Drug-Induced Gingival Hyperplasia: a Retrospective Study Using
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
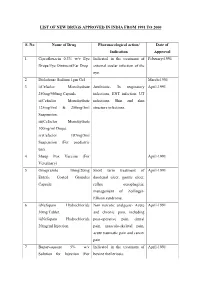
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

Parenteral Dilantin® (Phenytoin Sodium Injection, USP) WARNING
1 Parenteral Dilantin (Phenytoin Sodium Injection, USP) WARNING: CARDIOVASCULAR RISK ASSOCIATED WITH RAPID INFUSION The rate of intravenous Dilantin administration should not exceed 50 mg per minute in adults and 1-3 mg/kg/min (or 50 mg per minute, whichever is slower) in pediatric patients because of the risk of severe hypotension and cardiac arrhythmias. Careful cardiac monitoring is needed during and after administering intravenous Dilantin. Although the risk of cardiovascular toxicity increases with infusion rates above the recommended infusion rate, these events have also been reported at or below the recommended infusion rate. Reduction in rate of administration or discontinuation of dosing may be needed (see WARNINGS and DOSAGE AND ADMINISTRATION). DESCRIPTION Dilantin (phenytoin sodium injection, USP) is a ready-mixed solution of phenytoin sodium in a vehicle containing 40% propylene glycol and 10% alcohol in water for injection, adjusted to pH 12 with sodium hydroxide. Phenytoin sodium is related to the barbiturates in chemical structure, but has a five-membered ring. The chemical name is sodium 5,5-diphenyl-2, 4- imidazolidinedione represented by the following structural formula: CLINICAL PHARMACOLOGY Mechanism of Action Phenytoin is an anticonvulsant which may be useful in the treatment of generalized tonic clonic status epilepticus. The primary site of action appears to be the motor cortex where spread of seizure activity is inhibited. Possibly by promoting sodium efflux from neurons, phenytoin tends to stabilize the threshold against hyperexcitability caused by excessive stimulation or environmental changes capable of reducing membrane sodium gradient. This includes the reduction of posttetanic potentiation at synapses. Loss of posttetanic potentiation prevents cortical seizure foci from detonating adjacent cortical areas. -

Dilantin (Phenytoin Sodium) Extended Oral Capsule Three Times Daily and the Dosage Then Adjusted to Suit Individual Requirements
Dilantin® (Phenytoin Sodium) 100 mg Extended Oral Capsule DESCRIPTION Phenytoin sodium is an antiepileptic drug. Phenytoin sodium is related to the barbiturates in chemical structure, but has a five-membered ring. The chemical name is sodium 5,5-diphenyl-2, 4-imidazolidinedione, having the following structural formula: Each Dilantin— 100 mg Extended Oral Capsule—contains 100 mg phenytoin sodium. Also contains lactose monohydrate, NF; confectioner’s sugar, NF; talc, USP; and magnesium stearate, NF. The capsule body contains titanium dioxide, USP and gelatin, NF. The capsule cap contains FD&C red No. 28; FD&C yellow No. 6; and gelatin NF. Product in vivo performance is characterized by a slow and extended rate of absorption with peak blood concentrations expected in 4 to 12 hours as contrasted to Prompt Phenytoin Sodium Capsules, USP with a rapid rate of absorption with peak blood concentration expected in 1½ to 3 hours. CLINICAL PHARMACOLOGY Phenytoin is an antiepileptic drug which can be used in the treatment of epilepsy. The primary site of action appears to be the motor cortex where spread of seizure activity is inhibited. Possibly by promoting sodium efflux from neurons, phenytoin tends to stabilize the threshold against hyperexcitability caused by excessive stimulation or environmental changes capable of reducing membrane sodium gradient. This includes the reduction of posttetanic potentiation at synapses. Loss of posttetanic potentiation prevents cortical seizure foci from detonating adjacent cortical areas. Phenytoin reduces the maximal activity of brain stem centers responsible for the tonic phase of tonic-clonic (grand mal) seizures. The plasma half-life in man after oral administration of phenytoin averages 22 hours, with a range of 7 to 42 hours. -

Association of Hypertensive Status and Its Drug Treatment with Lipid and Haemostatic Factors in Middle-Aged Men: the PRIME Study
Journal of Human Hypertension (2000) 14, 511–518 2000 Macmillan Publishers Ltd All rights reserved 0950-9240/00 $15.00 www.nature.com/jhh ORIGINAL ARTICLE Association of hypertensive status and its drug treatment with lipid and haemostatic factors in middle-aged men: the PRIME Study P Marques-Vidal1, M Montaye2, B Haas3, A Bingham4, A Evans5, I Juhan-Vague6, J Ferrie`res1, G Luc2, P Amouyel2, D Arveiler3, D McMaster5, JB Ruidavets1, J-M Bard2, PY Scarabin4 and P Ducimetie`re4 1INSERM U518, Faculte´ de Me´decine Purpan, Toulouse, France; 2MONICA-Lille, Institut Pasteur de Lille, Lille, France; 3MONICA-Strasbourg, Laboratoire d’Epide´miologie et de Sante´ Publique, Strasbourg, France; 4INSERM U258, Hoˆ pital Broussais, Paris, France; 5Belfast-MONICA, Department of Epidemiology, The Queen’s University of Belfast, UK; 6Laboratory of Haematology, La Timone Hospital, Marseille, France Aims: To assess the association of hypertensive status this effect remained after multivariate adjustment. Cal- and antihypertensive drug treatment with lipid and hae- cium channel blockers decreased total cholesterol and mostatic levels in middle-aged men. apoproteins A-I and B; those differences remained sig- Methods and results: Hypertensive status, antihyperten- nificant after multivariate adjustment. ACE inhibitors sive drug treatment, total and high-density lipoprotein decreased total cholesterol, triglycerides, apoprotein B (HDL) cholesterol, triglyceride, apoproteins A-I and B, and LpE:B; and this effect remained after multivariate lipoparticles LpA-I, -

Dorset Medicines Advisory Group
DORSET CARDIOLOGY WORKING GROUP GUIDELINE FOR CALCIUM CHANNEL BLOCKERS IN HYPERTENSION SUMMARY The pan-Dorset cardiology working group continues to recommend the use of amlodipine (a third generation dihydropyridine calcium-channel blocker) as first choice calcium channel blocker on the pan-Dorset formulary for hypertension. Lercanidipine is second choice, lacidipine third choice and felodipine is fourth choice. This is due to preferable side effect profiles in terms of ankle oedema and relative costs of the preparations. Note: where angina is the primary indication or is a co-morbidity prescribers must check against the specific product characteristics (SPC) for an individual drug to confirm this is a licensed indication. N.B. Lacidipine and lercandipine are only licensed for use in hypertension. Chapter 02.06.02 CCBs section of the Formulary has undergone an evidence-based review. A comprehensive literature search was carried out on NHS Evidence, Medline, EMBASE, Cochrane Database, and UK Duets. This was for recent reviews or meta-analyses on calcium channel blockers from 2009 onwards (comparative efficacy and side effects) and randomised controlled trials (RCTs). REVIEW BACKGROUND Very little good quality evidence exists. No reviews, meta-analyses or RCTs were found covering all calcium channel blockers currently on the formulary. Another limitation was difficulty obtaining full text original papers for some of the references therefore having to use those from more obscure journals instead. Some discrepancies exist between classification of generations of dihydropyridine CCBs, depending upon the year of publication of the reference/authors’ interpretation. Dihydropyridine (DHP) CCBs tend to be more potent vasodilators than non-dihydropyridine (non-DHP) CCBs (diltiazem, verapamil), but the latter have greater inotropic effects. -

Potentially Harmful Drugs in the Elderly: Beers List
−This Clinical Resource gives subscribers additional insight related to the Recommendations published in− March 2019 ~ Resource #350301 Potentially Harmful Drugs in the Elderly: Beers List In 1991, Dr. Mark Beers and colleagues published a methods paper describing the development of a consensus list of medicines considered to be inappropriate for long-term care facility residents.12 The “Beers list” is now in its sixth permutation.1 It is intended for use by clinicians in outpatient as well as inpatient settings (but not hospice or palliative care) to improve the care of patients 65 years of age and older.1 It includes medications that should generally be avoided in all elderly, used with caution, or used with caution or avoided in certain elderly.1 There is also a list of potentially harmful drug-drug interactions in seniors, as well as a list of medications that may need to be avoided or have their dosage reduced based on renal function.1 This information is not comprehensive; medications and interactions were chosen for inclusion based on potential harm in relation to benefit in the elderly, and availability of alternatives with a more favorable risk/benefit ratio.1 The criteria no longer address drugs to avoid in patients with seizures or insomnia because these concerns are not unique to the elderly.1 Another notable deletion is H2 blockers as a concern in dementia; evidence of cognitive impairment is weak, and long-term PPIs pose risks.1 Glimepiride has been added as a drug to avoid. Some drugs have been added with cautions (dextromethorphan/quinidine, trimethoprim/sulfamethoxazole), and some have had cautions added (rivaroxaban, tramadol, SNRIs). -

Effect of Amlodipine and Indomethacin in Electrical and Picrotoxin Induced Convulsions in Mice
DOI: 10.5958/2319-5886.2014.00402.0 International Journal of Medical Research & Health Sciences www.ijmrhs.com Volume 3 Issue 3 Coden: IJMRHS Copyright @2014 ISSN: 2319-5886 Received: 9th Apr 2014 Revised: 3rd Jun 2014 Accepted: 14th Jun 2014 Research Article EFFECT OF AMLODIPINE AND INDOMETHACIN IN ELECTRICAL AND PICROTOXIN INDUCED CONVULSIONS IN MICE *Jagathi Devi N1, Prasanna V2 1Assistant Professor, 2Professor and Head, Department of Pharmacology, Osmania Medical College, Hyderabad *Corresponding author email: [email protected] ABSTRACT Background and Objectives: Antiepileptic drugs (AEDs) are the drugs used in the treatment of epilepsy. Many AEDs have been developed, but the ideal AED which can not only prevent but also abolish seizures by correcting the underlying pathophysiology is still not in sight. Calcium channel blockers (CCBs) may form such a group, as the initiation of epileptogenic activity in the neuron is connected with a phenomenon known as “intrinsic burst firing” which is activated by inward calcium current. In this study, Amlodipine, a CCB of the dihydropyridine class was evaluated for its anticonvulsant activity in mice. It was compared with Phenytoin sodium, one of the oldest anti epileptic drugs. Amlodipine was also combined with Indomethacin, a conventional NSAID, to look for any potentiating effect of this prostaglandin-synthesis inhibitor. Materials and Methods: A total of 48 adult Swiss albino mice of either sex weighing 20-30 G were used for this study; 48 were divided into 8 groups, each group containing 6 mice. Group 1-4 MES (50 m Amp for 0.1 secs) induced convulsion method, Group 5-8 evaluated by using the chemo-convulsant, picrotoxin (0.7 mg / kg). -

Drug Name Plate Number Well Location % Inhibition, Screen Axitinib 1 1 20 Gefitinib (ZD1839) 1 2 70 Sorafenib Tosylate 1 3 21 Cr
Drug Name Plate Number Well Location % Inhibition, Screen Axitinib 1 1 20 Gefitinib (ZD1839) 1 2 70 Sorafenib Tosylate 1 3 21 Crizotinib (PF-02341066) 1 4 55 Docetaxel 1 5 98 Anastrozole 1 6 25 Cladribine 1 7 23 Methotrexate 1 8 -187 Letrozole 1 9 65 Entecavir Hydrate 1 10 48 Roxadustat (FG-4592) 1 11 19 Imatinib Mesylate (STI571) 1 12 0 Sunitinib Malate 1 13 34 Vismodegib (GDC-0449) 1 14 64 Paclitaxel 1 15 89 Aprepitant 1 16 94 Decitabine 1 17 -79 Bendamustine HCl 1 18 19 Temozolomide 1 19 -111 Nepafenac 1 20 24 Nintedanib (BIBF 1120) 1 21 -43 Lapatinib (GW-572016) Ditosylate 1 22 88 Temsirolimus (CCI-779, NSC 683864) 1 23 96 Belinostat (PXD101) 1 24 46 Capecitabine 1 25 19 Bicalutamide 1 26 83 Dutasteride 1 27 68 Epirubicin HCl 1 28 -59 Tamoxifen 1 29 30 Rufinamide 1 30 96 Afatinib (BIBW2992) 1 31 -54 Lenalidomide (CC-5013) 1 32 19 Vorinostat (SAHA, MK0683) 1 33 38 Rucaparib (AG-014699,PF-01367338) phosphate1 34 14 Lenvatinib (E7080) 1 35 80 Fulvestrant 1 36 76 Melatonin 1 37 15 Etoposide 1 38 -69 Vincristine sulfate 1 39 61 Posaconazole 1 40 97 Bortezomib (PS-341) 1 41 71 Panobinostat (LBH589) 1 42 41 Entinostat (MS-275) 1 43 26 Cabozantinib (XL184, BMS-907351) 1 44 79 Valproic acid sodium salt (Sodium valproate) 1 45 7 Raltitrexed 1 46 39 Bisoprolol fumarate 1 47 -23 Raloxifene HCl 1 48 97 Agomelatine 1 49 35 Prasugrel 1 50 -24 Bosutinib (SKI-606) 1 51 85 Nilotinib (AMN-107) 1 52 99 Enzastaurin (LY317615) 1 53 -12 Everolimus (RAD001) 1 54 94 Regorafenib (BAY 73-4506) 1 55 24 Thalidomide 1 56 40 Tivozanib (AV-951) 1 57 86 Fludarabine -

Neurochemical Mechanisms Underlying Alcohol Withdrawal
Neurochemical Mechanisms Underlying Alcohol Withdrawal John Littleton, MD, Ph.D. More than 50 years ago, C.K. Himmelsbach first suggested that physiological mechanisms responsible for maintaining a stable state of equilibrium (i.e., homeostasis) in the patient’s body and brain are responsible for drug tolerance and the drug withdrawal syndrome. In the latter case, he suggested that the absence of the drug leaves these same homeostatic mechanisms exposed, leading to the withdrawal syndrome. This theory provides the framework for a majority of neurochemical investigations of the adaptations that occur in alcohol dependence and how these adaptations may precipitate withdrawal. This article examines the Himmelsbach theory and its application to alcohol withdrawal; reviews the animal models being used to study withdrawal; and looks at the postulated neuroadaptations in three systems—the gamma-aminobutyric acid (GABA) neurotransmitter system, the glutamate neurotransmitter system, and the calcium channel system that regulates various processes inside neurons. The role of these neuroadaptations in withdrawal and the clinical implications of this research also are considered. KEY WORDS: AOD withdrawal syndrome; neurochemistry; biochemical mechanism; AOD tolerance; brain; homeostasis; biological AOD dependence; biological AOD use; disorder theory; biological adaptation; animal model; GABA receptors; glutamate receptors; calcium channel; proteins; detoxification; brain damage; disease severity; AODD (alcohol and other drug dependence) relapse; literature review uring the past 25 years research- science models used to study with- of the reasons why advances in basic ers have made rapid progress drawal neurochemistry as well as a research have not yet been translated Din understanding the chemi- reluctance on the part of clinicians to into therapeutic gains and suggests cal activities that occur in the nervous consider new treatments. -

Management of Alcohol Withdrawal
Management of alcohol withdrawal Q2: What interventions are safe and effective for the management of alcohol withdrawal, including treatment for alcohol withdrawal seizures and prevention and treatment for acute Wernicke's encephalopathy? Background Alcohol withdrawal can be uncomfortable and occasionally life threatening. Pharmacological management of alcohol withdrawal is an essential component of alcohol dependence. Benzodiazepines (BZDs), non-sedating anticonvulsants and antipsychotics are commonly used in the treatment of alcohol withdrawal. Given that they are all potentially toxic medications, what is the evidence that the benefits of their use justify the risks? Which is more effective? Population/Intervention(s)/Comparison/Outcome(s) (PICO) Population: people with alcohol dependence commencing alcohol withdrawal Interventions: benzodiazepines anticonvulsants (non sedating i.e. non barbiturates and not chlormethiazole) antipsychotics Comparison: placebo and/or active treatment Outcomes: severity of withdrawal complications of withdrawal (seizures, delirium) completion of withdrawal death 1 Management of alcohol withdrawal List of the systematic reviews identified by the search process INCLUDED IN GRADE TABLES OR FOOTNOTES Ntais C et al (2005). Benzodiazepines for alcohol withdrawal. Cochrane Database of Systematic Review, (3):CD005063. Polycarpou A et al (2005). Anticonvulsants for alcohol withdrawal. Cochrane Database of Systematic Reviews, (3):CD005064. Mayo-Smith MF (1997). Pharmacological management of alcohol withdrawal. A -

Medicine for Prevention of and Treatment for Arteriosclerosis and Hypertension
Europäisches Patentamt *EP001604664A1* (19) European Patent Office Office européen des brevets (11) EP 1 604 664 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 158(3) EPC (43) Date of publication: (51) Int Cl.7: A61K 31/4422, A61K 45/06, 14.12.2005 Bulletin 2005/50 A61P 9/00, A61P 9/10, A61P 43/00, A61P 9/12, (21) Application number: 04706359.9 A61P 13/12 (22) Date of filing: 29.01.2004 (86) International application number: PCT/JP2004/000861 (87) International publication number: WO 2004/067003 (12.08.2004 Gazette 2004/33) (84) Designated Contracting States: • IWAI, Masaru AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Shigenobu-cho, Onsen-gun, HU IE IT LI LU MC NL PT RO SE SI SK TR Ehime 791-0204 (JP) Designated Extension States: • SADA, Toshio, Sankyo Company, Limited AL LT LV MK Tokyo 140-8710 (JP) • MIZUNO, Makoto, Sankyo Company, Limited (30) Priority: 31.01.2003 JP 2003022990 Tokyo 140-8710 (JP) 07.02.2003 JP 2003030830 (74) Representative: Gibson, Christian John Robert (71) Applicant: Sankyo Company, Limited Marks & Clerk Tokyo 103-8426 (JP) 90 Long Acre London WC2E 9RA (GB) (72) Inventors: • HORIUCHI, Masatsugu Onsen-gun, Ehime 791-0204 (JP) (54) MEDICINE FOR PREVENTION OF AND TREATMENT FOR ARTERIOSCLEROSIS AND HYPERTENSION (57) A medicament comprising the following composition: (A) an angiotensin II receptor antagonist selected from the group of a compound having a general formula (I), pharmacologically acceptable esters thereof and pharmacolog- ically acceptable salts thereof (for example, olmesartan medoxomil and the like); EP 1 604 664 A1 Printed by Jouve, 75001 PARIS (FR) (Cont. -

Guideline for Preoperative Medication Management
Guideline: Preoperative Medication Management Guideline for Preoperative Medication Management Purpose of Guideline: To provide guidance to physicians, advanced practice providers (APPs), pharmacists, and nurses regarding medication management in the preoperative setting. Background: Appropriate perioperative medication management is essential to ensure positive surgical outcomes and prevent medication misadventures.1 Results from a prospective analysis of 1,025 patients admitted to a general surgical unit concluded that patients on at least one medication for a chronic disease are 2.7 times more likely to experience surgical complications compared with those not taking any medications. As the aging population requires more medication use and the availability of various nonprescription medications continues to increase, so does the risk of polypharmacy and the need for perioperative medication guidance.2 There are no well-designed trials to support evidence-based recommendations for perioperative medication management; however, general principles and best practice approaches are available. General considerations for perioperative medication management include a thorough medication history, understanding of the medication pharmacokinetics and potential for withdrawal symptoms, understanding the risks associated with the surgical procedure and the risks of medication discontinuation based on the intended indication. Clinical judgement must be exercised, especially if medication pharmacokinetics are not predictable or there are significant risks associated with inappropriate medication withdrawal (eg, tolerance) or continuation (eg, postsurgical infection).2 Clinical Assessment: Prior to instructing the patient on preoperative medication management, completion of a thorough medication history is recommended – including all information on prescription medications, over-the-counter medications, “as needed” medications, vitamins, supplements, and herbal medications. Allergies should also be verified and documented.