Morphological, Ultrastructural, and Phylogenetic Analysis of Two Novel
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Estimating Migratory Fish Distribution from Altitude and Basin Area: a Case
Freshwater Biology (2012) 57, 2297–2305 doi:10.1111/fwb.12003 Estimating migratory fish distribution from altitude and basin area: a case study in a large Neotropical river JOSE´ RICARDO S. BARRADAS*, LUCAS G. SILVA*, BRET C. HARVEY† AND NELSON F. FONTOURA* *Departamento de Biodiversidade e Ecologia, Pontifı´cia Universidade Cato´lica do Rio Grande do Sul (PUCRS), Porto Alegre, RS, Brazil †USDA Forest Service, Pacific Southwest Research Station, Arcata, CA, U.S.A. SUMMARY 1. The objective of this study was to identify longitudinal distribution patterns of large migratory fish species in the Uruguay River basin, southern Brazil, and construct statistical distribution models for Salminus brasiliensis, Prochilodus lineatus, Leporinus obtusidens and Pseudoplatystoma corruscans. 2. The sampling programme resulted in 202 interviews with old residents and fishermen at 167 different sampling sites in the major rivers of the basin. Interviewees were asked whether these fish species were present in the nearby river segment, recently or in the past. The average age of respondents was 56 years, and the great majority had resided for more than 30 years in the area where they were interviewed. 3. Fish distribution patterns were modelled using altitude and basin area as environmental descriptors. Distributional patterns were estimated using logistic equations (LOGIT): À1 P ¼ eða0þa1 lnðAlÞþa2 lnðBAÞÞð1 þ eða0þa1 lnðAlÞþa2 lnðBAÞÞÞ , where Al is the altitude and BA is the basin area. 4. Accuracies of the distribution models were between 77 and 85%. These accuracies are similar to those of published distribution models for other fishes based on larger numbers of environmental descriptors. 5. The historical distributional ranges make clear that all operating or planned large hydropower dams in the Uruguay River basin, with one exception, limit or will limit the distribution of migratory fishes. -

First Evidence of a Spawning Site of the Endangered Fish Brycon
Short Research Note Acta Limnologica Brasiliensia, 2020, vol. 32, e23 https://doi.org/10.1590/S2179-975X2220 ISSN 2179-975X on-line version First evidence of a spawning site of the endangered fish Brycon orbignyanus (Valenciennes, 1850) (Characiformes, Bryconidae) in the Middle Uruguay River, Brazil Primeira evidência de um local de desova do peixe ameaçado de extinção Brycon orbignyanus (Valenciennes, 1850) (Characiformes, Bryconidae) no Médio rio Uruguai, Brasil David Augusto Reynalte-Tataje1* , Marlon da Luz Soares1 , Marthoni Vinicius Massaro1 , Rodrigo Bastian1 , Fernando Mayer Pelicice2 1 Universidade Federal da Fronteira Sul – UFFS. Rua Jacob Reinaldo Haupenthal, 1580, CEP 97900-000, Cerro Largo, RS, Brasil 2 Núcleo de Estudos Ambientais, Universidade Federal do Tocantins – UFT, Rua 3, Quadra 11 17, Jardim dos Ipês, CEP 77500-000, Porto Nacional, TO, Brasil *e-mail: [email protected] Cite as: Reynalte-Tataje, D.A. et al. First evidence of a spawning site of the endangered fish Brycon orbignyanus (Valenciennes, 1850) (Characiformes, Bryconidae) in the Middle Uruguay River, Brazil. Acta Limnologica Brasiliensia, 2020, vol. 32, e23. Abstract: Aim: This study provides the first direct evidence about the location of a spawning site of Brycon orbignyanus (Valenciennes, 1850) in the Middle Uruguay River. Methods: Sampling was carried out with a plankton net (500 µm) in November 2017 in the Uruguay River near the confluence with the Piratinim River, southern Brazil. Eggs were sorted in the field, and to confirm species identity we used the live ichthyoplankton technique. Results: We captured 47 eggs of B. orbignyanus in the channel of the Uruguay River, characterized by fast flow, shallow depth (approximately 3 m) and rocky bottom, located a few kilometers upstream from its confluence with the Piratinim River. -

Myxobolus Spp. (Cnidaria: Myxobolidae) in the Circulating Blood of Fishes from Goiás and Mato Grosso States, Brazil: Case Report Myxobolus Spp
ISSN 1519-6984 (Print) ISSN 1678-4375 (Online) THE INTERNATIONAL JOURNAL ON NEOTROPICAL BIOLOGY THE INTERNATIONAL JOURNAL ON GLOBAL BIODIVERSITY AND ENVIRONMENT Original Article Myxobolus spp. (Cnidaria: Myxobolidae) in the circulating blood of fishes from Goiás and Mato Grosso States, Brazil: case report Myxobolus spp. (Cnidaria: Myxobolidae) no sangue circulante de peixes dos estados de Goiás e Mato Grosso, Brasil: relato de caso L. P. Úngaria* , D. H. M. D. Vieiraa , A. L. Q. Santosb , R. J. da Silvaa and L. H. O’Dwyera a Setor de Parasitologia, Departamento de Bioestatística, Biologia vegetal, Parasitologia e Zoologia – DBBVPZ, Instituto de Biociências, Universidade Estadual Paulista – UNESP, Botucatu, SP, Brasil b Laboratório de Ensino e Pesquisa em Animais Silvestres – LAPAS, Faculdade de Medicina Veterinária, Universidade Federal de Uberlândia – UFU, Uberlândia, MG, Brasil Abstract Myxosporidiosis is an infectious disease caused by myxozoans of the Phylum Cnidaria, Class Myxosporea, and Order Bivalvulida, considered a common parasite in fresh and saltwater fishes that parasitize many organs, especially gills. In the present study, 49 specimens of fishes belonging to eight genera: Tetragonopterus, Leporinus, Myleus, Pirinampus, Rhapiodon, Pygocentrus, Ageneiosus, and Serrasalmus were collected and blood smears were made, fixed with absolute methanol, and stained with Giemsa 10% to survey hemoparasites. However, myxospores were found in the circulating blood of five (10.20%) fishes belonging to genus Tetragonopterus, Myleus, and Pygocentrus. Two morphological types of Myxobolus spp. were identified in all the five fish specimens analyzed. Usually, investigations on myxozoans in fish are carried out with the search for plasmodia or cysts in the fish organs and observation of the cavity of organs. -

January 2020 54. Milanin T, Bartholomew JL, Atkinson SD
Peer-reviewed Journal articles – S.D.Atkinson – January 2020 54. Milanin T, Bartholomew JL, Atkinson SD (2020) An introduced host with novel and introduced parasites: Myxobolus spp. (Cnidaria: Myxozoa) in yellow perch Perca flavescens. Parasitology Research DOI:10.1007/s00436-019-06585-3 53. Richey CA, Kenelty KV, Hopkins KVS, Stevens BN, Martínez-López B, Hallett SL, Atkinson SD, Bartholomew JL, Soto E (2020) Validation of environmental DNA sampling for determination of Ceratonova shasta (Noble, 1950) (Cnidaria: Myxozoa) distribution in Plumas National Forest, CA. Journal of Aquatic Animal Health epub DOI:10.1007/s00436-019-06509-1 52. Atkinson SD, Hallett SL, Díaz Morales D, Bartholomew JL, de Buron I (2019) First myxozoan infection (Cnidaria: Myxosporea) in a marine polychaete from North America, and erection of actinospore collective group Saccimyxon. Journal of Parasitology 105(2):252-262 DOI:10.1645/18-183 51. Alama-Bermejo, G, Viozzi GP, Waicheim MA, Flores VR, Atkinson SD (2019) Host-parasite relationship of Ortholinea lauquen n. sp. (Cnidaria:Myxozoa) and the fish Galaxias maculatus (Jenyns, 1842) in northwest Patagonia, Argentina. Diseases of Aquatic Organisms 136(2):163-174 DOI: 10.3354/dao03400 50. Borkhanuddin MH, Cech G, Molnár K, Shaharom-Harrison F, Duy Khoa TN, Samshuri MA, Mazelan S, Atkinson SD, Székely C (2019) Henneguya (Cnidaria: Myxosporea: Myxobolidae) infections of cultured barramundi, Lates calcarifer (Perciformes: Latidae) in an estuarine wetlands system of Malaysia: Description of Henneguya setiuensis n. sp., Henneguya voronini n. sp. and Henneguya calcarifer n. sp. Parasitology Research 119(1):85-96 DOI: 10.1007/s00436-019-06541-1 49. Breyta R, Atkinson SD, Bartholomew JL (2019) Evolutionary dynamics of Ceratonova species in the Klamath River basin reveals different host adaptation strategies. -

Aquaculture Production in Brazil 600000 562.533 500000
Transformations in Brazilian aquaculture: technological intensification and capital concentration Manoel Pedroza Embrapa Fisheries and Aquaculture Brazilian Agricultural Research Corporation What about the Brazilian aquaculture? » Significant growth in recent years Aquaculture production in Brazil 600000 562.533 500000 Tons 14º biggest - 400000 Inland 300000 world Marine 200000 producer 100000 Total Production Production 0 2008 2010 2012 2014 » Inland production semi-intensive and extensive systems Floating cages Earthen ponds Source: FAO, 2016 » Great diversity of species » Manly native » Geographical dispersion Aquaculture production in Brazil (2014) • White legged shrimp • Tilapia (42%) (O. niloticus) ≠’s strains (78%) (L.vannamei) (Thai/Chitralada/ Gift) 18% • Tambaqui (29%) (Colossoma macropomum) • Pacific oyster (C. gigas) + Brown mussel (P.perna) • Tambacu and tambatinga (9%) (hybrids) (22%) 82% • Carps (4%) (≠’s spp.) • Spotted catfish (4%) (Psedoplatystoma spp.) • Others (12%) Inland aquaculture Mariculture Source: IBGE, Ministry of Fisheries and Aquaculture Historical driving forces in the Brazilian aquaculture » Increase in demand of seafood » 2005 = 6,6 kg/habitant/year 2015 = 10,6 kg/habitant/year (+60,6%) » 1 billion US$ seafood imports » Low scale of producers » Most of producers with annual production of less than 5.000 tones » Few producers organizations » Low technological intensity » Innovation gap on native species Aquaculture production in Brazil by category of species (tons) Dourado (Salminus brasiliensis) 38 Peacock -

BMC Evolutionary Biology, 2014, 14
Abe et al. BMC Evolutionary Biology 2014, 14:152 http://www.biomedcentral.com/1471-2148/14/152 RESEARCH ARTICLE Open Access Systematic and historical biogeography of the Bryconidae (Ostariophysi: Characiformes) suggesting a new rearrangement of its genera and an old origin of Mesoamerican ichthyofauna Kelly T Abe, Tatiane C Mariguela, Gleisy S Avelino, Fausto Foresti and Claudio Oliveira* Abstract Background: Recent molecular hypotheses suggest that some traditional suprageneric taxa of Characiformes require revision, as they may not constitute monophyletic groups. This is the case for the Bryconidae. Various studies have proposed that this family (considered a subfamily by some authors) may be composed of different genera. However, until now, no phylogenetic study of all putative genera has been conducted. Results: In the present study, we analyzed 27 species (46 specimens) of all currently recognized genera of the Bryconidae (ingroup) and 208 species representing all other families and most genera of the Characiformes (outgroup). Five genes were sequenced: 16SrRNA, Cytochrome b, recombination activating gene 1 and 2 and myosin heavy chain 6 cardiac muscle. The final matrix contained 4699 bp and was analyzed by maximum likelihood, maximum parsimony and Bayesian analyses. The results show that the Bryconidae, composed of Brycon, Chilobrycon, Henochilus and Salminus, is monophyletic and is the sister group of Gasteropelecidae + Triportheidae. However, the genus Brycon is polyphyletic. Fossil studies suggest that the family originated approximately 47 million years ago (Ma) and that one of the two main lineages persisted only in trans-Andean rivers, including Central American rivers, suggesting a much older origin of Mesoamerican ichthyofauna than previously accepted. -

Genetic Structuring of Salminus Hilarii Valenciennes, 1850 (Teleostei
Neotropical Ichthyology, 13(3): 547-556, 2015 Copyright © 2015 Sociedade Brasileira de Ictiologia DOI: 10.1590/1982-0224-20150015 Genetic structuring of Salminus hilarii Valenciennes, 1850 (Teleostei: Characiformes) in the rio Paraná basin as revealed by microsatellite and mitochondrial DNA markers Juliana Viana da Silva1, Eric M. Hallerman2, Laura Helena Orfão3 and Alexandre Wagner Silva Hilsdorf1 Genetic variation of Salminus hilarii was assessed by screening microsatellite loci and mitochondrial D-loop DNA across four sampling in the upper rio Paraná basin of Brazil. Genetic diversity – measured as mean expected heterozygosity (0.904) and mean number of alleles across populations (13.7) – was reasonably high. Differentiation of microsatellite allele frequencies among populations was shown to be low but significant by AMOVAΦ ST (0.0192), and high by DEST (0.185). D-loop variation was high, with haplotypic diversity of 0.950 and nucleotide diversity of 0.011. Mitochondrial DNA-based estimates for population differentiation were high, with an overall ΦST of 0.173. The results of tests of nuclear and mitochondrial variation yielded no unequivocal inference of historical demographic bottleneck or expansion. Genetic differentiation observed among S. hilarii populations in the rio Grande may be caused by a combination of historical differentiation and recent gene-flow disruption caused by the dams followed by reproduction of isolated spawning assemblages in mid-sized tributaries of the respective reservoirs. We present spatially more intensive sampling of S. hilarii populations across the rio Paraná basin in order to more effectively distinguish between historical and contemporary differentiation. A variabilidade genética de Salminus hilarii foi avaliada por lócus microssatélites e sequências D-Loop do DNA mitocondrial em quatro populações da região da bacia do Alto Paraná. -

The Unique Karyotype of Henochilus Wheatlandii, a Critically Endangered Fish Living in a Fast-Developing Region in Minas Gerais State, Brazil
The Unique Karyotype of Henochilus wheatlandii,a Critically Endangered Fish Living in a Fast-Developing Region in Minas Gerais State, Brazil Priscilla C. Silva1*, Udson Santos1, Nata´lia M. Travenzoli1, Jose C. Zanuncio1, Marcelo de B. Cioffi2, Jorge A. Dergam1 1 Laborato´rio de Sistema´tica Molecular - Beagle, Departamento de Biologia Animal, Universidade Federal de Vic¸osa, Vic¸osa, Brazil, 2 Laborato´rio de biodiversidade molecular e citogene´tica, Departamento de Gene´tica e Evoluc¸a˜o, Universidade Federal de Sa˜o Carlos, Sa˜o Carlo, Brazil Abstract Henochilus wheatlandii, the only species of this genus, is critically endangered and was considered extinct for over a century. The rediscovery of this fish in 1996 made it possible to study its phylogenetic relationships with other species in the subfamily Bryconinae. The aim of this study was to characterise the karyotype of H. wheatlandii. Standard staining, C- positive heterochromatin and nucleolar organiser region (NOR) banding, chromomycin A3 staining, and fluorescent in situ hybridisation (FISH) using 5S rDNA and 18S rDNA probes were conducted on nineteen specimens collected in the Santo Antonio River, a sub-basin of the Doce River in Ferros municipality, Minas Gerais State, Brazil. Henochilus wheatlandii shared the same diploid number and chromosome morphology as other species of Bryconinae. However, its heterochromatin distribution patterns, NOR localisation, and FISH patterns revealed a cytogenetic profile unique among Neotropical Bryconinae, emphasizing the evolutionary uniqueness of this threatened species. Citation: Silva PC, Santos U, Travenzoli NM, Zanuncio JC, Cioffi MdB, et al. (2012) The Unique Karyotype of Henochilus wheatlandii, a Critically Endangered Fish Living in a Fast-Developing Region in Minas Gerais State, Brazil. -
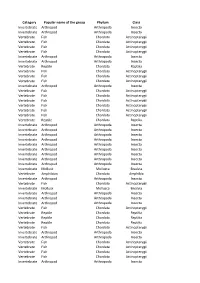
Category Popular Name of the Group Phylum Class Invertebrate
Category Popular name of the group Phylum Class Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Vertebrate Reptile Chordata Reptilia Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Invertebrate Arthropod Arthropoda Insecta Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Fish Chordata Actinopterygii Vertebrate Reptile Chordata Reptilia Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Mollusk Mollusca Bivalvia Vertebrate Amphibian Chordata Amphibia Invertebrate Arthropod Arthropoda Insecta Vertebrate Fish Chordata Actinopterygii Invertebrate Mollusk Mollusca Bivalvia Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Invertebrate Arthropod Arthropoda Insecta Vertebrate -

Download This PDF File
22777 Brazilian Journal of Development Estimation of the age and biometry of Salminus brasiliensis (Cuvier 1816) captured in the Funil hydroelectric plants Estimativa da idade e biometria de Salminus brasiliensis (Cuvier 1816) capturada nas hidrelétricas de Funil DOI:10.34117/bjdv6n4-441 Recebimento dos originais: 02/03/2020 Aceitação para publicação: 01/04/2020 Athalita Ester Mendonça da Silva Piva Ferreira Mestre em Engenharia Agrícola pela Universidade Federal de Lavras Instituição: Universidade Federal de Lavras Campus Universitário, Caixa Postal 3037, CEP: 37200-900, Lavras-MG, Brasil E-mail: [email protected] Carlos Cicinato Vieira Melo Doutor em Zootecnia pela Universidade Federal de Lavras Instituição: Centro Universitário Tocantinense Presidente Antônio Carlos Av. Filadélfia, 568 - St. Oeste, CEP: 77816-540, Araguaína-TO, Brasil E-mail: [email protected] Natália Michele Nonato Mourad Doutora em Zootecnia pela Universidade Federal de Lavras Instituição: Universidade Federal de Lavras Campus Universitário, Caixa Postal: 3037, CEP: 37200-900, Lavras-MG, Brasil E-mail:[email protected] Viviane de Oliveira Felizardo Doutora em Zootecnia pela Universidade Federal de Lavras Instituição: Universidade Federal de Lavras Campus Universitário, Caixa Postal 3037, CEP: 37200-900, Lavras-MG, Brasil E-mail: [email protected] William Franco Carneiro Doutorando em Zootecnia pela Universidade Federal de Lavras Instituição: Universidade Federal de Lavras Campus Universitário, Caixa Postal 3037, CEP: 37200-900, Lavras-MG, Brasil E-mail: [email protected] Rilke Tadeu Fonseca de Freitas Doutor em Zootecnia pela Universidade Federal de Viçosa Instituição: Universidade Federal de Lavras Campus Universitário, Caixa Postal: 3037, CEP: 37200-900, Lavras-MG, Brasil E-mail: [email protected] Luis David Solis Murgas* Doutor em Zootecnia pela Universidade Federal de Lavras Instituição: Universidade Federal de Lavras Braz. -

Cnidaria Myxosporea, Myxobolidae), a Myxozoan Parasite of Esomus Danrica Hamilton, 1882 from Ponds and Ditches of Thoubal, Manipur, India
Original Article Bulletin of Pure and Applied Sciences. Vol.36 A (Zoology), No.2, 2017: P.93-101 Print version ISSN 0970 0765 Online version ISSN 2320 3188 DOI 10.5958/2320-3188.2017.00013.4 Authors Affiliation: MYXOBOLUS DANRICI SP. N. (CNIDARIA 1Professor, MYXOSPOREA, MYXOBOLIDAE), A Parasitology Section, MYXOZOAN PARASITE OF ESOMUS DANRICA Life Sciences Department, Manipur University, HAMILTON, 1882 FROM PONDS AND Canchipur-795003 DITCHES OF THOUBAL, MANIPUR, INDIA E-mail: [email protected] N. Mohilal & T. Soni 2Research Scholar, Parasitology Section, Life Sciences Department, Manipur University, Canchipur-795003 E-mail: [email protected] *Corresponding Author: Prof. Naorem Mohilal Meitei Professor, Parasitology Section, Life Sciences Department, Manipur University, Canchipur-795003 E-mail: [email protected] Received on 12.10.2017 Accepted on 02.12.2017 Abstract: Myxobolus Myxobolus danrici A new species of the genus , sp. n. is obtained from an ornamental fish Esomus danrica (Hamilton, 1822) commonly called flying barb from Thoubal, Manipur, India. The diagnostic characters are: spores spherical with rounded ends in frontal view, biconvex-shaped in sutural view with thick straight sutural line. Polar capsules equal, pyriform with a prominent nipple-shaped anterior ends; distinct V- shaped intercapsular appendage; polar filament makes 5 - 6 turns of coil. Sporoplasm anchor-shaped, rise up between the two polar capsules which touches the tip of the intercapsular appendage. Keywords: Myxozoa, Myxobolus danrici, Esomus danrica, Thoubal, India. N. Mohilal & T. Soni / Myxobolus danrici sp. N. (cnidaria myxosporea, myxobolidae), A Myxozoan Parasite of Esomus Danrica Hamilton, 1882 From Ponds and Ditches of Thoubal, Manipur, India INTRODUCTION Fishes are parasitized by different groups of parasites. -

Exposure of Fishery Resources to Environmental and Socioeconomic Threats Within the Pantanal Wetland of South America
vv Life Sciences Group International Journal of Aquaculture and Fishery Sciences ISSN: 2455-8400 DOI CC By Cleber JR Alho1* and Roberto E Reis2 Review Article 1Professor, Graduate Program in the Environment, University Anhanguera-Uniderp, Alexandre Herculano Street, 1400 - Jardim Veraneio, Campo Grande, MS Exposure of Fishery Resources to 79037-280, Brazil 2Professor, Pontifícia Universidade Católica do Environmental and Socioeconomic Rio Grande do Sul, Laboratório de Sistemática de Vertebrados and Regional Chair for South America of the Freshwater Fish Specialist Group of IUCN / Threats within the Pantanal Wetland Wetlands International, Brazil Dates: Received: 06 April, 2017; Accepted: 03 May, of South America 2017; Published: 04 May, 2017 *Corresponding author: Cleber JR Alho, Professor, Graduate Program in the Environment, University Abstract Anhanguera-Uniderp, Alexandre Herculano Street, 1400 - Jardim Veraneio, Campo Grande, MS 79037- The huge Pantanal wetland, located in the central region of South America, mainly in Brazil, formed by 280, Brazil, Tel: +55 61 3365-3142; +55 61 99989- the Upper Paraguay River Basin, comprising 150,355 km² (approximately 140,000 km² in Brazil), is facing 3142; E-Mail: environmental and socioeconomic threats that are affecting fi sh populations and fi shery resources. The Paraguay River and its tributaries feed the Pantanal wetland, forming a complex aquatic ecosystem, Keywords: Biodiversity, Environmental threats; So- harboring more than 260 fi sh species, some of them with great subsistence and commercial values to cioeconomic threats; Fishery resources; Freshwater regional human communities. Sport fi shing is also preeminent in the region. The natural ecosystems and habitats; Pantanal wetland the increasing human population that depend on them are at risk from a number of identifi ed threats, https://www.peertechz.com including natural habitat disruptions and overfi shing.