from Brazil, Prof Roberto de Barros Faria. He also noted that Dr Michael Dennis and Prof Michael Hess were unable to be with us. Each of the attendees introduced himself and provided a brief bit of background information. Housekeeping details regarding breaks and lunch were announced and an invitation to a reception from the U. S. National Committee for IUPAC on Tuesday, August 20 was noted. 2.0 The agenda as circulated was approved with the addition of a report from Dr Moss on the activity on his website. 3.0 The minutes of the Division Committee Meeting in Cambridge, UK, January 25, 2002 as posted on the Webboard (http://www.rsc.org/IUPAC8/attachments/MinutesDivCommJan2002.rtf and http://www.rsc.org/IUPAC8/attachments/MinutesDivCommJan2002.pdf) were approved with the following corrections: 3.1 The name Dr Gerard Moss should be added to the members present listing. -

Combined PIANO Standard
Combined PIANO Standard Product #: VHG-PIANO-COM-0.1 Lot #: 711069047B Concentration Concentration Component Component (Wt.%) (Wt.%) Isoparaffins Isopentane 0.3371 4-Methylheptane 0.4939 2,3-Dimethylbutane 0.0691 3-Methylheptane 0.8406 2-Methylpentane 0.5031 3-Ethylhexane 0.1097 3-Methylpentane 0.8271 3,3-Dimethylheptane 0.2590 2,2-Dimethylpentane 0.2746 2,5-Dimethylheptane 0.8678 2,4-Dimethylpentane 0.5679 3,5-Dimethylheptane 0.1169 2,2,3-Trimethylbutane 0.6042 2,3-Dimethylheptane 0.2288 3,3-Dimethylpentane 0.2873 3,4-Dimethylheptane 0.5668 2-Methylhexane 0.3643 2-Methyloctane 0.5791 2,3-Dimethylpentane 0.2751 3-Methyloctane 0.8645 3-Methylhexane 0.2475 3,3-Diethylpentane 0.2424 3-Ethylpentane 0.0813 2,2-Dimethyloctane 0.5024 2,2-Dimethylhexane 0.2027 3,3-Dimethyloctane 0.4902 2,5-Dimethylhexane 0.5727 2,3-Dimethyloctane 0.5927 2,2,3-Trimethylpentane 0.2658 3-Ethyloctane 0.5694 2,4-Dimethylhexane 0.2542 2-Methylnonane 0.5727 2,3-Dimethylhexane 0.2492 3-Methylnonane 0.8889 2-Methylheptane 0.6744 Aromatics Benzene 1.6808 1-Methyl-3-n-propylbenzene 0.4972 Toluene 1.0772 1-Methyl-4-n-propylbenzene 0.5237 Ethylbenzene 1.5918 n-Butylbenzene 0.5198 m-Xylene 0.5341 1,2-Diethylbenzene 0.2577 p-Xylene 1.1328 1-Methyl-2-n-propylbenzene 0.5282 o-Xylene 0.5315 1,4-Dimethyl-2-ethylbenzene 0.5379 Isopropylbenzene 0.5235 1,3-Dimethyl-5-ethylbenzene 0.5176 n-Propylbenzene 1.0731 1,2-Dimethyl-4-ethylbenzene 0.5310 1-Methyl-3-ethylbenzene 0.5181 1,3-Dimethyl-2-ethylbenzene 0.2695 1-Methyl-4-ethylbenzene 0.5104 1,2-Dimethyl-3-ethylbenzene 0.5151 1,3,5-Trimethylbenzene -

Safety Data Sheet Acc
Page 1/10 Safety Data Sheet acc. to OSHA HCS Printing date 03/24/2019 Version Number 2 Reviewed on 03/24/2019 * 1 Identification · Product identifier · Trade name: Acrylamide · Part number: RCC-203 · CAS Number: 79-06-1 · EC number: 201-173-7 · Index number: 616-003-00-0 · Application of the substance / the mixture Reagents and Standards for Analytical Chemical Laboratory Use · Details of the supplier of the safety data sheet · Manufacturer/Supplier: Agilent Technologies, Inc. 5301 Stevens Creek Blvd. Santa Clara, CA 95051 USA · Information department: Telephone: 800-227-9770 e-mail: [email protected] · Emergency telephone number: CHEMTREC®: 1-800-424-9300 2 Hazard(s) identification · Classification of the substance or mixture GHS06 Skull and crossbones Acute Tox. 3 H301 Toxic if swallowed. GHS08 Health hazard Muta. 1B H340 May cause genetic defects. Carc. 1B H350 May cause cancer. Repr. 2 H361 Suspected of damaging fertility or the unborn child. STOT RE 1 H372 Causes damage to organs through prolonged or repeated exposure. GHS07 Acute Tox. 4 H312 Harmful in contact with skin. Acute Tox. 4 H332 Harmful if inhaled. Skin Irrit. 2 H315 Causes skin irritation. Eye Irrit. 2A H319 Causes serious eye irritation. Skin Sens. 1 H317 May cause an allergic skin reaction. · Label elements · GHS label elements The substance is classified and labeled according to the Globally Harmonized System (GHS). (Contd. on page 2) US 48.1.26 Page 2/10 Safety Data Sheet acc. to OSHA HCS Printing date 03/24/2019 Version Number 2 Reviewed on 03/24/2019 Trade name: Acrylamide (Contd. -

Chapter 16 Liquid and Solids
Homework #2 Chapter 16 Liquid and Solids 7. Vapor Pressure: The pressure exerted by the vapor of a liquid when the vapor and the liquid are in dynamic equilibrium. The vapor pressure reflects the fact that within a system there is a distribution of energies that molecules can have, therefore, some molecules will have enough energy to overcome the intermolecular forces and enter into the gas phase. All liquids have some vapor pressure. The stronger the intermolecular forces the smaller the vapor pressure. All solids also have a vapor pressure. This is why if you leave ice in the freezer for a long time it “disappears.” The vapor pressure of solids is less than the vapor pressure of liquids. As the temperature increases the molecules have more energy, therefore, more molecules can escape into the gas phase (vapor pressure increases). When the vapor pressure is equal to the atmospheric pressure the solution boils. 9. a) Surface Tension As the intermolecular forces increase (↑), surface tension increases (↑). b) Viscosity As the intermolecular forces increase (↑), the viscosity increases (↑). c) Melting Point As the intermolecular forces increase (↑), the melting point increases (↑). d) Boiling Point As the intermolecular forces increase (↑), the boiling point increases (↑). e) Vapor Pressure As the intermolecular forces increase (↑), the vapor pressure decreases (↓). 11. Intermolecular Forces: The forces of attraction/repulsion between molecules. Intramolecular Forces: The forces of attraction/repulsion within a molecule. Intramolecular forces are stronger the intermolecular forces. Types of intermolecular forces: Dipole-Dipole Forces: The interaction between two electric dipoles in different molecules. Hydrogen Bonding: The attraction between a hydrogen atom (that is bonded to an O, N, or F atom) and an O, N, or F atom in a neighboring molecule. -

SAFETY DATA SHEET Cyclopentane
SAFETY DATA SHEET Cyclopentane Section 1. Identification GHS product identifier : Cyclopentane Chemical name : cyclopentane Other means of : cyclopentane (dot); pentamethylene identification Product use : Synthetic/Analytical chemistry. Synonym : cyclopentane (dot); pentamethylene SDS # : 001124 Supplier's details : Airgas USA, LLC and its affiliates 259 North Radnor-Chester Road Suite 100 Radnor, PA 19087-5283 1-610-687-5253 24-hour telephone : 1-866-734-3438 Section 2. Hazards identification OSHA/HCS status : This material is considered hazardous by the OSHA Hazard Communication Standard (29 CFR 1910.1200). Classification of the : FLAMMABLE LIQUIDS - Category 2 substance or mixture AQUATIC HAZARD (ACUTE) - Category 3 AQUATIC HAZARD (LONG-TERM) - Category 3 GHS label elements Hazard pictograms : Signal word : Danger Hazard statements : Highly flammable liquid and vapor. May form explosive mixtures in Air. Harmful to aquatic life with long lasting effects. Precautionary statements General : Read label before use. Keep out of reach of children. If medical advice is needed, have product container or label at hand. Prevention : Wear protective gloves. Wear eye or face protection. Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. No smoking. Use explosion- proof electrical, ventilating, lighting and all material-handling equipment. Use only non- sparking tools. Take precautionary measures against static discharge. Keep container tightly closed. Avoid release to the environment. Response : IF ON SKIN (or hair): Take off immediately all contaminated clothing. Rinse skin with water or shower. Storage : Store in a well-ventilated place. Keep cool. Disposal : Dispose of contents and container in accordance with all local, regional, national and international regulations. Hazards not otherwise : None known. -
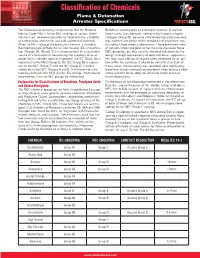
Classification of Chemicals
Classification of Chemicals Flame & Detonation Arrester Specifications PROTECTOSEAL ® The Protectoseal Company recommends that the National Butadiene would qualify as a Group D material. In each of Electric Code (NEC) Article 500, rankings of various chemi - these cases, the chemicals were primarly listed in a higher cals be used, whenever possible, to determine the suitability category (Group B), because of relatively high pressure read - of a detonation arrester for use with a particular chemical. ings noted in one phase of the standard test procedure con - When no NEC rating of the particular chemical is available, ducted by Underwriters Laboratories. These pressures were the International Electrotechnical Commission (IEC) classifica - of concern when categorizing the chemicals because these tion (Groups IIA, IIB and IIC) is recommended as a secondary NEC groupings are also used as standard indicators for the source of information for determining the suitability of an ar - design strength requirements of electrical boxes, apparatus, rester for its intended service. In general, the IEC Group IIA is etc. that must withstand the pressures generated by an igni - equivalent to the NEC Group D; the IEC Group IIB is equiva - tion within the container. It should be noted that, in each of lent to the NEC Group C; and the IEC Group IIC includes these cases, the test pressures recorded were significantly chemicals in the NEC Groups A and B. In the event of a dis - lower than those commonly encountered when testing a deto - crepancy between the NEC and the IEC ratings, Protectoseal nation arrester for its ability to withstand stable and over - recommends that the NEC groups be referenced. -

WHO Guidelines for Indoor Air Quality : Selected Pollutants
WHO GUIDELINES FOR INDOOR AIR QUALITY WHO GUIDELINES FOR INDOOR AIR QUALITY: WHO GUIDELINES FOR INDOOR AIR QUALITY: This book presents WHO guidelines for the protection of pub- lic health from risks due to a number of chemicals commonly present in indoor air. The substances considered in this review, i.e. benzene, carbon monoxide, formaldehyde, naphthalene, nitrogen dioxide, polycyclic aromatic hydrocarbons (especially benzo[a]pyrene), radon, trichloroethylene and tetrachloroethyl- ene, have indoor sources, are known in respect of their hazard- ousness to health and are often found indoors in concentrations of health concern. The guidelines are targeted at public health professionals involved in preventing health risks of environmen- SELECTED CHEMICALS SELECTED tal exposures, as well as specialists and authorities involved in the design and use of buildings, indoor materials and products. POLLUTANTS They provide a scientific basis for legally enforceable standards. World Health Organization Regional Offi ce for Europe Scherfi gsvej 8, DK-2100 Copenhagen Ø, Denmark Tel.: +45 39 17 17 17. Fax: +45 39 17 18 18 E-mail: [email protected] Web site: www.euro.who.int WHO guidelines for indoor air quality: selected pollutants The WHO European Centre for Environment and Health, Bonn Office, WHO Regional Office for Europe coordinated the development of these WHO guidelines. Keywords AIR POLLUTION, INDOOR - prevention and control AIR POLLUTANTS - adverse effects ORGANIC CHEMICALS ENVIRONMENTAL EXPOSURE - adverse effects GUIDELINES ISBN 978 92 890 0213 4 Address requests for publications of the WHO Regional Office for Europe to: Publications WHO Regional Office for Europe Scherfigsvej 8 DK-2100 Copenhagen Ø, Denmark Alternatively, complete an online request form for documentation, health information, or for per- mission to quote or translate, on the Regional Office web site (http://www.euro.who.int/pubrequest). -

MSDS Cyclopentane (1).Pdf
MATERIAL SAFETY DATA SHEET (MSDS) CYCLOPENTANE 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION SUBSTANCE OR PREPATION TRADE NAME Cyclopentane CHEMICAL CLASSIFICATION Cycloparaffin, Naphthene COMPANY/ UNDERTAKING NAME AND Haldia Petrochemicals Limited, ADDRESS PO Box No 12, Haldia Plant PO Durgachak, Dist Midnapore West Bengal, India PIN 721 602 TELEPHONE 091-3224-274384 / 274400 EMERGENCY TELEPHONE NUMBER 091-3224-275916 2. COMPOSTION AND INFORMATION ON INGREDIENTS CHEMICAL CHEMICLAL CONTENT CAS EXPOSURE LIMITS IN AIR NAME FORMULA NUMBER (ppm) ACGIH ACGIH IDLH TLV- TLV- TWA STEL Cyclopentane C5H10 98.20 wt% 287-92-3 600 NA NA 2,2 Dimethyl (CH3)3CCH2CH3 0.28 wt% 75-83-2 500 1000 NA butane n- pentane n- C5H12 1.47 wt% 109-66-0 600 750 NA i- Pentane i- C5H12 0.05 wt% 78-78-4 600 750 NA Total Sulpher S < 1.0 wt. ppm 7704-34-9 NA NA NA 3. HAZARD CLASSIFICATION Highly flammable liquid and vapour. Vapour EMERGENCY OVERVIEW may cause flash fire. Harmful or Fatal is enter lungs and cause damage POTENTIAL HEALTH HAZARDS EYE SKIN INHALATION INGESTION OTHERS ACUTE To cause Not expected Dizziness, Abdominal prolonged or to be harmful headache, pain, significant to internal nausea, diarrhoea, eye irritation organs if Unconsciousness, dizziness, absorbed weakness nausea, sore through the throat skin. CHRONIC Repeated or prolonged contact with skin may cause dermatitis NFPA HAZARD HEALTH FLAMMABILITY REACTIVITY SPECIAL SIGNALS 2 3 0 - HAZCHEM CODE 3YE GHS-Classification Flammable liquids Category 2 Aspiration hazard Category 1 Chronic aquatic toxicity, Category 3 Acute aquatic toxicity, Category 3 Target Organ Systemic Toxicant - Single exposure, Category 3, Inhalation, Central nervous system Document Compiled By Approved By Issue No Rev. -

Revealing Injection Vulnerabilities by Leveraging Existing Tests
Revealing Injection Vulnerabilities by Leveraging Existing Tests Katherine Hough1, Gebrehiwet Welearegai2, Christian Hammer2 and Jonathan Bell1 1George Mason University, Fairfax, VA, USA 2University of Potsdam, Potsdam, Germany [email protected],[email protected],[email protected],[email protected] Abstract just one of over 8,200 similar code injection exploits discovered in Code injection attacks, like the one used in the high-prole 2017 recent years in popular software [44]. Code injection vulnerabilities Equifax breach, have become increasingly common, now ranking have been exploited in repeated attacks on US election systems [10, #1 on OWASP’s list of critical web application vulnerabilities. Static 18, 39, 61], in the theft of sensitive nancial data [56], and in the analyses for detecting these vulnerabilities can overwhelm develop- theft of millions of credit card numbers [33]. In the past several ers with false positive reports. Meanwhile, most dynamic analyses years, code injection attacks have persistently ranked at the top rely on detecting vulnerabilities as they occur in the eld, which of the Open Web Application Security Project (OWASP) top ten can introduce a high performance overhead in production code. most dangerous web aws [46]. Injection attacks can be damaging This paper describes a new approach for detecting injection vul- even for applications that are not traditionally considered critical nerabilities in applications by harnessing the combined power of targets, such as personal websites, because attackers can use them human developers’ test suites and automated dynamic analysis. as footholds to launch more complicated attacks. Our new approach, Rivulet, monitors the execution of developer- In a code injection attack, an adversary crafts a malicious in- written functional tests in order to detect information ows that put that gets interpreted by the application as code rather than may be vulnerable to attack.