Mental Health Revolution
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Optumrx Brand Pipeline Forecast
RxOutlook® 1st Quarter 2019 OptumRx brand pipeline forecast Route of Regulatory Estimated Specialty Orphan Drug name Generic name Company Drug class Therapeutic use administration status release date drug drug 2019 Possible launch date Ophthalmological DS-300 DS-300 Eton undisclosed SC Filed NDA 2019 unknown N disease anti-sclerostin Evenity romosozumab Amgen Osteoporosis SC Filed NDA 2/2019 Y N monoclonal antibody tetrahydrofolate iclaprim iclaprim Motif Bio Bacterial infections IV Filed NDA 2/13/2019 Y Y dehydrogenase inhibitor tazarotene/ IDP-118 Valeant retinoid/ corticosteroid Psoriasis TOP Filed NDA 2/15/2019 N N halobetasol adenosine deaminase Mavenclad cladribine Merck/ Teva resistant Multiple sclerosis PO Filed NDA 2/15/2019 Y N deoxyadenosine analog Lotemax Gel loteprednol Valeant corticosteroid Ocular inflammation OP Filed NDA 2/25/2019 N N Nex Gen etabonate turoctocog alfa glyco-PEGylated factor NN-7088 Novo Nordisk Hemophilia IV/SC Filed BLA 2/27/2019 Y N pegol VIII derivative selective sphingosine-1 BAF-312 siponimod Novartis phosphate receptor Multiple sclerosis PO Filed NDA 3/1/2019 Y N agonist midazolam midazolam UCB benzodiazepine Seizures Intranasal Filed NDA 3/1/2019 N Y (USL-261) XeriSol glucagon Xeris glucagon analog Diabetes mellitus SC Filed NDA 3/1/2019 N N Glucagon optum.com/optumrx 1 RxOutlook® 1st Quarter 2019 Route of Regulatory Estimated Specialty Orphan Drug name Generic name Company Drug class Therapeutic use administration status release date drug drug dopamine receptor JZP-507 sodium oxybate Jazz Narcolepsy -

Pristiq (Desvenlafaxine Succinate) – First-Time Generic
Pristiq® (desvenlafaxine succinate) – First-time generic • On March 1, 2017, Teva launched AB-rated generic versions of Pfizer’s Pristiq (desvenlafaxine succinate) 25 mg, 50 mg, and 100 mg extended-release tablets for the treatment of major depressive disorder. — Teva launched the 25 mg tablet with 180-day exclusivity. — In addition, Alembic/Breckenridge, Mylan, and West-Ward have launched AB-rated generic versions of Pristiq 50 mg and 100 mg extended-release tablets. — Greenstone’s launch plans for authorized generic versions of Pristiq 25 mg, 50 mg, and 100 mg tablets are pending. — Lupin and Sandoz received FDA approval of AB-rated generic versions of Pristiq 50 mg and 100 mg tablets on June 29, 2015. Lupin’s and Sandoz’s launch plans are pending. • Other serotonin-norepinephrine reuptake inhibitors approved for the treatment of major depressive disorder include desvenlafaxine fumarate, duloxetine, Fetzima™ (levomilnacipran), Khedezla™ (desvenlafaxine) , venlafaxine, and venlafaxine extended-release. • Pristiq and the other serotonin-norepinephrine reuptake inhibitors carry a boxed warning for suicidal thoughts and behaviors. • According to IMS Health data, the U.S. sales of Pristiq were approximately $883 million for the 12 months ending on December 31, 2016. optumrx.com OptumRx® specializes in the delivery, clinical management and affordability of prescription medications and consumer health products. We are an Optum® company — a leading provider of integrated health services. Learn more at optum.com. All Optum® trademarks and logos are owned by Optum, Inc. All other brand or product names are trademarks or registered marks of their respective owners. This document contains information that is considered proprietary to OptumRx and should not be reproduced without the express written consent of OptumRx. -

Integrating Treatment for Autism Spectrum Disorders Through the Life Cycle
Integrating Treatment for Autism Spectrum Disorders Through the Life Cycle Robert L Hendren, DO Professor of Psychiatry and Behavioral Science Idaho Autism Summit November 2, 2019 Faculty Disclosure • Grants — Curemark, Roche, Otsuka • Advisory Board — Curemark, BioMarin, Janssen, Axial Biotherapeutics • Honoraria/Royalties: Oxford University Press, Taylor & Francis • Dr. Hendren does intend to discuss the use of off- label/unapproved use of drugs Learning Objectives • Identify successes and challenges in the developmental progression through the life cycle for people with developmental disabilities and their families • Identify and effectively treat comorbid medical, emotional and behavioral symptoms associated with autism spectrum disorders (ASD) • Consider integrating biomedical treatments for ASD including conventional psychotropic medication and what has been referred to as CAM/CIM into a comprehensive program. 2010 1 in 68; 2014 1 in 59 CDC Prevalence of Autism • Possible explanations include – Diagnostic expansion and substitution – Better reporting – Increased recognition – Increasing acceptability – Immigration for services – Environmental toxins – Infectious and immune vulnerability – Epigenetic processes Rutter M. Acta Pediatr. 2005;94(1):2-15. Centers for Disease Control and Prevention. Autism Spectrum Disorders. www.cdc.gov/ncbddd/autism. Accessed June 16, 2015. Hagerman R, Hendren RL (Eds). Treatment of Neurodevelopmental Disorders: Targeting Neurobiological Mechanisms. Oxford University Press; 2014. ASD Genetic Etiology (Levels 1 & 2) • Multiple genes: NRXN12q, 7q11.23, 15q11-13, 16p11.2, SHANK 3, 2, NLGN4, MTHFR 677>T, SEMA5A, 2Q22.1, GRIN2B, 5P14.1, CDH9, 10, FRX, PTEN • Identical twins: 60% to 90% – Fraternal twins: 0% to 36% – Siblings: 4% to 19% • Clear genetic etiology accounts for 25% of autism cases • Hundreds of genetic mutations, some de novo, lead to many ways to develop and treat autism • Is Precision Medicine Possible? Weiss KM, Issues Science and Technology in 2017 Levy D, et al. -

Pharmacological Management of Autism Spectrum Disorders
Pharmacological Management of Autism Spectrum Disorders Lightin the Way 2018 David Ermer Md Pharmacotherapy common in Autism Spectrum iIlnesses (ASDs) ◼ 45% of children and adolescents with ASDs treated with psychotropic medications ◼ 75% of adults with ASDs treated with psychotropic medications Research into Psychotropic Medications in ASD’s is Relatively New ◼ Before 2006 there were no FDA approved medications for use in Autism ◼ With the increased incidence of ASDs being reported, there is increased interest from the pharmaceutical industry Treatment Strategies for Pharmacologic Intervention ◼ Pharmacologic treatments are available and significantly beneficial ◼ Educational and behavioral supports are the mainstays of treatment ◼ It is essential to integrate behavioral and pharmacologic treatments Realistic Expectations Must be Set ◼ Expectation that symptoms remit more quickly with pharmacologic treatment over behavioral treatments ◼ Expectation that response will be more complete with pharmacologic treatments ◼ “magic bullets” Must Optimize Environment ◼ Evaluate school setting ◼ Work with caregivers on home environment ◼ Pharmacologic strategies ineffective in unsupportive and inadequate environments Parental Collaboration is Essential ◼ Monitor patient and provide information to provider ◼ Administer medication ◼ Observe side effects ◼ Note emotional and behavioral effects ◼ Collaborate their information with school Focus on Symptom Clusters ◼ Most of the core symptoms are likely to remain ◼ Must focus on specific measurable symptoms -

Desvenlafaxine
https://providers.amerigroup.com CONTAINS CONFIDENTIAL PATIENT INFORMATION desvenlafaxine Prior Authorization of Benefits (PAB) Form Complete form in its entirety and fax to: Prior Authorization of Benefits Center at 1-844-512-9004 Provider Help Desk 1-800-454-3730 1. PATIENT INFORMATION 2. PHYSICIAN INFORMATION Patient Name: Prescribing Physician: Patient ID #: Physician Address: Patient DOB: Physician Phone #: Date of Rx: Physician Fax #: Patient Phone #: Physician Specialty: Patient Email Address: Physician DEA: Physician NPI #: Physician Email Address: 3. MEDICATION 4. STRENGTH 5. DIRECTIONS 6. QUANTITY PER 30 DAYS desvenlafaxine Specify: 7. DIAGNOSIS: 8. APPROVAL CRITERIA: CHECK ALL BOXES THAT APPLY NOTE: Any areas not filled out are considered not applicable to your patient & MAY AFFECT THE OUTCOME of this request. □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with two preferred generic SSRIs* has been provided If No: □ Yes □ No Documented evidence is provided that the use of these agents would be medically contraindicated □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with one preferred generic SNRI** has been provided If No: □ Yes □ No Documented evidence is provided that the use of these agents would be medically contraindicated □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with one non-SSRI/SNRI generic antidepressant** has been provided If No: □ Yes □ No Documented evidence is provided that the use of these -

Psychotropic Medication Concerns When Treating Individuals with Developmental Disabilities
Tuesday, 2:30 – 4:00, C2 Psychotropic Medication Concerns when Treating Individuals with Developmental Disabilities Richard Berchou 248-613-6716 [email protected] Objective: Identify effective methods for the practical application of concepts related to improving the delivery of services for persons with developmental disabilities Notes: Medication Assistance On-Line Resources OBTAINING MEDICATION: • Needy Meds o Needymeds.com • Partnership for Prescriptions Assistance o Pparx.org • Patient Assistance Program Center o Rxassist.org • Insurance coverage & Prior authorization forms for most drug plans o Covermymeds.com REMINDERS TO TAKE MEDICATION: • Medication reminder by Email, Phone call, or Text message o Sugaredspoon.com ANSWER MOST QUESTIONS ABOUT MEDICATIONS: • Univ. of Michigan/West Virginia Schools of Pharmacy o Justaskblue.com • Interactions between medications, over-the-counter (OTC) products and some foods; also has a pictorial Pill Identifier: May input an entire list of medications o Drugs.com OTHER TRUSTED SITES: • Patient friendly information about disease and diagnoses o Mayoclinic.com, familydoctor.org • Package inserts, boxed warnings, “Dear Doctor” letters (can sign up to receive e- mail alerts) o Dailymed.nlm.nih.gov • Communications about drug safety o www.Fda.gov/cder/drug/drugsafety/drugindex.htm • Purchasing medications on-line o Pharnacychecker.com Updated 2013 Psychotropic Medication for Persons with Developmental Disabilities April 23, 2013 Richard Berchou, Pharm. D. Assoc. Clinical Prof., Dept. Psychiatry & Behavioral -
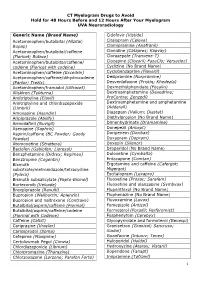
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology Generic Name (Brand Name) Cidofovir (Vistide) Acetaminophen/butalbital (Allzital; Citalopram (Celexa) Bupap) Clomipramine (Anafranil) Acetaminophen/butalbital/caffeine Clonidine (Catapres; Kapvay) (Fioricet; Butace) Clorazepate (Tranxene-T) Acetaminophen/butalbital/caffeine/ Clozapine (Clozaril; FazaClo; Versacloz) codeine (Fioricet with codeine) Cyclizine (No Brand Name) Acetaminophen/caffeine (Excedrin) Cyclobenzaprine (Flexeril) Acetaminophen/caffeine/dihydrocodeine Desipramine (Norpramine) (Panlor; Trezix) Desvenlafaxine (Pristiq; Khedezla) Acetaminophen/tramadol (Ultracet) Dexmethylphenidate (Focalin) Aliskiren (Tekturna) Dextroamphetamine (Dexedrine; Amitriptyline (Elavil) ProCentra; Zenzedi) Amitriptyline and chlordiazepoxide Dextroamphetamine and amphetamine (Limbril) (Adderall) Amoxapine (Asendin) Diazepam (Valium; Diastat) Aripiprazole (Abilify) Diethylpropion (No Brand Name) Armodafinil (Nuvigil) Dimenhydrinate (Dramamine) Asenapine (Saphris) Donepezil (Aricept) Aspirin/caffeine (BC Powder; Goody Doripenem (Doribax) Powder) Doxapram (Dopram) Atomoxetine (Strattera) Doxepin (Silenor) Baclofen (Gablofen; Lioresal) Droperidol (No Brand Name) Benzphetamine (Didrex; Regimex) Duloxetine (Cymbalta) Benztropine (Cogentin) Entacapone (Comtan) Bismuth Ergotamine and caffeine (Cafergot; subcitrate/metronidazole/tetracycline Migergot) (Pylera) Escitalopram (Lexapro) Bismuth subsalicylate (Pepto-Bismol) Fluoxetine (Prozac; Sarafem) -

Clinical Guideline Drug/Drug Class: Antipsychotics Prepared By
MassHealth Drug Utilization Review Program Commonwealth Medicine University of Massachusetts Medical School P.O. Box 2586 Worcester MA, 01613-2586 Clinical Guideline Drug/Drug Class: Antipsychotics Prepared by: Drug Utilization Review Program Prepared for: MassHealth Pharmacy Program Purpose: The purpose of this guideline is to clarify the procedures for approving and denying prior authorization (PA) requests for: Polypharmacy with two or more antipsychotics for members ≥ 18 years old (including first- generation [typical] and second-generation [atypical]) for greater than 60 days (excluding clozapine and injectable formulations) Orally disintegrating dosage forms and Versacloz® (clozapine) oral suspension Medication exceeding defined quantity limits Fanapt® (iloperidone), Invega® (paliperidone), Latuda® (lurasidone), Rexulti® (brexpiprazole), Saphris® (asenapine), and Vraylar® (cariprazine) for members of all ages and all quantities Abilify® (aripiprazole) and Seroquel XR® (quetiapine extended-release) for members 18 years of age and older and all quantities Background: Since 2003, MassHealth has determined that oral second-generation (atypical) antipsychotics (with the exception of clozapine and injectables) would require prior authorization for polypharmacy, defined as two or more second-generation (atypical) antipsychotics for greater than 60 days. Clozapine is excluded from the polypharmacy requirement because the guidelines for the treatment of schizophrenia recognize that combinations including clozapine have been reported. 1 In 2016, the adult antipsychotic polypharmacy criteria was updated and the PA restriction was expanded to include first-generation (typical), and second-generation (atypical) antipsychotics, excluding clozapine and injectable formulations. Orally disintegrating tablets (ODT) of aripiprazole, clozapine, olanzapine and risperidone, as well as Versacloz® (clozapine) oral suspension also require PA since there are more cost-effective alternatives available. -

Amoxapine | Memorial Sloan Kettering Cancer Center
PATIENT & CAREGIVER EDUCATION Amoxapine This information from Lexicomp® explains what you need to know about this medication, including what it’s used for, how to take it, its side effects, and when to call your healthcare provider. Warning Drugs like this one have raised the chance of suicidal thoughts or actions in children and young adults. The risk may be greater in people who have had these thoughts or actions in the past. All people who take this drug need to be watched closely. Call the doctor right away if signs like low mood (depression), nervousness, restlessness, grouchiness, panic attacks, or changes in mood or actions are new or worse. Call the doctor right away if any thoughts or actions of suicide occur. This drug is not approved for use in children. Talk with the doctor. What is this drug used for? It is used to treat low mood (depression). It may be given to you for other reasons. Talk with the doctor. What do I need to tell my doctor BEFORE I take this drug? If you are allergic to this drug; any part of this drug; or any other drugs, foods, or substances. Tell your doctor about the allergy and what signs you had. If you have had a recent heart attack. If you have taken certain drugs for depression or Parkinson’s disease in the last 14 days. This includes isocarboxazid, phenelzine, tranylcypromine, Amoxapine 1/7 selegiline, or rasagiline. Very high blood pressure may happen. If you are taking any of these drugs: Linezolid or methylene blue. -

Antidepressants, Other Review 04/14/2009
Antidepressants, Other Review 04/14/2009 Copyright © 2004 - 2009 by Provider Synergies, L.L.C. All rights reserved. Printed in the United States of America. All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopying, recording, digital scanning, or via any information storage and retrieval system without the express written consent of Provider Synergies, L.L.C. All requests for permission should be mailed to: Attention: Copyright Administrator Intellectual Property Department Provider Synergies, L.L.C. 5181 Natorp Blvd., Suite 205 Mason, Ohio 45040 The materials contained herein represent the opinions of the collective authors and editors and should not be construed to be the official representation of any professional organization or group, any state Pharmacy and Therapeutics committee, any state Medicaid Agency, or any other clinical committee. This material is not intended to be relied upon as medical advice for specific medical cases and nothing contained herein should be relied upon by any patient, medical professional or layperson seeking information about a specific course of treatment for a specific medical condition. All readers of this material are responsible for independently obtaining medical advice and guidance from their own physician and/or other medical professional in regard to the best course of treatment for their specific medical condition. This publication, inclusive of all forms contained herein, is intended to be -

Still the Leading Antidepressant After 40 Years of Randomised Controlle
BRITISH JOURNAL OF PSYCHIATRY "2001), 178, 129^144 REVIEW ARTICLE Amitriptyline vv.therest:stilltheleading METHOD Inclusion criteria antidepressant after 40 years of randomised All RCTs comparing amitriptyline with any y other tricyclic,heterocyclic or SSRI were in- controlled trials cluded. Crossover studies were excluded. Studies adopting any criteria to define CORRADO BARBUI and MATTHEW HOTOPF patients suffering from depression were included; a concurrent diagnosis of another psychiatric disorder was not considered an exclusion criterion. Trials in patients with depression with a concomitant medical ill- Background Tricyclic antidepressants Amitriptyline is one of the first `reference' ness were not included in this review. have similar efficacy and slightly lower tricyclic antidepressants TCAs). Over the past 40 years a number of newer tricyclics, tolerability than selective serotonin Search strategy heterocyclics and selective serotonin re- Relevant studies were located by searching reuptakeinhibitorsreuptake inhibitors SSRIs).However, uptake inhibitors SSRIs) have been intro- the Cochrane Collaboration Depression, there are no systematic reviews assessing duced Garattini et aletal,1998). Despite Anxiety and Neurosis Controlled Trials several large systematic reviews comparing amitriptyline, the reference tricyclic drug, Register CCDANCTR). This specialised tricyclics and SSRIs there is no clear agree- vv. other tricyclics and SSRIs directly. register is regularly updated by electronic ment over first-line treatment of depression Medline,Embase,PsycINFO,LILACS, SongSong et aletal,1993; Anderson & Tomenson, Aims ToreviewTo review the tolerability and Psyndex,CINAHL,SIGLE) and non-electro- 1995; Montgomery & Kasper,1995; efficacy of amitriptyline inthe nicnicliterature searches. The register was HotopfHotopf et aletal,1996; Canadian Coordinating management of depression. searched using the following terms: Office for Health Technology Assessment, AMITRIPTYLIN**AMITRIPTYLIN oror AMITRILAMITRIL oror ELA-ELA- 19971997aa). -

Desvenlafaxine Drug Assessment Pristiq® in Major Depressive Report Disorder in Adults @DTB Navarre.Es More Cost for Less
03 2015 DESVENLAFAXINE DRUG ASSESSMENT Pristiq® in major depressive REPORT www.dtb.navarra.es disorder in adults @DTB_Navarre.es More cost for less... ABSTRACT Desvenlafaxine is an active me- tabolite of venlafaxine. Indications1 In the study against escitalopram4 flexible Treatment of major depressive disorder doses of desvenlafaxine were used (100 and In three placebo-controlled 200 mg daily) in postmenopausal women. No trials, the results on the reduc- 1 Mechanism of action advantage of desvenlafaxine over escitalopram tion in HAM-D17 score were not This is an active metabolite of venlafaxine, that was found. consistent. inhibits the reuptake of serotonine and nora- There is only one long-term study16 that eva- dernaline. Its bioavailability reaches up to 80% luated relapse prevention. Patients responding The most common adverse while elimination occurs without alteration after 8 weeks of treatment with desvenlafaxine effects are of gastrointestinal through the urine (45%) and metabolism by 50 mg daily and with a stable response up to origin or sleep disorders. There glucurono conjugation (19%). week 20 were randomized either to placebo or are no available long-term safe- desvenlafaxine 50 mg daily for 6 months. The ty data. Dosage and administration1 endpoint was time to relapse (defined as HAM- The recommended dose is 50 mg daily. The ta- D17 score ≥16), treatment withdrawal due to In the only head-to-head trial blets are swallowed wholly, with liquid with or unsatisfactory response, hospital admission carried out in post-menopause without food and at the same time. The increa- due to depression, suicide attempt or suicide. women, desvenlafaxine at high se in doses should be gradual and up to a maxi- Time to relapse was significantly lower in the doses did not show superiority mum of 200 mg daily and at intervals of at least case of placebo compared to desvenlafaxine versus escitalopram.