PPAR-Mediated Toxicology and Applied Pharmacology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Analysis of Estrogen Receptor Isoforms and Variants in Breast Cancer Cell Lines
EXPERIMENTAL AND THERAPEUTIC MeDICINE 2: 537-544, 2011 Analysis of estrogen receptor isoforms and variants in breast cancer cell lines MAIE AL-BADER1, CHRISTOPHER FORD2, BUSHRA AL-AYADHY3 and ISSAM FRANCIS3 Departments of 1Physiology, 2Surgery, and 3Pathology, Faculty of Medicine, Kuwait University, Safat 13110, Kuwait Received November 22, 2010; Accepted February 14, 2011 DOI: 10.3892/etm.2011.226 Abstract. In the present study, the expression of estrogen domain C, the DNA binding domain; domains D/E, bearing receptor (ER)α and ERβ isoforms in ER-positive (MCF7, both the activation function-2 (AF-2) and the ligand binding T-47D and ZR-75-1) and ER-negative (MDA-MB-231, SK-BR-3, domains; and finally, domain F, the C-terminal domain (6,7). MDA-MB-453 and HCC1954) breast cancer cell lines was The actions of estrogens are mediated by binding to ERs investigated. ERα mRNA was expressed in ER-positive and (ERα and/or ERβ). These receptors, which are co-expressed some ER-negative cell lines. ERα ∆3, ∆5 and ∆7 spliced in a number of tissues, form functional homodimers or variants were present in MCF7 and T-47D cells; ERα ∆5 heterodimers. When bound to estrogens as homodimers, the and ∆7 spliced variants were detected in ZR-75-1 cells. transcription of target genes is activated (8,9), while as heterodi- MDA-MB-231 and HCC1954 cells expressed ERα ∆5 and ∆7 mers, ERβ exhibits an inhibitory action on ERα-mediated gene spliced variants. The ERβ1 variant was expressed in all of the expression and, in many instances, opposes the actions of ERα cell lines and the ERβ2 variant in all of the ER-positive and (7,9). -

Chemicals in the Fourth Report and Updated Tables Pdf Icon[PDF
Chemicals in the Fourth National Report on Human Exposure to Environmental Chemicals: Updated Tables, March 2021 CDC’s Fourth National Report on Human Exposure to Environmental Chemicals: Updated Tables, March 2021 provides exposure data on the following chemicals or classes of chemicals. The Updated Tables contain cumulative data from national samples collected beginning in 1999–2000 and as recently as 2015-2016. Not all chemicals were measured in each national sample. The data tables are available at https://www.cdc.gov/exposurereport. An asterisk (*) indicates the chemical has been added since publication of the Fourth National Report on Human Exposure to Environmental Chemicals in 2009. Adducts of Hemoglobin Acrylamide Formaldehyde* Glycidamide Tobacco Alkaloids and Metabolites Anabasine* Anatabine* Cotinine Cotinine-n-oxide* Hydroxycotinine* Trans-3’-hydroxycotinine* 1-(3-Pyridyl)-1-butanol-4-carboxylic acid* Nicotine* Nicotine-N’-oxide* Nornicotine* Tobacco-Specific Nitrosamines (TSNAs) N’-Nitrosoanabasine (NAB)* N’-Nitrosoanatabine (NAT)* N’-Nitrosonornicotine (NNN)* Total 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol) (NNAL)* Volatile N-nitrosamines (VNAs) N-Nitrosodiethylamine (NDEA)* N-Nitrosoethylmethylamine (NMEA)* N-Nitrosomorpholine (NMOR)* N-Nitrosopiperidine (NPIP)* N-Nitrosopyrrolidine (NPYR)* Disinfection By-Products Bromodichloromethane Dibromochloromethane Tribromomethane (Bromoform) Trichloromethane (Chloroform) Personal Care and Consumer Product Chemicals and Metabolites Benzophenone-3 Bisphenol A Bisphenol F* Bisphenol -

The Antidiabetic Drug Lobeglitazone Has the Potential to Inhibit PTP1B T Activity ⁎ Ruth F
Bioorganic Chemistry 100 (2020) 103927 Contents lists available at ScienceDirect Bioorganic Chemistry journal homepage: www.elsevier.com/locate/bioorg The antidiabetic drug lobeglitazone has the potential to inhibit PTP1B T activity ⁎ Ruth F. Rochaa, Tiago Rodriguesc, Angela C.O. Menegattia,b, , Gonçalo J.L. Bernardesc,d, Hernán Terenzia a Centro de Biologia Molecular Estrutural, Departamento de Bioquímica, Universidade Federal de Santa Catarina, Campus Trindade, 88040-900 Florianópolis, SC, Brazil b Universidade Federal do Piauí, CPCE, 64900-000 Bom Jesus, PI, Brazil c Instituto de Medicina Molecular, Faculdade de Medicina, Universidade de Lisboa, Avenida Professor Egas Moniz, 1649-028 Lisbon, Portugal d Department of Chemistry, University of Cambridge, Lensfield Road, CB2 1EW Cambridge, UK ARTICLE INFO ABSTRACT Keywords: Protein tyrosine phosphatase 1B (PTP1B) is considered a potential therapeutic target for the treatment of type 2 Thiazolidinediones diabetes mellitus (T2DM), since this enzyme plays a significant role to down-regulate insulin and leptin sig- Lobeglitazone nalling and its over expression has been implicated in the development of insulin resistance, T2DM and obesity. PPAR-γ Some thiazolidinediones (TZD) derivatives have been reported as promising PTP1B inhibitors with anti hy- PTP1B perglycemic effects. Recently, lobeglitazone, a new TZD, was described as an antidiabetic drug that targetsthe Non-competitive inhibitors PPAR-γ (peroxisome γ proliferator-activated receptor) pathway, but no information on its effects on PTP1B have been reported to date. We investigated the effects of lobeglitazone on PTP1B activity in vitro. Surprisingly, lobeglitazone led to moderate inhibition on PTP1B (IC50 42.8 ± 3.8 µM) activity and to a non-competitive reversible mechanism of action. -

Telmisartan Use in Rats with Preexisting Osteoporotics Bone
Life Sciences 237 (2019) 116890 Contents lists available at ScienceDirect Life Sciences journal homepage: www.elsevier.com/locate/lifescie Telmisartan use in rats with preexisting osteoporotics bone disorders T increases bone microarchitecture alterations via PPARγ Antonio Marcos Birocalea, Antonio Ferreira de Melo Jr.b, Pollyana Peixotob, Phablo Wendell Costalonga Oliveirab, Leandro Dias Gonçalves Ruffonic, Liliam Masako Takayamad, Breno Valentim Nogueirae, Keico Okino Nonakac, ∗ Rosa Maria Rodrigues Pereirad, José Martins de Oliveira Jr.f, Nazaré Souza Bissolib, a Department of Health Integrated Education, Federal University of Espirito Santo, Vitória, ES, Brazil b Department of Physiological Sciences, Federal University of Espirito Santo, Vitória, ES, Brazil c Department of Physiological Sciences, Federal University of São Carlos, São Carlos, SP, Brazil d Department of Medical Clinic, Medicine College, University of São Paulo, São Paulo, SP, Brazil e Department of Morphology, Federal University of Espirito Santo, Vitoria, ES, Brazil f Laboratory of Applied Nuclear Physics, University of Sorocaba, Sorocaba, SP, Brazil ARTICLE INFO ABSTRACT Keywords: Aims: Telmisartan (TEL), an angiotensin II type I receptor blocker and PPARγ partial agonist, has been used for Telmisartan to treat hypertension. It is known that PPARγ activation induces bone loss. Therefore, we evaluate the effects of PPARγ telmisartan on PPARγ protein expression, biomechanics, density and bone microarchitecture of femurs and Hypertension lumbar vertebrae in SHR ovariectomized animals, a model of hypertension in which preexisting bone impair- Ovariectomy ment has been demonstrated. Bone Main methods: SHR females (3 months old) were distributed into four groups: sham (S), sham + TEL (ST), OVX (C) and OVX + TEL (CT). TEL (5 mg/kg/day) or vehicle were administered according to the groups. -

Dualism of Peroxisome Proliferator-Activated Receptor Α/Γ: a Potent Clincher in Insulin Resistance
AEGAEUM JOURNAL ISSN NO: 0776-3808 Dualism of Peroxisome Proliferator-Activated Receptor α/γ: A Potent Clincher in Insulin Resistance Mr. Ravikumar R. Thakar1 and Dr. Nilesh J. Patel1* 1Faculty of Pharmacy, Shree S. K. Patel College of Pharmaceutical Education & Research, Ganpat University, Gujarat, India. [email protected] Abstract: Diabetes mellitus is clinical syndrome which is signalised by augmenting level of sugar in blood stream, which produced through lacking of insulin level and defective insulin activity or both. As per worldwide epidemiology data suggested that the numbers of people with T2DM living in developing countries is increasing with 80% of people with T2DM. Peroxisome proliferator-activated receptors are a family of ligand-activated transcription factors; modulate the expression of many genes. PPARs have three isoforms namely PPARα, PPARβ/δ and PPARγ that play a central role in regulating glucose, lipid and cholesterol metabolism where imbalance can lead to obesity, T2DM and CV ailments. It have pathogenic role in diabetes. PPARα is regulates the metabolism of lipids, carbohydrates, and amino acids, activated by ligands such as polyunsaturated fatty acids, and drugs used as Lipid lowering agents. PPAR β/δ could envision as a therapeutic option for the correction of diabetes and a variety of inflammatory conditions. PPARγ is well categorized, an element of the PPARs, also pharmacological effective as an insulin resistance lowering agents, are used as a remedy for insulin resistance integrated with type- 2 diabetes mellitus. There are mechanistic role of PPARα, PPARβ/δ and PPARγ in diabetes mellitus and insulin resistance. From mechanistic way, it revealed that dual PPAR-α/γ agonist play important role in regulating both lipids as well as glycemic levels with essential safety issues. -

Combination Treatment with Pioglitazone and Fenofibrate
179 Combination treatment with pioglitazone and fenofibrate attenuates pioglitazone-mediated acceleration of bone loss in ovariectomized rats Rana Samadfam, Malaika Awori, Agnes Be´nardeau1, Frieder Bauss2, Elena Sebokova1, Matthew Wright1 and Susan Y Smith Charles River Laboratories, 22022 Transcanadienne, Senneville, Montre´al, Que´bec, Canada H9X 3R3 1F. Hoffmann-La Roche AG, Basel, CH-4070 Switzerland 2Roche Diagnostics GmbH, Penzberg, DE-82377 Germany (Correspondence should be addressed to S Y Smith; Email: [email protected]) Abstract Peroxisome proliferator-activated receptor (PPAR) g ago- mineral content (w45%) and bone mineral density (BMD; nists, such as pioglitazone (Pio), improve glycemia and lipid w60%) at the lumbar spine. Similar effects of treatments were profile but are associated with bone loss and fracture risk. Data observed at the femur, most notably at sites rich in trabecular regarding bone effects of PPARa agonists (including bone. At the proximal tibial metaphysis, concomitant fenofibrate (Feno)) are limited, although animal studies treatment with PioCFeno prevented Pio exacerbation of suggest that Feno may increase bone mass. This study ovariectomy-induced loss of trabecular bone, resulting in investigated the effects of a 13-week oral combination BMD values in the PioCFeno group comparable to OVX treatment with Pio (10 mg/kg per day)CFeno (25 mg/kg controls. Discontinuation of Pio or Feno treatment of per day) on body composition and bone mass parameters OVX rats was associated with partial reversal of effects on compared with Pio or Feno alone in adult ovariectomized bone loss or bone mass gain, respectively, while values in the (OVX) rats, with a 4-week bone depletion period, followed PioCFeno group remained comparable to OVX controls. -

Comparative Transcriptional Network Modeling of Three PPAR-A/C Co-Agonists Reveals Distinct Metabolic Gene Signatures in Primary Human Hepatocytes
Comparative Transcriptional Network Modeling of Three PPAR-a/c Co-Agonists Reveals Distinct Metabolic Gene Signatures in Primary Human Hepatocytes Rene´e Deehan1, Pia Maerz-Weiss2, Natalie L. Catlett1, Guido Steiner2, Ben Wong1, Matthew B. Wright2*, Gil Blander1¤a, Keith O. Elliston1¤b, William Ladd1, Maria Bobadilla2, Jacques Mizrahi2, Carolina Haefliger2, Alan Edgar{2 1 Selventa, Cambridge, Massachusetts, United States of America, 2 F. Hoffmann-La Roche AG, Basel, Switzerland Abstract Aims: To compare the molecular and biologic signatures of a balanced dual peroxisome proliferator-activated receptor (PPAR)-a/c agonist, aleglitazar, with tesaglitazar (a dual PPAR-a/c agonist) or a combination of pioglitazone (Pio; PPAR-c agonist) and fenofibrate (Feno; PPAR-a agonist) in human hepatocytes. Methods and Results: Gene expression microarray profiles were obtained from primary human hepatocytes treated with EC50-aligned low, medium and high concentrations of the three treatments. A systems biology approach, Causal Network Modeling, was used to model the data to infer upstream molecular mechanisms that may explain the observed changes in gene expression. Aleglitazar, tesaglitazar and Pio/Feno each induced unique transcriptional signatures, despite comparable core PPAR signaling. Although all treatments inferred qualitatively similar PPAR-a signaling, aleglitazar was inferred to have greater effects on high- and low-density lipoprotein cholesterol levels than tesaglitazar and Pio/Feno, due to a greater number of gene expression changes in pathways related to high-density and low-density lipoprotein metabolism. Distinct transcriptional and biologic signatures were also inferred for stress responses, which appeared to be less affected by aleglitazar than the comparators. In particular, Pio/Feno was inferred to increase NFE2L2 activity, a key component of the stress response pathway, while aleglitazar had no significant effect. -
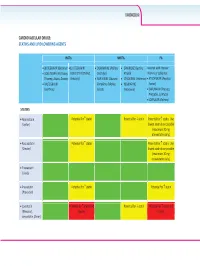
Statins and Lipid Lowering Agents
CARDIOVASCULAR Z/Ks^h>ZZh'^͗ ^dd/E^E>/W/>KtZ/E''Ed^ /E^d/Ɛ EEZd/Ɛ W/Ɛ x/d'Zs/Z ;ŝŬƚĂƌǀLJͿ x>s/d'Zs/Zͬ x KZs/Z/E ;WŝĨĞůƚƌŽ͕ x &s/ZE ;^ƵƐƚŝǀĂ͕ ŽŽƐƚĞĚǁŝƚŚƌŝƚŽŶĂǀŝƌ xK>hd'Zs/Z ;dŝǀŝĐĂLJ͕ K//^dd ;^ƚƌŝďŝůĚ͕ ĞůƐƚƌŝŐŽͿ ƚƌŝƉůĂͿ ;EŽƌǀŝƌͿŽƌĐŽďŝĐŝƐƚĂƚ dƌŝƵŵĞƋ͕:ƵůƵĐĂ͕ŽǀĂƚŽͿ 'ĞŶǀŽLJĂͿ x Z/>W/s/Z/E ;ĚƵƌĂŶƚ͕ x dZs/Z/E ;/ŶƚĞůĞŶĐĞͿ xdEs/Z ;ZĞLJĂƚĂnj͕ xZ>d'Zs/Z ŽŵƉůĞƌĂ͕KĚĞĨƐĞLJ͕ x Es/ZW/E ǀŽƚĂnjͿ ;/ƐĞŶƚƌĞƐƐͿ :ƵůƵĐĂͿ ;sŝƌĂŵƵŶĞͿ xZhEs/Z ;WƌĞnjŝƐƚĂ͕ WƌĞnjĐŽďŝdž͕^LJŵƚƵnjĂͿ x>KW/Es/Z ;<ĂůĞƚƌĂͿ ^dd/E^ xƚŽƌǀĂƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ pƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ͘hƐĞ ;>ŝƉŝƚŽƌͿ ůŽǁĞƐƚƐƚĂƚŝŶĚŽƐĞƉŽƐƐŝďůĞ ;ŵĂdžŝŵƵŵϮϬŵŐ ĂƚŽƌǀĂƐƚĂƚŝŶĚĂŝůLJͿ͘ xZŽƐƵǀĂƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ͘hƐĞ ;ƌĞƐƚŽƌͿ ůŽǁĞƐƚƐƚĂƚŝŶĚŽƐĞƉŽƐƐŝďůĞ ;ŵĂdžŝŵƵŵϭϬŵŐ ƌŽƐƵǀĂƐƚĂƚŝŶĚĂŝůLJͿ͘ xWŝƚĂǀĂƐƚĂƚŝŶ ;>ŝǀĂůŽͿ xWƌĂǀĂƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ ;WƌĂǀĂĐŚŽůͿ x>ŽǀĂƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶĂŶĚ WŽƚĞŶƚŝĂůĨŽƌ pƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶĂŶĚ ;DĞǀĂĐŽƌͿ͕ ƚŽdžŝĐŝƚLJ ƚŽdžŝĐŝƚLJ ƐŝŵǀĂƐƚĂƚŝŶ ;ŽĐŽƌͿ CARDIOVASCULAR INSTIs NNRTIs PIs xBICTEGRAVIR (Biktarvy) xELVITEGRAVIR/ x DORAVIRINE (Pifeltro, x EFAVIRENZ (Sustiva, Boosted with ritonavir xDOLUTEGRAVIR (Tivicay, COBICISTAT (Stribild, Delstrigo) Atripla) (Norvir) or cobicistat Triumeq, Juluca) Genvoya) x RILPIVIRINE (Edurant, x ETRAVIRINE (Intelence) xATAZANAVIR (Reyataz, xRALTEGRAVIR Complera, Odefsey, x NEVIRAPINE Evotaz) (Isentress) Juluca) (Viramune) xDARUNAVIR (Prezista, Prezcobix, Symtuza) xLOPINAVIR (Kaletra) FIBRATES xFenofibrate, bezafibrate, gemfibrozil CHOLESTEROL ABSORPTION INHIBITOR xEzetimibe -

Estrogen Receptor a Binds to Peroxisome Proliferator ^ Activated
Human Cancer Biology Estrogen Receptor A Binds to Peroxisome Proliferator ^ Activated Receptor Response Element and Negatively Interferes with Peroxisome Proliferator ^ Activated Receptor ; Signaling in Breast Cancer Cells Daniela Bonofiglio,1Sabrina Gabriele,1Saveria Aquila,1Stefania Catalano,1Mariaelena Gentile,2 Emilia Middea,1Francesca Giordano,2 and Sebastiano Ando' 2,3 Abstract Purpose: The molecular mechanisms involved in the repressive effects exerted by estrogen receptors (ER) on peroxisome proliferator ^ activated receptor (PPAR) g^ mediated transcriptional activity remain to be elucidated. The aim of the present study was to provide new insight into the crosstalk between ERa and PPARg pathways in breast cancer cells. Experimental Design: Using MCF7 and HeLa cells as model systems, we did transient trans- fections and electrophoretic mobility shift assay and chromatin immunoprecipitation studies to evaluate the ability of ERa to influence PPAR response element ^ mediated transcription. A pos- sible direct interaction between ERa and PPARg was ascertained by coimmunoprecipitation assay, whereas their modulatory role in the phosphatidylinositol 3-kinase (PI3K)/AKT pathway was evaluated by determining PI3K activity and AKT phosphorylation. As a biological counter- part, we investigated the growth response to the cognate ligands of both receptors in hormone-dependent MCF7 breast cancer cells. Results:Our data show for the first time that ERa binds to PPAR response element and represses its transactivation. Moreover, we have documented the physical and functional interactions of ERa and PPARg, which also involve the p85 regulatory subunit of PI3K. Interestingly, ERa and PPARg pathways have an opposite effect on the regulation of the PI3K/AKT transduction cascade, explaining, at least in part, the divergent response exerted by the cognate ligands 17 h-estradiol and BRL49653on MCF7 cell proliferation. -

Classification Decisions Taken by the Harmonized System Committee from the 47Th to 60Th Sessions (2011
CLASSIFICATION DECISIONS TAKEN BY THE HARMONIZED SYSTEM COMMITTEE FROM THE 47TH TO 60TH SESSIONS (2011 - 2018) WORLD CUSTOMS ORGANIZATION Rue du Marché 30 B-1210 Brussels Belgium November 2011 Copyright © 2011 World Customs Organization. All rights reserved. Requests and inquiries concerning translation, reproduction and adaptation rights should be addressed to [email protected]. D/2011/0448/25 The following list contains the classification decisions (other than those subject to a reservation) taken by the Harmonized System Committee ( 47th Session – March 2011) on specific products, together with their related Harmonized System code numbers and, in certain cases, the classification rationale. Advice Parties seeking to import or export merchandise covered by a decision are advised to verify the implementation of the decision by the importing or exporting country, as the case may be. HS codes Classification No Product description Classification considered rationale 1. Preparation, in the form of a powder, consisting of 92 % sugar, 6 % 2106.90 GRIs 1 and 6 black currant powder, anticaking agent, citric acid and black currant flavouring, put up for retail sale in 32-gram sachets, intended to be consumed as a beverage after mixing with hot water. 2. Vanutide cridificar (INN List 100). 3002.20 3. Certain INN products. Chapters 28, 29 (See “INN List 101” at the end of this publication.) and 30 4. Certain INN products. Chapters 13, 29 (See “INN List 102” at the end of this publication.) and 30 5. Certain INN products. Chapters 28, 29, (See “INN List 103” at the end of this publication.) 30, 35 and 39 6. Re-classification of INN products. -

Clinofibrate Improved Canine Lipid Metabolism in Some but Not All Breeds
NOTE Internal Medicine Clinofibrate improved canine lipid metabolism in some but not all breeds Yohtaro SATO1), Nobuaki ARAI2), Hidemi YASUDA3) and Yasushi MIZOGUCHI4)* 1)Graduate School of Agriculture, Meiji University, 1-1-1 Higashimita, Tama-ku, Kawasaki, Kanagawa 214-8571, Japan 2)Spectrum Lab Japan, 1-5-22-201 Midorigaoka, Meguro-ku, Tokyo 152-0034, Japan 3)Yasuda Veterinary Clinic, 1-5-22 Midorigaoka, Meguro-ku, Tokyo 152-0034, Japan 4)School of Agriculture, Meiji University, 1-1-1 Higashimita, Tama-ku, Kawasaki, Kanagawa 214-8571, Japan ABSTRACT. The objectives of this study were to assess if Clinofibrate (CF) treatment improved J. Vet. Med. Sci. lipid metabolism in dogs, and to clarify whether its efficacy is influenced by canine characteristics. 80(6): 945–949, 2018 We collected medical records of 306 dogs and performed epidemiological analyses. Lipid values of all lipoproteins were significantly decreased by CF medication, especially VLDL triglyceride doi: 10.1292/jvms.17-0703 (TG) concentration (mean reduction rate=54.82%). However, 17.65% of dogs showed drug refractoriness in relation to TG level, and Toy Poodles had a lower CF response than other breeds (OR=5.36, 95% CI=2.07–13.90). Therefore, our study suggests that genetic factors may have an Received: 22 December 2017 effect on CF response, so genetic studies on lipid metabolism-related genes might be conducted Accepted: 9 March 2018 to identify variations in CF efficacy. Published online in J-STAGE: KEY WORDS: clinofibrate, descriptive epidemiology, drug response, dyslipidemia, Toy Poodle 26 March 2018 High serum cholesterol (Cho) and triglyceride (TG) concentrations in dogs are caused by various factors such as lack of exercise, high fat diets, obesity, neutralization, age, diseases and breed [6, 21, 24]. -

Conditional Expression of Human Pparδ and a Dominant Negative Variant of Hpparδ in Vivo
Hindawi Publishing Corporation PPAR Research Volume 2012, Article ID 216817, 12 pages doi:10.1155/2012/216817 Research Article Conditional Expression of Human PPARδ and a Dominant Negative Variant of hPPARδ In Vivo Larry G. Higgins,1 Wojciech G. Garbacz,1 Mattias C. U. Gustafsson,2 Sitheswaran Nainamalai,3 Peter R. Ashby,1 C. Roland Wolf,1 and Colin N. A. Palmer1 1 CRUK Molecular Pharmacology Unit, Medical Research Institute, University of Dundee, Ninewells Hospital and Medical School, Dundee DD1 9SY, UK 2 Department of Laboratory Medicine, Division of Medical Microbiology, Lund University, Solvegatan¨ 23, SE-223 62 Lund, Sweden 3 Division of Molecular Medicine, Medical Science Institute, College of Life Sciences, University of Dundee, Dundee DD1 4HN, UK Correspondence should be addressed to Colin N. A. Palmer, [email protected] Received 21 September 2011; Revised 7 December 2011; Accepted 20 December 2011 Academic Editor: James P. Hardwick Copyright © 2012 Larry G. Higgins et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. The nuclear receptor, NR1C2 or peroxisome proliferator-activated receptor (PPAR)-δ, is ubiquitously expressed and important for placental development, fatty acid metabolism, wound healing, inflammation, and tumour development. PPARδ has been hypothesized to function as both a ligand activated transcription factor and a repressor of transcription in the absence of agonist. In this paper, treatment of mice conditionally expressing human PPARδ with GW501516 resulted in a marked loss in body weight that was not evident in nontransgenic animals or animals expressing a dominant negative derivative of PPARδ.