A View of Extraterrestrial Soils G
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Book Review1.Fm
Meteoritics & Planetary Science 42, Nr 9, 1695–1696 (2007) http://meteoritics.org Book Review Protostars and planets V, edited by Reipurth B, Jewitt D., and Keil K. Tucson, Arizona: The University of Arizona Press, 2007, 1024 p., cloth (ISBN 978-0-8165-2654-3). Some thirty years ago, planetary science and the study of star formation were different scientific fields with different practitioners who rarely interacted with one another. Planetary scientists, mostly geochemists and geophysicists, resided in geology departments whereas star formation was the domain of astronomers. All this has changed starting with Tom Gehrels who organized the first Protostars and Planets conference in January 1978 in Tucson. In the book that followed this conference, which was edited by Gehrels, he expressed the hope “to develop the interface between studies of star formation and those of the origin of the solar system.” This hope has been fulfilled beyond expectations. The first volume has been followed by four more in intervals of about seven years. Each book was preceded by a conference. The Protostars and Planets V Conference took place at the Hilton Waikoloa Village on the Big Island of Hawai‘i on October 24–28, 2005. The subsequent book appeared early this year. As its forerunners, it is part of the University of Arizona The arrangement of the chapters roughly follows the Space Science Series, which is devoted to different aspects of inferred prehistory of our own solar system. In the dense core solar system science. of a molecular cloud, gravity overcomes thermal and The rapid growth of this new interdisciplinary field magnetic pressures, leading to the formation of a star, in most between astronomy and planetary science is demonstrated by case actually of a binary star system. -

And Abiogenesis
Historical Development of the Distinction between Bio- and Abiogenesis. Robert B. Sheldon NASA/MSFC/NSSTC, 320 Sparkman Dr, Huntsville, AL, USA ABSTRACT Early greek philosophers laid the philosophical foundations of the distinction between bio and abiogenesis, when they debated organic and non-organic explanations for natural phenomena. Plato and Aristotle gave organic, or purpose-driven explanations for physical phenomena, whereas the materialist school of Democritus and Epicurus gave non-organic, or materialist explanations. These competing schools have alternated in popularity through history, with the present era dominated by epicurean schools of thought. Present controversies concerning evidence for exobiology and biogenesis have many aspects which reflect this millennial debate. Therefore this paper traces a selected history of this debate with some modern, 20th century developments due to quantum mechanics. It ¯nishes with an application of quantum information theory to several exobiology debates. Keywords: Biogenesis, Abiogenesis, Aristotle, Epicurus, Materialism, Information Theory 1. INTRODUCTION & ANCIENT HISTORY 1.1. Plato and Aristotle Both Plato and Aristotle believed that purpose was an essential ingredient in any scienti¯c explanation, or teleology in philosophical nomenclature. Therefore all explanations, said Aristotle, answer four basic questions: what is it made of, what does it represent, who made it, and why was it made, which have the nomenclature material, formal, e±cient and ¯nal causes.1 This aristotelean framework shaped the terms of the scienti¯c enquiry, invisibly directing greek science for over 500 years. For example, \organic" or \¯nal" causes were often deemed su±cient to explain natural phenomena, so that a rock fell when released from rest because it \desired" its own kind, the earth, over unlike elements such as air, water or ¯re. -

ET First Extract
May 2018 Evangelical TIMES 23 What is man (1): Are we alone in the universe? The first of three edited extracts from Edgar Andrews’ new book, What is Man? Adam, alien or ape? Prof Edgar Andrews It’s easy to confuse SETI with YETI because both relate to are more likely to swallow small bugs than little green men. the National Science Foundation, Ohio State University began creatures that probably don’t exist. The Yeti is a proverbial I should make it clear that the planet’s discoverers said constructing a radio observatory called ‘Big Ear’ — which beast that inhabits the Himalayas and walks upright like a nothing about bugs — that was an invention by the BBC. later undertook the world’s first continuous SETI program. human, but leaves huge footprints in the snow and strikes But their claim that Gliese 581c is a rocky planet like Earth On 15 August 1977, Jerry Ehman, a project volunteer at fear into the local populace. Related to America’s Bigfoot, it and has liquid water on its surface is itself based on a string the observatory, observed a strong signal and wrote ‘Wow!’ is sometimes called the Abominable Snowman (apparently of assumptions. on the recorded trace. Unsurprisingly known as the Wow! due to a mistranslation of its Nepalese name). An informed website respondent commented: ‘You must signal, some enthusiasts consider it the best candidate to date SETI, on the other hand, stands for ‘the search for extra- remember that neither the mass nor the radius of this planet for a cosmic radio signal from an artificial source. -

Viscosity of Gases References
VISCOSITY OF GASES Marcia L. Huber and Allan H. Harvey The following table gives the viscosity of some common gases generally less than 2% . Uncertainties for the viscosities of gases in as a function of temperature . Unless otherwise noted, the viscosity this table are generally less than 3%; uncertainty information on values refer to a pressure of 100 kPa (1 bar) . The notation P = 0 specific fluids can be found in the references . Viscosity is given in indicates that the low-pressure limiting value is given . The dif- units of μPa s; note that 1 μPa s = 10–5 poise . Substances are listed ference between the viscosity at 100 kPa and the limiting value is in the modified Hill order (see Introduction) . Viscosity in μPa s 100 K 200 K 300 K 400 K 500 K 600 K Ref. Air 7 .1 13 .3 18 .5 23 .1 27 .1 30 .8 1 Ar Argon (P = 0) 8 .1 15 .9 22 .7 28 .6 33 .9 38 .8 2, 3*, 4* BF3 Boron trifluoride 12 .3 17 .1 21 .7 26 .1 30 .2 5 ClH Hydrogen chloride 14 .6 19 .7 24 .3 5 F6S Sulfur hexafluoride (P = 0) 15 .3 19 .7 23 .8 27 .6 6 H2 Normal hydrogen (P = 0) 4 .1 6 .8 8 .9 10 .9 12 .8 14 .5 3*, 7 D2 Deuterium (P = 0) 5 .9 9 .6 12 .6 15 .4 17 .9 20 .3 8 H2O Water (P = 0) 9 .8 13 .4 17 .3 21 .4 9 D2O Deuterium oxide (P = 0) 10 .2 13 .7 17 .8 22 .0 10 H2S Hydrogen sulfide 12 .5 16 .9 21 .2 25 .4 11 H3N Ammonia 10 .2 14 .0 17 .9 21 .7 12 He Helium (P = 0) 9 .6 15 .1 19 .9 24 .3 28 .3 32 .2 13 Kr Krypton (P = 0) 17 .4 25 .5 32 .9 39 .6 45 .8 14 NO Nitric oxide 13 .8 19 .2 23 .8 28 .0 31 .9 5 N2 Nitrogen 7 .0 12 .9 17 .9 22 .2 26 .1 29 .6 1, 15* N2O Nitrous -

Cumulated Bibliography of Biographies of Ocean Scientists Deborah Day, Scripps Institution of Oceanography Archives Revised December 3, 2001
Cumulated Bibliography of Biographies of Ocean Scientists Deborah Day, Scripps Institution of Oceanography Archives Revised December 3, 2001. Preface This bibliography attempts to list all substantial autobiographies, biographies, festschrifts and obituaries of prominent oceanographers, marine biologists, fisheries scientists, and other scientists who worked in the marine environment published in journals and books after 1922, the publication date of Herdman’s Founders of Oceanography. The bibliography does not include newspaper obituaries, government documents, or citations to brief entries in general biographical sources. Items are listed alphabetically by author, and then chronologically by date of publication under a legend that includes the full name of the individual, his/her date of birth in European style(day, month in roman numeral, year), followed by his/her place of birth, then his date of death and place of death. Entries are in author-editor style following the Chicago Manual of Style (Chicago and London: University of Chicago Press, 14th ed., 1993). Citations are annotated to list the language if it is not obvious from the text. Annotations will also indicate if the citation includes a list of the scientist’s papers, if there is a relationship between the author of the citation and the scientist, or if the citation is written for a particular audience. This bibliography of biographies of scientists of the sea is based on Jacqueline Carpine-Lancre’s bibliography of biographies first published annually beginning with issue 4 of the History of Oceanography Newsletter (September 1992). It was supplemented by a bibliography maintained by Eric L. Mills and citations in the biographical files of the Archives of the Scripps Institution of Oceanography, UCSD. -
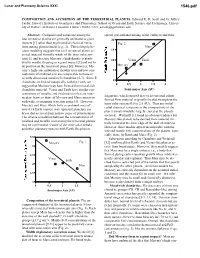
Composition and Accretion of the Terrestrial Planets
Lunar and Planetary Science XXXI 1546.pdf COMPOSITION AND ACCRETION OF THE TERRESTRIAL PLANETS. Edward R. D. Scott and G. Jeffrey Taylor, Hawai’i Institute of Geophysics and Planetology, School of Ocean and Earth Science and Technology, Univer- sity of Hawai’i at Manoa, Honolulu, Hawai’i 96822, USA; [email protected] Abstract: Compositional variations among the spread gravitational mixing of the embryos and their four terrestrial planets are generally attributed to giant 20 impacts [1] rather than to primordial chemical varia- Fig. 2 tions among planetesimals [e.g., 2]. This is largely be- Mars cause modeling suggests that each terrestrial planet ac- 15 creted material from the whole of the inner solar sys- tem [1], and because Mercury’s high density is attrib- 10 Venus Earth uted to mantle stripping in a giant impact [3] and not to its position as the innermost planet [4]. However, Mer- Mercury cury’s high concentration of metallic iron and low con- 5 centration of oxidized iron are comparable to those in recently discovered metal-rich chondrites [5-7]. Since E 0 chondrites are linked isotopically with the Earth, we 0 0.5 1 1.5 2 suggest that Mercury may have formed from metal-rich chondritic material. Venus and Earth have similar con- Semi-major Axis (AU) centrations of metallic and oxidized iron that are inter- fragments, which ensured that each terrestrial planet mediate between those of Mercury and Mars consistent formed from material originally located throughout the with wide, overlapping accretion zones [1]. However, inner solar system (0.5 to 2.5 AU). -

POSSIBLE STRUCTURE MODELS for the TRANSITING SUPER-EARTHS:KEPLER-10B and 11B
43rd Lunar and Planetary Science Conference (2012) 1290.pdf POSSIBLE STRUCTURE MODELS FOR THE TRANSITING SUPER-EARTHS:KEPLER-10b AND 11b. P. Futó1 1 Department of Physical Geography, University of West Hungary, Szombathely, Károlyi Gáspár tér, H- 9700, Hungary ([email protected]) Introduction:Up to january of 2012,10 super- The planet Kepler-11b has a large radius (1.97 R⊕) for Earths have been announced by Kepler-mission [1] its mass (4.3 M⊕),therefore this planet must have a that is designed to detect hundreds of transiting exo- spherical shell that is composed of low-density materi- planets.Kepler was launched on 6th March,2009 and the als.Considering the planet's average density,it must primary purpose of its scientific program is to search have a metallic core with different possible fractional for terrestrial-sized planets in the habitable zone of mass.Accordinghly,I have made a possible structure Solar-like stars.For the case of high number of discov- model for Kepler-11b in which the selected core mass eries we will be able to estimate the frequency of fraction is 32.59% (similarly to that of Earth) and the Earth-sized planets in our galaxy.Results of the Kepler- water ice layer has a relatively great fractional 's measurements show that the small-sized planets are volume.For case of the selected composition, the icy frequent in the spiral galaxies.A catalog of planetary surface sublimated to form a water vapor as the planet candidates,including objects with small-sized candidate moved inward the central star during its migration. -

Planetary Science
Scientific Research National Aeronautics and Space Administration Planetary Science MSFC planetary scientists are active in Peering into the history The centerpiece of the Marshall efforts is research at Marshall, building on a long history of the solar system the Marshall Noble Gas Research Laboratory of scientific support to NASA’s human explo- (MNGRL), a unique facility within NASA. Noble- ration program planning, whether focused Marshall planetary scientists use multiple anal- gas isotopes are a well-established technique on the Moon, asteroids, or Mars. Research ysis techniques to understand the formation, for providing detailed temperature-time histories areas of expertise include planetary sample modification, and age of planetary materials of rocks and meteorites. The MNGRL lab uses analysis, planetary interior modeling, and to learn about their parent planets. Sample Ar-Ar and I-Xe radioactive dating to find the planetary atmosphere observations. Scientists analysis of this type is well-aligned with the formation age of rocks and meteorites, and at Marshall are involved in several ongoing priorities for scientific research and analysis Ar/Kr/Ne cosmic-ray exposure ages to under- planetary science missions, including the Mars in NASA’s Planetary Science Division. Multiple stand when the meteorites were launched from Exploration Rovers, the Cassini mission to future missions are poised to provide new their parent planets. Saturn, and the Gravity Recovery and Interior sample-analysis opportunities. Laboratory (GRAIL) mission to the Moon. Marshall is the center for program manage- ment for the Agency’s Discovery and New Frontiers programs, providing programmatic oversight into a variety of missions to various planetary science destinations throughout the solar system, from MESSENGER’s investiga- tions of Mercury to New Frontiers’ forthcoming examination of the Pluto system. -

Lunar & Planetary Surface Power Management & Distribution
NASA SBIR 2020 Phase I Solicitation Z1.05 Lunar & Planetary Surface Power Management & Distribution Lead Center: GRC Participating Center(s): GSFC, JSC Technology Area: TA3 Space Power and Energy Storage Scope Title Innovative ways to transmit high power for lunar & Mars surface missions Scope Description The Global Exploration Roadmap (January 2018) and the Space Policy Directive (December 2017) detail NASA’s plans for future human-rated space missions. A major factor in this involves establishing bases on the lunar surface and eventually Mars. Surface power for bases is envisioned to be located remotely from the habitat modules and must be efficiently transferred over significant distances. The International Space Station (ISS) has the highest power (100kW), and largest space power distribution system with eight interleaved micro-grids providing power functions similar to a terrestrial power utility. Planetary bases will be similar to the ISS with expectations of multiple power sources, storage, science, and habitation modules, but at higher power levels and with longer distribution networks providing interconnection. In order to enable high power (>100kW) and longer distribution systems on the surface of the moon or Mars, NASA is in need of innovative technologies in the areas of lower mass/higher efficiency power electronic regulators, switchgear, cabling, connectors, wireless sensors, power beaming, power scavenging, and power management control. The technologies of interest would need to operate in extreme temperature environments, including lunar night, and could experience temperature changes from -153C to 123C for lunar applications, and -125C to 80C for Mars bases. In addition to temperature extremes, technologies would need to withstand (have minimal degradation from) lunar dust/regolith, Mars dust storms, and space radiation levels. -

Habitability of Super-Earths: Gliese 581C & 581D
Exoplanets: Detection, Formation and Dynamics Proceedings IAU Symposium No. 249, 2007 c 2008 International Astronomical Union Y.-S. Sun, S. Ferraz-Mello and J.-L. Zhou, eds. doi:10.1017/S1743921308017031 Habitability of super-Earths: Gliese 581c & 581d W. von Bloh1, C. Bounama1,M.Cuntz2 and S. Franck1 1 Potsdam Institute for Climate Impact Research, P.O. Box 601203, Potsdam, Germany, email: [email protected], [email protected], [email protected] 2 University of Texas at Arlington, P.O. Box 19059, Arlington, TX 76019, USA email: [email protected] Abstract. The unexpected diversity of exoplanets includes a growing number of super-Earth planets, i.e., exoplanets with masses smaller than 10 Earth masses. Unlike the larger exoplanets previously found, these smaller planets are more likely to have similar chemical and mineralogical composition to the Earth. We present a thermal evolution model for super-Earth planets to identify the sources and sinks of atmospheric carbon dioxide. The photosynthesis-sustaining habitable zone (pHZ) is determined by the limits of biological productivity on the planetary surface. We apply our model to calculate the habitability of the two super-Earths in the Gliese 581 system. The super-Earth Gl 581c is clearly outside the pHZ, while Gl 581d is at the outer edge of the pHZ. Therefore, it could at least harbor some primitive forms of life. Keywords. Astrobiology, planetary systems, stars: individual (Gliese 581) 1. Introduction Very recently, Udry et al. (2007) announced the detection of two super-Earth planets in the Gliese 581 system; namely, Gl 581c with a mass of 5.06 M⊕ and a semi-major axis of 0.073 AU, and Gl 581d with 8.3 M⊕ and 0.25 AU. -

Sanjay Limaye US Lead-Investigator Ludmila Zasova Russian Lead-Investigator Steering Committee K
Answer to the Call for a Medium-size mission opportunity in ESA’s Science Programme for a launch in 2022 (Cosmic Vision 2015-2025) EuropEan VEnus ExplorEr An in-situ mission to Venus Eric chassEfièrE EVE Principal Investigator IDES, Univ. Paris-Sud Orsay & CNRS Universite Paris-Sud, Orsay colin Wilson Co-Principal Investigator Dept Atm. Ocean. Planet. Phys. Oxford University, Oxford Takeshi imamura Japanese Lead-Investigator sanjay Limaye US Lead-Investigator LudmiLa Zasova Russian Lead-Investigator Steering Committee K. Aplin (UK) S. Lebonnois (France) K. Baines (USA) J. Leitner (Austria) T. Balint (USA) S. Limaye (USA) J. Blamont (France) J. Lopez-Moreno (Spain) E. Chassefière(F rance) B. Marty (France) C. Cochrane (UK) M. Moreira (France) Cs. Ferencz (Hungary) S. Pogrebenko (The Neth.) F. Ferri (Italy) A. Rodin (Russia) M. Gerasimov (Russia) J. Whiteway (Canada) T. Imamura (Japan) C. Wilson (UK) O. Korablev (Russia) L. Zasova (Russia) Sanjay Limaye Ludmilla Zasova Eric Chassefière Takeshi Imamura Colin Wilson University of IKI IDES ISAS/JAXA University of Oxford Wisconsin-Madison Laboratory of Planetary Space Science and Spectroscopy Univ. Paris-Sud Orsay & Engineering Center Space Research Institute CNRS 3-1-1, Yoshinodai, 1225 West Dayton Street Russian Academy of Sciences Universite Paris-Sud, Bat. 504. Sagamihara Dept of Physics Madison, Wisconsin, Profsoyusnaya 84/32 91405 ORSAY Cedex Kanagawa 229-8510 Parks Road 53706, USA Moscow 117997, Russia FRANCE Japan Oxford OX1 3PU Tel +1 608 262 9541 Tel +7-495-333-3466 Tel 33 1 69 15 67 48 Tel +81-42-759-8179 Tel 44 (0)1-865-272-086 Fax +1 608 235 4302 Fax +7-495-333-4455 Fax 33 1 69 15 49 11 Fax +81-42-759-8575 Fax 44 (0)1-865-272-923 [email protected] [email protected] [email protected] [email protected] [email protected] European Venus Explorer – Cosmic Vision 2015 – 2025 List of EVE Co-Investigators NAME AFFILIATION NAME AFFILIATION NAME AFFILIATION AUSTRIA Migliorini, A. -

USGS Open-File Report 2006-1263
Abstracts of the Annual Meeting of Planetary Geologic Mappers, Nampa, Idaho 2006 Edited By Tracy K.P. Gregg,1 Kenneth L. Tanaka,2 and R. Stephen Saunders3 Open-File Report 2006-1263 2006 Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. U.S. DEPARTMENT OF THE INTERIOR U.S. GEOLOGICAL SURVEY 1 The State University of New York at Buffalo, Department of Geology, 710 Natural Sciences Complex, Buffalo, NY 14260-3050. 2 U.S. Geological Survey, 2255 N. Gemini Drive, Flagstaff, AZ 86001. 3 NASA Headquarters, Office of Space Science, 300 E. Street SW, Washington, DC 20546. Report of the Annual Mappers Meeting Northwest Nazarene University Nampa, Idaho June 30 – July 2, 2006 Approximately 18 people attended this year’s mappers meeting, and many more submitted abstracts and maps in absentia. The meeting was held on the campus of Northwest Nazarene University (NNU), and was graciously hosted by NNU’s School of Health and Science. Planetary mapper Dr. Jim Zimbelman is an alumnus of NNU, and he was pivotal in organizing the meeting at this location. Oral and poster presentations were given on Friday, June 30. Drs. Bill Bonnichsen and Marty Godchaux led field excursions on July 1 and 2. USGS Astrogeology Team Chief Scientist Lisa Gaddis led the meeting with a brief discussion of the status of the planetary mapping program at USGS, and a more detailed description of the Lunar Mapping Program. She indicated that there is now a functioning website (http://astrogeology.usgs.gov/Projects/PlanetaryMapping/Lunar/) which shows which lunar quadrangles are available to be mapped.