New Catalysts for Clean Environment 2Nd Symposium of the VTT Research Programme on Chemical Reaction Mechanisms
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Carbonylation of Protected Or Non-Protected 2-Bromobenzaldehyde Catalyzed by Cobalt Carbonyl
772 Bull. Korean Chem. Soc. 1994, Vol. 15, No. 9 Sang Chui Shim et al. Carbonylation of Protected or Non-protected 2-Bromobenzaldehyde Catalyzed by Cobalt Carbonyl Sang Chui S버 m*, Dong Yub Lee, Heung Jin Choi, Chil Hoon Doh1, and Keun Tai Huh* ^Department of Industrial Chemistry, Kyungpook National University, Taegu 701-702, Korea ^Department of Materials Science and Engineering Kyungsung University, Pusan 608-736, Korea Korea Electrotechnology Research Institute, Changwon, Korea Received May 11, 1994 The cobalt catalyzed carbonylation of bromobenzene having protected aldehyde group gives the corresponding ester in good yields, but 2-bromobenzaldehyde gives 3-alkoxyphthalide in the noticeable yield instead of alkyl 2-formylben- zoates. Introduction Table 1. Carbonylation of 2-Bromobenzenes Having Protected Aldehyde Groups to 2-Substituted Alkyl Benzoates Catalyzed by The metal-catalyzed carbonyl가 ion of aryl halides has ver Cobalt CarbonyF satile utility in the preparative organic chemistry1. Of many Run Reactant catalysts2 used in the carbonylation of aryl halides cobalt Alcohol Product Yield (%y carbonyl species have been conducted under mild conditions, 1 1 CH3CH2OH 2b 88 room temperature and one atmospheric pressure of carbon 2 1 CH3CH2OH 2b 75 。 monoxide. More recently, using dicobalt octacarbonyl at room 3 1 CH3CH2OH 2b 38d temperature under one atmospheric pressure of carbon mo 4 1 CH3OH 2a 80 noxide many applications were reported on the carbonylation 5 1 CH3CH2CH2OH 2c 85 of benzal halides,3-5 and halo (halomethyl)benzenes6-8. 6 (CH3)2CHOH 21 However, studies2,9-11 on the carbonylation of aryl halides 1 2d 7 CH3CH2CH2CH2OH having other reactive functional groups are rare. -

Direct Carboniiatiom of Aromatic Nttriles Using
DIRECT CARBONIIATIOM OF AROMATIC NTTRILES USING DICOBALT OCTACARBONIL by JOSEPH EDMUND GERVAX B..Sc Honours, University of Montreal, Loyola College, l?6l A THESIS SUBMITTED IN PARTIAL FUIFIIMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE in the Department of Chemistry We accept this; thesis as conforming to the required standard THE UNIVERSITY OF BRITISH COLUMBIA June, 1963 In presenting this thesis in partial fulfilment of the requirements for an advanced degree at the University of British Columbia, I agree that the Library shall make it freely available for reference and study. I further agree that per• mission for extensive copying of this thesis for scholarly purposes may be granted by the Head of my Department or by his representatives. It is understood that copying, or publi• cation of this thesis for financial gain shall not be allowed without my written permission. Department of CHEMISTRY The University of British Columbia,. Vancouver 8, Canada. Date June 2°» W ABSTRACT A new method of synthesizing N-substituted phthalimidines is described. When benzonitrile was reacted with carbon monoxide containing about 0.0k volume percent of hydrogen and in the presence of dicobalt octacarbonyl and pyridine in benzene solution at 235° and 3U00 p.s.i. pressure the following compounds were produced: MTbenzyphthalimidine (8$), N-phenylphthalimidine (3»7%), and benzamide (3*9%)- When lj.0 p.s.i. hydrogen was added under the same reaction conditions, the yield of N-benzylphthalimidine increased (16%)., When benzonitrile was subjected to the same reaction conditions using 2U0 p.s.d* hydrogen and no pyridine, N-benzylphthalimidine (1%%) and sym-dibenzylurea (8$) were produced. -

Chemical Intercalation of Zerovalent Metals Into 2D Layered Bi2se3 Nanoribbons † † ‡ † † † § Kristie J
Article pubs.acs.org/JACS Chemical Intercalation of Zerovalent Metals into 2D Layered Bi2Se3 Nanoribbons † † ‡ † † † § Kristie J. Koski, Colin D. Wessells, Bryan W. Reed, Judy J. Cha, Desheng Kong, and Yi Cui*, , † Department of Materials Science and Engineering, Stanford University, Stanford, California 94305, United States ‡ Physical and Life Sciences Directorate, Lawrence Livermore National Laboratory, 7000 East Avenue, Livermore, California 94550, United States § SLAC National Accelerator Laboratory, Stanford Institute for Materials and Energy Sciences, 2575 Sand Hill Road, Menlo Park, California 94025, United States *S Supporting Information ABSTRACT: We have developed a chemical method to intercalate a variety of zerovalent metal atoms into two-dimen- sional (2D) layered Bi2Se3 chalcogenide nanoribbons. We use a chemical reaction, such as a disproportionation redox reaction, to generate dilute zerovalent metal atoms in a refluxing solution, which intercalate into the layered Bi2Se3 structure. The zerovalent nature of the intercalant allows superstoichiometric intercalation of metal atoms such as Ag, Au, Co, Cu, Fe, In, Ni, and Sn. We foresee the impact of this methodology in establishing novel fundamental physical behaviors and in possible energy applications. 1. INTRODUCTION Ni, and Sn. Some interesting effects that could arise with − 7−10 intercalation are superconductivity, such as in Cu Bi2Se3, Intercalation is the insertion of a guest species into a host 6 lattice. Intercalation into layered materials is essential to battery enhanced conductivity, or possibly opening a surface state gap electrodes, electrochromics, detergents, and solid lubricants and in topological insulator Bi2Se3. This method of zerovalent metal is important in exotic fundamental two-dimensional (2D) intercalation may also be extended to other layered materials. -

Bond Distances and Bond Orders in Binuclear Metal Complexes of the First Row Transition Metals Titanium Through Zinc
Metal-Metal (MM) Bond Distances and Bond Orders in Binuclear Metal Complexes of the First Row Transition Metals Titanium Through Zinc Richard H. Duncan Lyngdoh*,a, Henry F. Schaefer III*,b and R. Bruce King*,b a Department of Chemistry, North-Eastern Hill University, Shillong 793022, India B Centre for Computational Quantum Chemistry, University of Georgia, Athens GA 30602 ABSTRACT: This survey of metal-metal (MM) bond distances in binuclear complexes of the first row 3d-block elements reviews experimental and computational research on a wide range of such systems. The metals surveyed are titanium, vanadium, chromium, manganese, iron, cobalt, nickel, copper, and zinc, representing the only comprehensive presentation of such results to date. Factors impacting MM bond lengths that are discussed here include (a) n+ the formal MM bond order, (b) size of the metal ion present in the bimetallic core (M2) , (c) the metal oxidation state, (d) effects of ligand basicity, coordination mode and number, and (e) steric effects of bulky ligands. Correlations between experimental and computational findings are examined wherever possible, often yielding good agreement for MM bond lengths. The formal bond order provides a key basis for assessing experimental and computationally derived MM bond lengths. The effects of change in the metal upon MM bond length ranges in binuclear complexes suggest trends for single, double, triple, and quadruple MM bonds which are related to the available information on metal atomic radii. It emerges that while specific factors for a limited range of complexes are found to have their expected impact in many cases, the assessment of the net effect of these factors is challenging. -

Intramolecular Ene Reactions of Functionalised Nitroso Compounds
INTRAMOLECULAR ENE REACTIONS OF FUNCTIONALISED NITROSO COMPOUNDS A Thesis Presented by Sandra Luengo Arratta In Partial Fulfilment of the Requirements for the Award of the Degree of DOCTOR OF PHILOSOPHY OF UNIVERSITY COLLEGE LONDON Christopher Ingold Laboratories Department of Chemistry University College London 20 Gordon Street London WC1H 0AJ July 2010 DECLARATION I Sandra Luengo Arratta, confirm that the work presented in this thesis is my own. Where work has been derived from other sources, I confirm that this has been indicated in the thesis. ABSTRACT This thesis concerns the generation of geminally functionalised nitroso compounds and their subsequent use in intramolecular ene reactions of types I and II, in order to generate hydroxylamine derivatives which can evolve to the corresponding nitrones. The product nitrones can then be trapped in the inter- or intramolecular mode by a variety of reactions, including 1,3-dipolar cycloadditions, thereby leading to diversity oriented synthesis. The first section comprises the chemistry of the nitroso group with a brief discussion of the current methods for their generation together with the scope and limitations of these methods for carrying out nitroso ene reactions, with different examples of its potential as a powerful synthetic method to generate target drugs. The second chapter describes the results of the research programme and opens with the development of methods for the generation of functionalised nitroso compounds from different precursors including oximes and nitro compounds, using a range of reactants and conditions. The application of these methods in intramolecular nitroso ene reactions is then discussed. Chapter three presents the conclusions which have been drawn from the work presented in chapter two, and provides suggestions for possible directions of this research in the future. -

Palladium-Bismuth Intermetallic and Surface-Poisoned Catalysts for the Semi-Hydrogenation of 2-Methyl-3-Butyn-2-Ol
Accepted Manuscript Title: Palladium-bismuth intermetallic and surface-poisoned catalysts for the semi-hydrogenation of 2-methyl-3-butyn-2-ol Author: Nikolay Cherkasov Alex O. Ibhadon Alan McCue James A. Anderson Shaun K. Johnston PII: S0926-860X(15)00136-2 DOI: http://dx.doi.org/doi:10.1016/j.apcata.2015.02.038 Reference: APCATA 15280 To appear in: Applied Catalysis A: General Received date: 1-12-2014 Revised date: 4-2-2015 Accepted date: 20-2-2015 Please cite this article as: N. Cherkasov, A.O. Ibhadon, A. McCue, J.A. Anderson, S.K. Johnston, Palladium-bismuth intermetallic and surface-poisoned catalysts for the semi-hydrogenation of 2-methyl-3-butyn-2-ol, Applied Catalysis A, General (2015), http://dx.doi.org/10.1016/j.apcata.2015.02.038 This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain. Highlights • Pd-Bi catalysts were studied in the 2-methyl-3-butyn-3-ol semi-hydrogenation • Poisoning with Bi increases alkene selectivity, but decreases activity • Poisoning with Bi hinders the formation of Pd beta-hydride phase • Kinetic modelling suggets significant ligand effects at high Bi content Accepted Manuscript 1 Page 1 of 25 Palladium-bismuth intermetallic and surface-poisoned catalysts for the semi- hydrogenation of 2-methyl-3-butyn-2-ol Nikolay Cherkasova, Alex O. -
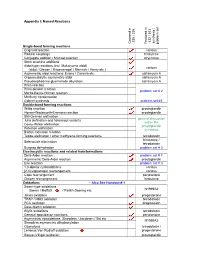
Appendix I: Named Reactions Single-Bond Forming Reactions Co
Appendix I: Named Reactions 235 / 335 432 / 533 synthesis / / synthesis Covered in Covered Featured in problem set problem Single-bond forming reactions Grignard reaction various Radical couplings hirstutene Conjugate addition / Michael reaction strychnine Stork enamine additions Aldol-type reactions (incl. Mukaiyama aldol) various (aldol / Claisen / Knoevenagel / Mannich / Henry etc.) Asymmetric aldol reactions: Evans / Carreira etc. saframycin A Organocatalytic asymmetric aldol saframycin A Pseudoephedrine glycinamide alkylation saframycin A Prins reaction Prins-pinacol reaction problem set # 2 Morita-Baylis-Hillman reaction McMurry condensation Gabriel synthesis problem set #3 Double-bond forming reactions Wittig reaction prostaglandin Horner-Wadsworth-Emmons reaction prostaglandin Still-Gennari olefination general discussion Julia olefination and heteroaryl variants within the Corey-Winter olefination prostaglandin Peterson olefination synthesis Barton extrusion reaction Tebbe olefination / other methylene-forming reactions tetrodotoxin hirstutene / Selenoxide elimination tetrodotoxin Burgess dehydration problem set # 3 Electrocyclic reactions and related transformations Diels-Alder reaction problem set # 1 Asymmetric Diels-Alder reaction prostaglandin Ene reaction problem set # 3 1,3-dipolar cycloadditions various [2,3] sigmatropic rearrangement various Cope rearrangement periplanone Claisen rearrangement hirstutene Oxidations – Also See Handout # 1 Swern-type oxidations (Swern / Moffatt / Parikh-Doering etc. N1999A2 Jones oxidation -

Steam Cracking: Chemical Engineering
Steam Cracking: Kinetics and Feed Characterisation João Pedro Vilhena de Freitas Moreira Thesis to obtain the Master of Science Degree in Chemical Engineering Supervisors: Professor Doctor Henrique Aníbal Santos de Matos Doctor Štepánˇ Špatenka Examination Committee Chairperson: Professor Doctor Carlos Manuel Faria de Barros Henriques Supervisor: Professor Doctor Henrique Aníbal Santos de Matos Member of the Committee: Specialist Engineer André Alexandre Bravo Ferreira Vilelas November 2015 ii The roots of education are bitter, but the fruit is sweet. – Aristotle All I am I owe to my mother. – George Washington iii iv Acknowledgments To begin with, my deepest thanks to Professor Carla Pinheiro, Professor Henrique Matos and Pro- fessor Costas Pantelides for allowing me to take this internship at Process Systems Enterprise Ltd., London, a seven-month truly worthy experience for both my professional and personal life which I will certainly never forget. I would also like to thank my PSE and IST supervisors, who help me to go through this final journey as a Chemical Engineering student. To Stˇ epˇ an´ and Sreekumar from PSE, thank you so much for your patience, for helping and encouraging me to always keep a positive attitude, even when harder problems arose. To Prof. Henrique who always showed availability to answer my questions and to meet in person whenever possible. Gostaria tambem´ de agradecer aos meus colegas de casa e de curso Andre,´ Frederico, Joana e Miguel, com quem partilhei casa. Foi uma experienciaˆ inesquec´ıvel que atravessamos´ juntos e cer- tamente que a vossa presenc¸a diaria´ apos´ cada dia de trabalho ajudou imenso a aliviar as saudades de casa. -

Hydrocarbons Thermal Cracking Selectivity Depending on Their Structure and Cracking Parameters
Master in Chemical Engineering Hydrocarbons Thermal Cracking Selectivity Depending on Their Structure and Cracking Parameters Thesis of the Master’s Degree Development Project in Foreign Environment Cláudia Sofia Martins Angeira Erasmus coordinator: Eng. Miguel Madeira Examiner in FEUP: Eng. Fernando Martins Supervisor in ICT: Doc. Ing Petr Zámostný July 2008 Acknowledgements I would like to thank Doc. Ing. Petr Zámostný for the opportunity to hold a master's thesis in this project, the orientation, the support given during the laboratory work and suggestions for improvement through the work. i Hydrocarbons Thermal Cracking Selectivity Depending on Their Structure and Cracking Parameters Abstract This research deals with the study of hydrocarbon thermal cracking with the aim of producing ethylene, one of the most important raw materials in Chemical Industry. The main objective was the study of cracking reactions of hydrocarbons by means of measuring the selectivity of hydrocarbons primary cracking and evaluating the relationship between the structure and the behavior. This project constitutes one part of a bigger project involving the study of more than 30 hydrocarbons with broad structure variability. The work made in this particular project was focused on the study of the double bond position effect in linear unsaturated hydrocarbons. Laboratory experiments were carried out in the Laboratory of Gas and Pyrolysis Chromatography at the Department of Organic Technology, Institute of Chemical Technology, Prague, using for all experiments the same apparatus, Pyrolysis Gas Chromatograph, to increase the reliability and feasibility of results obtained. Linear octenes with different double bond position in hydrocarbon chain were used as model compounds. In order to achieve these goals, the primary cracking reactions were studied by the method of primary selectivities. -

Metal Carbonyls
MODULE 1: METAL CARBONYLS Key words: Carbon monoxide; transition metal complexes; ligand substitution reactions; mononuclear carbonyls; dinuclear carbonyls; polynuclear carbonyls; catalytic activity; Monsanto process; Collman’s reagent; effective atomic number; 18-electron rule V. D. Bhatt / Selected topics in coordination chemistry / 2 MODULE 1: METAL CARBONYLS LECTURE #1 1. INTRODUCTION: Justus von Liebig attempted initial experiments on reaction of carbon monoxide with metals in 1834. However, it was demonstrated later that the compound he claimed to be potassium carbonyl was not a metal carbonyl at all. After the synthesis of [PtCl2(CO)2] and [PtCl2(CO)]2 reported by Schutzenberger (1868) followed by [Ni(CO)4] reported by Mond et al (1890), Hieber prepared numerous compounds containing metal and carbon monoxide. Compounds having at least one bond between carbon and metal are known as organometallic compounds. Metal carbonyls are the transition metal complexes of carbon monoxide containing metal-carbon bond. Lone pair of electrons are available on both carbon and oxygen atoms of carbon monoxide ligand. However, as the carbon atoms donate electrons to the metal, these complexes are named as carbonyls. A variety of such complexes such as mono nuclear, poly nuclear, homoleptic and mixed ligand are known. These compounds are widely studied due to industrial importance, catalytic properties and structural interest. V. D. Bhatt / Selected topics in coordination chemistry / 3 Carbon monoxide is one of the most important π- acceptor ligand. Because of its π- acidity, carbon monoxide can stabilize zero formal oxidation state of metals in carbonyl complexes. 2. SYNTHESIS OF METAL CARBONYLS Following are some of the general methods of preparation of metal carbonyls. -

The Action of Carbon Monoxide and Hydrogen on Deoxybenzoin Oxime and on 2-Acetonaphthone Oxime
THE ACTION OF CARBON MONOXIDE AND HYDROGEN ON DEOXYBENZOIN OXIME AND ON 2-ACETONAPHTHONE OXIME by Arthur Ronald Hubsoher A THESIS SUBMITTED IN PARTIAL FULFILMENT OF THE REQUIREMENTS POR THE DEGREE OP MASTER OF SCIENCE in the Department of Chemistry We accept this thesis as conforming to the required standard THE UNIVERSITY OF BRITISH COLUMBIA May, 1959 ABSTRACT When deoxybenzoin oxime was reacted with carbon monoxide and hydrogen in the presence of dicobalt ootacarbonyl at elevated temperatures and pressures 5-benzylphthalimidine and 3-phenyl-3,4- dihydroisocarbostyryl were produced. When 2-acetonaphthone oxime was reacted under similar conditions 2-(^-naphthyl)-4-methyl- benzo[h] quinoline, 2-methylbenzoff]phthalimidine and 1- (^-naphthyl)ethylurea were produced. Verification of the structure of 2-(^-naphthyl)-4-methylbenzo[h]- quinoline was attained in part through the hydro• chloride salt, the methiodide salt, the picrate derivative and the 2-(^-naphthyl)-4-formylbenzofh]- quinoline derivative. 2-(^-K.aphthyl )-4-methyl- benzofh] quinoline was also synthesized by reacting 2- acetonaphthone oxime with 2-acetonaphthone at elevated temperatures. The infrared spectra of the above compounds are described. In presenting this thesis in partial fulfilment of the requirements for an advanced degree at the University of British Columbia, I agree that the Library shall make it freely available for reference and study. I further agree that permission for extensive copying of this thesis for scholarly purposes may be granted by the Head of my Department or by his representatives. It is understood that copying or publication of this thesis for financial gain shall not be allowed without my written permission. -

Metal Catalyzed Outer Sphere Alkylations of Unactivated Olefins and Alkynes
Metal Catalyzed Outer Sphere Alkylations of Unactivated Olefins and Alkynes Stephen Goble Organic Super-Group Meeting Literature Presentation October 6, 2004 1 Outline I. Background • Introduction to Carbometallation • “Inner Sphere” vs. “Outer Sphere” • Review of Seminal Work II. Catalytic Palladium Systems III. Catalytic Platinum Systems IV. Catalytic Gold Systems 2 I. Background: Carbometallation is the formal addition of a metal and a carbon atom across a double bond. • Cis addition would involve olefin insertion in to an σ-alkyl-metal species. This is an “Inner Coordination Sphere” process. cis addition [M] [M] R olefin insertion R • The alkyl-metal species could arise from: 1. Oxidative addition of the metal to an electrophile. 2. Transmetallation of a nucleophile to the metal. 3. Direct nucleophilic attack on the palladium. • Trans addition would involve an “Outer Coordination Sphere” nucleophilic attack on the metal-olefin coordination complex. trans addition [M] [M] R- R 3 First Example: Carbopalladation of an Olefin Tsuji, J. and Takahashi, H. J. Am. Chem. Soc. 1965. 87(14), 3275-3276. [Pd] O O RO2C CO R PdCl2 RO OR 2 Na2CO3 [Pd] Cl • Amino and Oxo-palladations (i.e. Wacker Process) were previously known and have since been much more widely studied. • No distinction between cis and trans nucleophilic addition made. 4 Carbopalladation on Styrene: Different Modes of Attack Proposed different modes of attack based on different observed products with MeLi vs. Na-Malonate: Murahashi, S. et. al. J. Org. Chem. 1977. 42 (17), 2870-2874. Nucleophilic Attack on Pd Olefin Insertion B-Hydride Elimination CH3Li [Pd] [Pd] CH3 PdX2 CH3 [Pd]-H Inner Sphere Mechanism Palladium Coordination Nucleophilic Attack B-hydride elimination CO R CO2R 2 NaCH(CO2R) CO R [Pd] CO2R 2 PdX2 [Pd] [Pd]-H • Outer Sphere attack usually occurs at the more substituted carbon - more stabilization of + + charge (In intermolecular processes).