Functional Characterization of Arabidopsis Sorting Nexin Atsnx2b Nguyen Phan Iowa State University
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Trafficking Routes to the Plant Vacuole: Connecting Alternative and Classical Pathways
This is a repository copy of Trafficking routes to the plant vacuole: connecting alternative and classical pathways. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/124374/ Version: Accepted Version Article: Di Sansebastiano, GP, Barozzi, F, Piro, G et al. (2 more authors) (2018) Trafficking routes to the plant vacuole: connecting alternative and classical pathways. Journal of Experimental Botany, 69 (1). pp. 79-90. ISSN 0022-0957 https://doi.org/10.1093/jxb/erx376 © The Author(s) 2017. Published by Oxford University Press on behalf of the Society for Experimental Biology. This is an author produced version of a paper published in Journal of Experimental Botany. Uploaded in accordance with the publisher's self-archiving policy. Reuse Items deposited in White Rose Research Online are protected by copyright, with all rights reserved unless indicated otherwise. They may be downloaded and/or printed for private study, or other acts as permitted by national copyright laws. The publisher or other rights holders may allow further reproduction and re-use of the full text version. This is indicated by the licence information on the White Rose Research Online record for the item. Takedown If you consider content in White Rose Research Online to be in breach of UK law, please notify us by emailing [email protected] including the URL of the record and the reason for the withdrawal request. [email protected] https://eprints.whiterose.ac.uk/ 1 Trafficking routes to the Plant Vacuole: 2 connecting alternative and classical pathways. 3 4 Gian Pietro Di Sansebastiano1*, Fabrizio Barozzi1, Gabriella Piro1, Jurgen Denecke2 and 5 Carine de Marcos Lousa2,3 * 6 1.! DiSTeBA (Dipartimento di Scienze e Tecnologie Biologiche ed Ambientali), 7 University of Salento, Campus ECOTEKNE, 73100 Lecce, Italy; 8 [email protected] and [email protected]. -

Antibodies to Snare Complex Proteins
ANTIBODIES TO SNARE COMPLEX PROTEINS Antibodies to SNARE Complex Proteins Synaptophysin VAMP/Synaptobrevin SNAP-25 Syntaxin CDCrel-1 Synaptotagmin-1 Munc18-1 Synapsin-1 INTERNATIONAL VERSION www.cedarlanelabs.com/SNARE CEDARLANE® is an ISO 9001 and ISO 13485 registered company Antibodies to SNARE Complex Proteins SNARE proteins are a large protein superfamily consisting of more than 60 members in yeast and mammals. The primary role of these proteins is to mediate fusion of vesicles with their target membrane-bound compartments (such as lysosomes). The most well studied SNARE proteins are those involved in mediating synaptic vesicle docking at the pre-synaptic membrane of neurons. During this process, syntaxin-1, SNAP-25 and munc18-1 associate and form a complex at the pre-synaptic membrane. This complex interacts with synaptobrevin-2 and synaptotagmin-1 located in synaptic vesicles and initiates docking, priming and fusion at the membrane. This fusion event leads to release of the vesicle's cargo into the synaptic cleft, where it can ultimately interact with the post-synaptic neuron. Antibodies to: Synaptophysin Synaptophysin is a 38 KDa synaptic vesicle (SV) glycoprotein containing four transmembrane domains. It is present in SVs of the neuroendocrine system, brain, spinal cord, retina, adrenal medulla and at neuromuscular junctions. Synaptophysin acts as a marker for neuroendocrine tumours and has been used to study the distribution of synapses within the brain due to its ubiquity at these regions. Although the exact function of synaptophysin is still unknown, several lines of evidence suggest it may have many important roles in SV exo and endocytosis. These include regulation of SNARE assembly, fusion pore formation initiating neurotransmitter release, and activation of SV endocytosis. -

Sorting Nexins in Protein Homeostasis Sara E. Hanley1,And Katrina F
Preprints (www.preprints.org) | NOT PEER-REVIEWED | Posted: 6 November 2020 doi:10.20944/preprints202011.0241.v1 Sorting nexins in protein homeostasis Sara E. Hanley1,and Katrina F. Cooper2* 1Department of Molecular Biology, Graduate School of Biomedical Sciences, Rowan University, Stratford, NJ, 08084, USA 1 [email protected] 2 [email protected] * [email protected] Tel: +1 (856)-566-2887 1Department of Molecular Biology, Graduate School of Biomedical Sciences, Rowan University, Stratford, NJ, 08084, USA Abstract: Sorting nexins (SNXs) are a highly conserved membrane-associated protein family that plays a role in regulating protein homeostasis. This family of proteins is unified by their characteristic phox (PX) phosphoinositides binding domain. Along with binding to membranes, this family of SNXs also comprises a diverse array of protein-protein interaction motifs that are required for cellular sorting and protein trafficking. SNXs play a role in maintaining the integrity of the proteome which is essential for regulating multiple fundamental processes such as cell cycle progression, transcription, metabolism, and stress response. To tightly regulate these processes proteins must be expressed and degraded in the correct location and at the correct time. The cell employs several proteolysis mechanisms to ensure that proteins are selectively degraded at the appropriate spatiotemporal conditions. SNXs play a role in ubiquitin-mediated protein homeostasis at multiple levels including cargo localization, recycling, degradation, and function. In this review, we will discuss the role of SNXs in three different protein homeostasis systems: endocytosis lysosomal, the ubiquitin-proteasomal, and the autophagy-lysosomal system. The highly conserved nature of this protein family by beginning with the early research on SNXs and protein trafficking in yeast and lead into their important roles in mammalian systems. -

Structural and Functional Insights Into Sorting Nexin 5/6 Interaction with Bacterial Effector Ince
OPEN Citation: Signal Transduction and Targeted Therapy (2017) 2, e17030; doi:10.1038/sigtrans.2017.30 www.nature.com/sigtrans ARTICLE Structural and functional insights into sorting nexin 5/6 interaction with bacterial effector IncE Qingxiang Sun1,5, Xin Yong1,2,5, Xiaodong Sun3,5, Fan Yang1,2,5, Zhonghua Dai4, Yanqiu Gong1, Liming Zhou3, Xia Zhang1, Dawen Niu1, Lunzhi Dai1, Jia-Jia Liu4 and Da Jia1,2 The endosomal trafficking pathways are essential for many cellular activities. They are also important targets by many intracellular pathogens. Key regulators of the endosomal trafficking include the retromer complex and sorting nexins (SNXs). Chlamydia trachomatis effector protein IncE directly targets the retromer components SNX5 and SNX6 and suppresses retromer-mediated transport, but the exact mechanism has remained unclear. We present the crystal structure of the PX domain of SNX5 in complex with IncE, showing that IncE binds to a highly conserved hydrophobic groove of SNX5. The unique helical hairpin of SNX5/6 is essential for binding, explaining the specificity of SNX5/6 for IncE. The SNX5/6–IncE interaction is required for cellular localization of IncE and its inhibitory function. Mechanistically, IncE inhibits the association of CI-MPR cargo with retromer-containing endosomal subdomains. Our study provides new insights into the regulation of retromer-mediated transport and illustrates the intricate competition between host and pathogens in controlling cellular trafficking. Signal Transduction and Targeted Therapy (2017) 2, e17030; doi:10.1038/sigtrans.2017.30; -

Direct Lysosome-Based Autophagy of Lipid Droplets in Hepatocytes
Direct lysosome-based autophagy of lipid droplets in hepatocytes Ryan J. Schulzea,b,1, Eugene W. Kruegera,b,1, Shaun G. Wellera,b, Katherine M. Johnsona,b, Carol A. Caseyc, Micah B. Schotta,b, and Mark A. McNivena,b,2 aDepartment of Biochemistry and Molecular Biology, Mayo Clinic, Rochester, MN 55905; bDivision of Gastroenterology and Hepatology, Mayo Clinic, Rochester, MN 55905; and cDepartment of Internal Medicine, University of Nebraska Medical Center, Omaha, NE 68198 Edited by Tobias C. Walther, Harvard School of Public Health, Boston, MA, and accepted by Editorial Board Member Joseph L. Goldstein October 13, 2020 (received for review June 4, 2020) Hepatocytes metabolize energy-rich cytoplasmic lipid droplets (LDs) process that appears to play an especially important role in the in the lysosome-directed process of autophagy. An organelle- degradation of hepatocellular LDs (15). A selective form of LD- selective form of this process (macrolipophagy) results in the engulf- centric autophagy known as lipophagy is thought to involve the ment of LDs within double-membrane delimited structures (auto- recognition of as-of-yet unidentified LD-specific receptors to phagosomes) before lysosomal fusion. Whether this is an promote the localized assembly and extension of a sequestering exclusive autophagic mechanism used by hepatocytes to catabolize phagophore around the perimeter of the LD (16, 17). How this LDs is unclear. It is also unknown whether lysosomes alone might be phagophore is targeted to (and extended around) the LD surface to sufficient to mediate LD turnover in the absence of an autophago- facilitate lipophagy remains unclear. Once fully enclosed, the somal intermediate. -

Defining the Kv2.1–Syntaxin Molecular Interaction Identifies a First-In-Class Small Molecule Neuroprotectant
Defining the Kv2.1–syntaxin molecular interaction identifies a first-in-class small molecule neuroprotectant Chung-Yang Yeha,b,1, Zhaofeng Yec,d,1, Aubin Moutale, Shivani Gaura,b, Amanda M. Hentonf,g, Stylianos Kouvarosf,g, Jami L. Salomana, Karen A. Hartnett-Scotta,b, Thanos Tzounopoulosa,f,g, Rajesh Khannae, Elias Aizenmana,b,g,2, and Carlos J. Camachoc,2 aDepartment of Neurobiology, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261; bPittsburgh Institute for Neurodegenerative Diseases, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261; cDepartment of Computational and Systems Biology, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261; dSchool of Medicine, Tsinghua University, Beijing 100871, China; eDepartment of Pharmacology, College of Medicine, University of Arizona, Tucson, AZ 85724; fDepartment of Otolaryngology, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261; and gPittsburgh Hearing Research Center, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261 Edited by Lily Yeh Jan, University of California, San Francisco, CA, and approved June 19, 2019 (received for review February 27, 2019) + The neuronal cell death-promoting loss of cytoplasmic K follow- (13). The Kv2.1-dependent cell death pathway is normally initiated ing injury is mediated by an increase in Kv2.1 potassium channels in by the oxidative liberation of zinc from intracellular metal-binding the plasma membrane. This phenomenon relies on Kv2.1 binding to proteins (14), leading to the sequential phosphorylation of syntaxin 1A via 9 amino acids within the channel intrinsically disor- Kv2.1 residues Y124 and S800 by Src and p38 kinases, respectively dered C terminus. Preventing this interaction with a cell and blood- (15–17). -

Identification of Synaptic Proteins and Their Isoform Mrnas In
Proc. Natl. Acad. Sci. USA Vol. 91, pp. 12487-12491, December 1994 Cell Biology Identification of synaptic proteins and their isoform mRNAs in compartments of pancreatic endocrine cells (exocytosis/secretion/insulin/diabetes) GUNILLA JACOBSSON*, ANDREW J. BEANt, RICHARD H. SCHELLERt, LISA JUNTTI-BERGGRENt, JUDE T. DEENEYt, PER-OLOF BERGGRENt AND BJORN MEISTER*§ *Department of Neuroscience and tRolf Luft's Center for Diabetes Research, Department of Molecular Medicine, Karolinska Institute, S-171 77 Stockholm, Sweden; and tDepartment of Molecular and Cellular Physiology, Howard Hughes Medical Institute, Beckman Center, Stanford University, Stanford, CA 94305 Communicated by Tomas Hokfelt, August 30, 1994 ABSTRACT Several proteins that are of importance for clostridial neurotoxins, including tetanus toxin and botuli- membrane trafficking in the nerve terminal have recently been num neurotoxin B, whereas botulinum neurotoxins D and F characterized. We have used Western blot and immunohis- are capable of cleaving both forms of VAMP (10-12). tochemistry to show that synaptotagmin, synaptobrevin/VAMP VAMP-1 and VAMP-2 are encoded by two distinct genes (13) (vesicle-associated membrane protein), SNAP-25 (synaptosom- and are differentially expressed in the nervous system (14). al-associated protein of 25 kDa), and syntaxin proteins are Cellubrevin is a homologue of VAMP, which is present in a present in cells of the islets of Langerhans in the endocrine wide variety of tissues and may be a membrane trafficking pancreas. Synaptotagmin-like immunoreactivity (-LI) was lo- protein of a constitutively recycling pathway (15). calized to granules within the cytoplasm of a few endocrine cells In contrast to synaptotagmin and VAMP, the synaptoso- located in the periphery of the islets, identified as somatostatin- mal-associated protein of 25 kDa (SNAP-25) is located at the containing cells, and in many nerve fibers within the islets. -

Sorting Nexin 27 Regulates the Lysosomal Degradation of Aquaporin-2 Protein in the Kidney Collecting Duct
cells Article Sorting Nexin 27 Regulates the Lysosomal Degradation of Aquaporin-2 Protein in the Kidney Collecting Duct Hyo-Jung Choi 1,2, Hyo-Ju Jang 1,3, Euijung Park 1,3, Stine Julie Tingskov 4, Rikke Nørregaard 4, Hyun Jun Jung 5 and Tae-Hwan Kwon 1,3,* 1 Department of Biochemistry and Cell Biology, School of Medicine, Kyungpook National University, Taegu 41944, Korea; [email protected] (H.-J.C.); [email protected] (H.-J.J.); [email protected] (E.P.) 2 New Drug Development Center, Daegu-Gyeongbuk Medical Innovation Foundation, Taegu 41061, Korea 3 BK21 Plus KNU Biomedical Convergence Program, Department of Biomedical Science, School of Medicine, Kyungpook National University, Taegu 41944, Korea 4 Department of Clinical Medicine, Aarhus University, Aarhus 8200, Denmark; [email protected] (S.J.T.); [email protected] (R.N.) 5 Division of Nephrology, Department of Medicine, Johns Hopkins University School of Medicine, Baltimore, MD 21205, USA; [email protected] * Correspondence: [email protected]; Tel.: +82-53-420-4825; Fax: +82-53-422-1466 Received: 30 March 2020; Accepted: 11 May 2020; Published: 13 May 2020 Abstract: Sorting nexin 27 (SNX27), a PDZ (Postsynaptic density-95/Discs large/Zonula occludens 1) domain-containing protein, cooperates with a retromer complex, which regulates intracellular trafficking and the abundance of membrane proteins. Since the carboxyl terminus of aquaporin-2 (AQP2c) has a class I PDZ-interacting motif (X-T/S-X-F), the role of SNX27 in the regulation of AQP2 was studied. Co-immunoprecipitation assay of the rat kidney demonstrated an interaction of SNX27 with AQP2. -
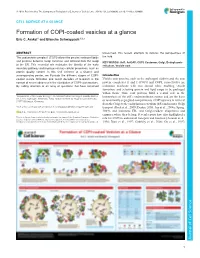
Formation of COPI-Coated Vesicles at a Glance Eric C
© 2018. Published by The Company of Biologists Ltd | Journal of Cell Science (2018) 131, jcs209890. doi:10.1242/jcs.209890 CELL SCIENCE AT A GLANCE Formation of COPI-coated vesicles at a glance Eric C. Arakel1 and Blanche Schwappach1,2,* ABSTRACT unresolved, this review attempts to refocus the perspectives of The coat protein complex I (COPI) allows the precise sorting of lipids the field. and proteins between Golgi cisternae and retrieval from the Golgi KEY WORDS: Arf1, ArfGAP, COPI, Coatomer, Golgi, Endoplasmic to the ER. This essential role maintains the identity of the early reticulum, Vesicle coat secretory pathway and impinges on key cellular processes, such as protein quality control. In this Cell Science at a Glance and accompanying poster, we illustrate the different stages of COPI- Introduction coated vesicle formation and revisit decades of research in the Vesicle coat proteins, such as the archetypal clathrin and the coat context of recent advances in the elucidation of COPI coat structure. protein complexes II and I (COPII and COPI, respectively) are By calling attention to an array of questions that have remained molecular machines with two central roles: enabling vesicle formation, and selecting protein and lipid cargo to be packaged within them. Thus, coat proteins fulfil a central role in the 1Department of Molecular Biology, Universitätsmedizin Göttingen, Humboldtallee homeostasis of the cell’s endomembrane system and are the basis 23, 37073 Göttingen, Germany. 2Max-Planck Institute for Biophysical Chemistry, 37077 Göttingen, Germany. of functionally segregated compartments. COPI operates in retrieval from the Golgi to the endoplasmic reticulum (ER) and in intra-Golgi *Author for correspondence ([email protected]) transport (Beck et al., 2009; Duden, 2003; Lee et al., 2004a; Spang, E.C.A., 0000-0001-7716-7149; B.S., 0000-0003-0225-6432 2009), and maintains ER- and Golgi-resident chaperones and enzymes where they belong. -

Molecular Mechanism of Membrane Targeting by the GRP1 PH Domain
Supplemental Material can be found at: http://www.jlr.org/cgi/content/full/M800150-JLR200/DC1 Molecular mechanism of membrane targeting by the GRP1 PH domain † † † Ju He,* Rachel M. Haney, ,§ Mohsin Vora, Vladislav V. Verkhusha,** Robert V. Stahelin, ,§ and Tatiana G. Kutateladze1,* Department of Pharmacology,* University of Colorado Health Sciences Center, Aurora, CO; † Department of Biochemistry and Molecular Biology, Indiana University School of Medicine, South Bend, IN; Department of Chemistry and Biochemistry and The Walther Center for Cancer Research,§ University of Notre Dame, South Bend, IN; and Department of Anatomy and Structural Biology,** Downloaded from Albert Einstein College of Medicine, Bronx, NY Abstract The general receptor for phosphoinositides iso- Supplementary key words general receptor for phosphoinositides iso- • • • form 1 (GRP1) is recruited to the plasma membrane in re- form 1 pleckstrin homology domain phosphoinositide phosphati- dylinositol 3,4,5-trisphosphate sponse to activation of phosphoinositide 3-kinases and www.jlr.org accumulation of phosphatidylinositol 3,4,5-trisphosphate ʼ [PtdIns(3,4,5)P3]. GRP1 s pleckstrin homology (PH) do- main recognizes PtdIns(3,4,5)P3 with high specificity and af- The signaling lipid phosphatidylinositol 3,4,5-trisphos- finity, however, the precise mechanism of its association phate [PtdIns(3,4,5)P3] is produced in plasma membranes at Albert Einstein College of Medicine Library on July 14, 2008 with membranes remains unclear. Here, we detail the mo- in response to stimulation of cell surface receptors by lecular basis of membrane anchoring by the GRP1 PH do- growth factors and hormones (1). Class I phosphoinositide main. Our data reveal a multivalent membrane docking (PI) 3-kinases phosphorylate the inositol headgroup of the involving PtdIns(3,4,5)P binding, regulated by pH and fa- 3 relatively abundant phosphatidylinositol 4,5-bisphosphate cilitated by electrostatic interactions with other anionic lip- [Ptdns(4,5)P2], transiently elevating the level of PtdIns ids. -

Predicting Clinical Response to Treatment with a Soluble Tnf-Antagonist Or Tnf, Or a Tnf Receptor Agonist
(19) TZZ _ __T (11) EP 2 192 197 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 02.06.2010 Bulletin 2010/22 C12Q 1/68 (2006.01) (21) Application number: 08170119.5 (22) Date of filing: 27.11.2008 (84) Designated Contracting States: (72) Inventor: The designation of the inventor has not AT BE BG CH CY CZ DE DK EE ES FI FR GB GR yet been filed HR HU IE IS IT LI LT LU LV MC MT NL NO PL PT RO SE SI SK TR (74) Representative: Habets, Winand Designated Extension States: Life Science Patents AL BA MK RS PO Box 5096 6130 PB Sittard (NL) (71) Applicant: Vereniging voor Christelijk Hoger Onderwijs, Wetenschappelijk Onderzoek en Patiëntenzorg 1081 HV Amsterdam (NL) (54) Predicting clinical response to treatment with a soluble tnf-antagonist or tnf, or a tnf receptor agonist (57) The invention relates to methods for predicting a clinical response to a therapy with a soluble TNF antagonist, TNF or a TNF receptor agonist and a kit for use in said methods. EP 2 192 197 A1 Printed by Jouve, 75001 PARIS (FR) EP 2 192 197 A1 Description [0001] The invention relates to methods for predicting a clinical response to a treatment with a soluble TNF antagonist, with TNF or a TNF receptor agonist using expression levels of genes of the Type I INF pathway and a kit for use in said 5 methods. In another aspect, the invention relates to a method for evaluating a pharmacological effect of a treatment with a soluble TNF antagonist, TNF or a TNF receptor agonist. -

The PX Domain Protein Interaction Network in Yeast
The PX domain protein interaction network in yeast Zur Erlangung des akademischen Grades eines DOKTORS DER NATURWISSENSCHAFTEN (Dr. rer. nat.) der Fakultät für Chemie und Biowissenschaften der Universität Karlsruhe (TH) vorgelegte DISSERTATION von Dipl. Biol. Carolina S. Müller aus Buenos Aires Dekan: Prof. Dr. Manfred Kappes Referent: Dr. Nils Johnsson Korreferent: HD. Dr. Adam Bertl Tag der mündlichen Prüfung: 17.02.2005 I dedicate this work to my Parents and Alex TABLE OF CONTENTS Table of contents Introduction 1 Yeast as a model organism in proteome analysis 1 Protein-protein interactions 2 Protein Domains in Yeast 3 Classification of protein interaction domains 3 Phosphoinositides 5 Function 5 Structure 5 Biochemistry 6 Localization 7 Lipid Binding Domains 8 The PX domain 10 Function of PX domain containing proteins 10 PX domain structure and PI binding affinities 10 Yeast PX domain containing proteins 13 PX domain and protein-protein interactions 13 Lipid binding domains and protein-protein interactions 14 The PX-only proteins Grd19p and Ypt35p and their phenotypes 15 Aim of my PhD work 16 Project outline 16 Searching for interacting partners 16 Confirmation of obtained interactions via a 16 second independent method Mapping the interacting region 16 The Two-Hybrid System 17 Definition 17 Basic Principle of the classical Yeast-Two Hybrid System 17 Peptide Synthesis 18 SPOT synthesis technique 18 Analysis of protein- peptide contact sites based on SPOT synthesis 19 TABLE OF CONTENTS Experimental procedures 21 Yeast two-hybrid assay