Unveiling the Structural Basis for Translational Ambiguity Tolerance in a Human Fungal Pathogen
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Patients Experiencing Statin-Induced Myalgia Exhibit a Unique Program of Skeletal Muscle Gene Expression Following Statin Re-Challenge
RESEARCH ARTICLE Patients experiencing statin-induced myalgia exhibit a unique program of skeletal muscle gene expression following statin re-challenge Marshall B. Elam1,2,3☯*, Gipsy Majumdar1,2, Khyobeni Mozhui4, Ivan C. Gerling1,3, Santiago R. Vera1, Hannah Fish-Trotter3¤, Robert W. Williams5, Richard D. Childress1,3, Rajendra Raghow1,2☯* 1 Department of Veterans Affairs Medical Center-Memphis, Memphis, Tennessee, United States of America, 2 Department of Pharmacology, University of Tennessee Health Sciences Center, Memphis, Tennessee, a1111111111 United States of America, 3 Department of Medicine, University of Tennessee Health Sciences Center, a1111111111 Memphis, Tennessee, United States of America, 4 Department of Preventive Medicine, University of a1111111111 Tennessee Health Sciences Center, Memphis, Tennessee, United States of America, 5 Department of a1111111111 Genetics, Genomics and Informatics, College of Medicine, University of Tennessee Health Science Center, a1111111111 Memphis, Tennessee, United States of America ☯ These authors contributed equally to this work. ¤ Current address: Department of Rehabilitation Medicine, Vanderbilt University, Nashville, Tennessee, United States of America. * [email protected] (MBE); [email protected] (RR) OPEN ACCESS Citation: Elam MB, Majumdar G, Mozhui K, Gerling IC, Vera SR, Fish-Trotter H, et al. (2017) Patients Abstract experiencing statin-induced myalgia exhibit a unique program of skeletal muscle gene Statins, the 3-hydroxy-3-methyl-glutaryl (HMG)-CoA reductase inhibitors, are widely pre- expression following statin re-challenge. PLoS ONE scribed for treatment of hypercholesterolemia. Although statins are generally well tolerated, 12(8): e0181308. https://doi.org/10.1371/journal. pone.0181308 up to ten percent of statin-treated patients experience myalgia symptoms, defined as mus- cle pain without elevated creatinine phosphokinase (CPK) levels. -

Location Analysis of Estrogen Receptor Target Promoters Reveals That
Location analysis of estrogen receptor ␣ target promoters reveals that FOXA1 defines a domain of the estrogen response Jose´ e Laganie` re*†, Genevie` ve Deblois*, Ce´ line Lefebvre*, Alain R. Bataille‡, Franc¸ois Robert‡, and Vincent Gigue` re*†§ *Molecular Oncology Group, Departments of Medicine and Oncology, McGill University Health Centre, Montreal, QC, Canada H3A 1A1; †Department of Biochemistry, McGill University, Montreal, QC, Canada H3G 1Y6; and ‡Laboratory of Chromatin and Genomic Expression, Institut de Recherches Cliniques de Montre´al, Montreal, QC, Canada H2W 1R7 Communicated by Ronald M. Evans, The Salk Institute for Biological Studies, La Jolla, CA, July 1, 2005 (received for review June 3, 2005) Nuclear receptors can activate diverse biological pathways within general absence of large scale functional data linking these putative a target cell in response to their cognate ligands, but how this binding sites with gene expression in specific cell types. compartmentalization is achieved at the level of gene regulation is Recently, chromatin immunoprecipitation (ChIP) has been used poorly understood. We used a genome-wide analysis of promoter in combination with promoter or genomic DNA microarrays to occupancy by the estrogen receptor ␣ (ER␣) in MCF-7 cells to identify loci recognized by transcription factors in a genome-wide investigate the molecular mechanisms underlying the action of manner in mammalian cells (20–24). This technology, termed 17-estradiol (E2) in controlling the growth of breast cancer cells. ChIP-on-chip or location analysis, can therefore be used to deter- We identified 153 promoters bound by ER␣ in the presence of E2. mine the global gene expression program that characterize the Motif-finding algorithms demonstrated that the estrogen re- action of a nuclear receptor in response to its natural ligand. -

Fine Mapping Studies of Quantitative Trait Loci for Baseline Platelet Count in Mice and Humans
Fine mapping studies of quantitative trait loci for baseline platelet count in mice and humans A thesis submitted in fulfillment of the requirements for the degree of Doctor of Philosophy Melody C Caramins December 2010 University of New South Wales ORIGINALITY STATEMENT ‘I hereby declare that this submission is my own work and to the best of my knowledge it contains no materials previously published or written by another person, or substantial proportions of material which have been accepted for the award of any other degree or diploma at UNSW or any other educational institution, except where due acknowledgement is made in the thesis. Any contribution made to the research by others, with whom I have worked at UNSW or elsewhere, is explicitly acknowledged in the thesis. I also declare that the intellectual content of this thesis is the product of my own work, except to the extent that assistance from others in the project's design and conception or in style, presentation and linguistic expression is acknowledged.’ Signed …………………………………………….............. Date …………………………………………….............. This thesis is dedicated to my father. Dad, thanks for the genes – and the environment! ACKNOWLEDGEMENTS “Nothing can come out of nothing, any more than a thing can go back to nothing.” - Marcus Aurelius Antoninus A PhD thesis is never the work of one person in isolation from the world at large. I would like to thank the following people, without whom this work would not have existed. Thank you firstly, to all my teachers, of which there have been many. Undoubtedly, the greatest debt is owed to my supervisor, Dr Michael Buckley. -

Core Circadian Clock Transcription Factor BMAL1 Regulates Mammary Epithelial Cell
bioRxiv preprint doi: https://doi.org/10.1101/2021.02.23.432439; this version posted February 23, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 Title: Core circadian clock transcription factor BMAL1 regulates mammary epithelial cell 2 growth, differentiation, and milk component synthesis. 3 Authors: Theresa Casey1ǂ, Aridany Suarez-Trujillo1, Shelby Cummings1, Katelyn Huff1, 4 Jennifer Crodian1, Ketaki Bhide2, Clare Aduwari1, Kelsey Teeple1, Avi Shamay3, Sameer J. 5 Mabjeesh4, Phillip San Miguel5, Jyothi Thimmapuram2, and Karen Plaut1 6 Affiliations: 1. Department of Animal Science, Purdue University, West Lafayette, IN, USA; 2. 7 Bioinformatics Core, Purdue University; 3. Animal Science Institute, Agriculture Research 8 Origination, The Volcani Center, Rishon Letsiyon, Israel. 4. Department of Animal Sciences, 9 The Robert H. Smith Faculty of Agriculture, Food, and Environment, The Hebrew University of 10 Jerusalem, Rehovot, Israel. 5. Genomics Core, Purdue University 11 Grant support: Binational Agricultural Research Development (BARD) Research Project US- 12 4715-14; Photoperiod effects on milk production in goats: Are they mediated by the molecular 13 clock in the mammary gland? 14 ǂAddress for correspondence. 15 Theresa M. Casey 16 BCHM Room 326 17 175 South University St. 18 West Lafayette, IN 47907 19 Email: [email protected] 20 Phone: 802-373-1319 21 22 bioRxiv preprint doi: https://doi.org/10.1101/2021.02.23.432439; this version posted February 23, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. -

Chromosome 20 Shows Linkage with DSM-IV Nicotine Dependence in Finnish Adult Smokers
Nicotine & Tobacco Research, Volume 14, Number 2 (February 2012) 153–160 Original Investigation Chromosome 20 Shows Linkage With DSM-IV Nicotine Dependence in Finnish Adult Smokers Kaisu Keskitalo-Vuokko, Ph.D.,1 Jenni Hällfors, M.Sc.,1,2 Ulla Broms, Ph.D.,1,3 Michele L. Pergadia, Ph.D.,4 Scott F. Saccone, Ph.D.,4 Anu Loukola, Ph.D.,1,3 Pamela A. F. Madden, Ph.D.,4 & Jaakko Kaprio, M.D., Ph.D.1,2,3 1 Hjelt Institute, Department of Public Health, University of Helsinki, Helsinki, Finland 2 Institute for Molecular Medicine Finland (FIMM), University of Helsinki, Helsinki, Finland 3 National Institute for Health and Welfare (THL), Helsinki, Finland 4 Department of Psychiatry, Washington University School of Medicine, St. Louis, MO Corresponding Author: Jaakko Kaprio, M.D., Ph.D., Department of Public Health, University of Helsinki, PO Box 41 (Manner- heimintie 172), Helsinki 00014, Finland. Telephone: +358-9-191-27595; Fax: +358-9-19127570; E-mail: [email protected] Received February 5, 2011; accepted June 21, 2011 2009). Despite several gene-mapping studies, the genes underlying Abstract liability to nicotine dependence (ND) remain largely unknown. Introduction: Chromosome 20 has previously been associated Recently, Han, Gelernter, Luo, and Yang (2010) performed a with nicotine dependence (ND) and smoking cessation. Our meta-analysis of 15 genome-wide linkage scans of smoking aim was to replicate and extend these findings. behavior. Linkage signals were observed on chromosomal regions 17q24.3–q25.3, 5q33.1–q35.2, 20q13.12–32, and 22q12.3–13.32. Methods: First, a total of 759 subjects belonging to 206 Finnish The relevance of the chromosome 20 finding is highlighted families were genotyped with 18 microsatellite markers residing by the fact that CHRNA4 encoding the nicotinic acetylcholine on chromosome 20, in order to replicate previous linkage findings. -

The DNA Sequence and Comparative Analysis of Human Chromosome 20
articles The DNA sequence and comparative analysis of human chromosome 20 P. Deloukas, L. H. Matthews, J. Ashurst, J. Burton, J. G. R. Gilbert, M. Jones, G. Stavrides, J. P. Almeida, A. K. Babbage, C. L. Bagguley, J. Bailey, K. F. Barlow, K. N. Bates, L. M. Beard, D. M. Beare, O. P. Beasley, C. P. Bird, S. E. Blakey, A. M. Bridgeman, A. J. Brown, D. Buck, W. Burrill, A. P. Butler, C. Carder, N. P. Carter, J. C. Chapman, M. Clamp, G. Clark, L. N. Clark, S. Y. Clark, C. M. Clee, S. Clegg, V. E. Cobley, R. E. Collier, R. Connor, N. R. Corby, A. Coulson, G. J. Coville, R. Deadman, P. Dhami, M. Dunn, A. G. Ellington, J. A. Frankland, A. Fraser, L. French, P. Garner, D. V. Grafham, C. Grif®ths, M. N. D. Grif®ths, R. Gwilliam, R. E. Hall, S. Hammond, J. L. Harley, P. D. Heath, S. Ho, J. L. Holden, P. J. Howden, E. Huckle, A. R. Hunt, S. E. Hunt, K. Jekosch, C. M. Johnson, D. Johnson, M. P. Kay, A. M. Kimberley, A. King, A. Knights, G. K. Laird, S. Lawlor, M. H. Lehvaslaiho, M. Leversha, C. Lloyd, D. M. Lloyd, J. D. Lovell, V. L. Marsh, S. L. Martin, L. J. McConnachie, K. McLay, A. A. McMurray, S. Milne, D. Mistry, M. J. F. Moore, J. C. Mullikin, T. Nickerson, K. Oliver, A. Parker, R. Patel, T. A. V. Pearce, A. I. Peck, B. J. C. T. Phillimore, S. R. Prathalingam, R. W. Plumb, H. Ramsay, C. M. -

Chromatin Conformation Links Distal Target Genes to CKD Loci
BASIC RESEARCH www.jasn.org Chromatin Conformation Links Distal Target Genes to CKD Loci Maarten M. Brandt,1 Claartje A. Meddens,2,3 Laura Louzao-Martinez,4 Noortje A.M. van den Dungen,5,6 Nico R. Lansu,2,3,6 Edward E.S. Nieuwenhuis,2 Dirk J. Duncker,1 Marianne C. Verhaar,4 Jaap A. Joles,4 Michal Mokry,2,3,6 and Caroline Cheng1,4 1Experimental Cardiology, Department of Cardiology, Thoraxcenter Erasmus University Medical Center, Rotterdam, The Netherlands; and 2Department of Pediatrics, Wilhelmina Children’s Hospital, 3Regenerative Medicine Center Utrecht, Department of Pediatrics, 4Department of Nephrology and Hypertension, Division of Internal Medicine and Dermatology, 5Department of Cardiology, Division Heart and Lungs, and 6Epigenomics Facility, Department of Cardiology, University Medical Center Utrecht, Utrecht, The Netherlands ABSTRACT Genome-wide association studies (GWASs) have identified many genetic risk factors for CKD. However, linking common variants to genes that are causal for CKD etiology remains challenging. By adapting self-transcribing active regulatory region sequencing, we evaluated the effect of genetic variation on DNA regulatory elements (DREs). Variants in linkage with the CKD-associated single-nucleotide polymorphism rs11959928 were shown to affect DRE function, illustrating that genes regulated by DREs colocalizing with CKD-associated variation can be dysregulated and therefore, considered as CKD candidate genes. To identify target genes of these DREs, we used circular chro- mosome conformation capture (4C) sequencing on glomerular endothelial cells and renal tubular epithelial cells. Our 4C analyses revealed interactions of CKD-associated susceptibility regions with the transcriptional start sites of 304 target genes. Overlap with multiple databases confirmed that many of these target genes are involved in kidney homeostasis. -

Identification of Novel Regulatory Genes in Acetaminophen
IDENTIFICATION OF NOVEL REGULATORY GENES IN ACETAMINOPHEN INDUCED HEPATOCYTE TOXICITY BY A GENOME-WIDE CRISPR/CAS9 SCREEN A THESIS IN Cell Biology and Biophysics and Bioinformatics Presented to the Faculty of the University of Missouri-Kansas City in partial fulfillment of the requirements for the degree DOCTOR OF PHILOSOPHY By KATHERINE ANNE SHORTT B.S, Indiana University, Bloomington, 2011 M.S, University of Missouri, Kansas City, 2014 Kansas City, Missouri 2018 © 2018 Katherine Shortt All Rights Reserved IDENTIFICATION OF NOVEL REGULATORY GENES IN ACETAMINOPHEN INDUCED HEPATOCYTE TOXICITY BY A GENOME-WIDE CRISPR/CAS9 SCREEN Katherine Anne Shortt, Candidate for the Doctor of Philosophy degree, University of Missouri-Kansas City, 2018 ABSTRACT Acetaminophen (APAP) is a commonly used analgesic responsible for over 56,000 overdose-related emergency room visits annually. A long asymptomatic period and limited treatment options result in a high rate of liver failure, generally resulting in either organ transplant or mortality. The underlying molecular mechanisms of injury are not well understood and effective therapy is limited. Identification of previously unknown genetic risk factors would provide new mechanistic insights and new therapeutic targets for APAP induced hepatocyte toxicity or liver injury. This study used a genome-wide CRISPR/Cas9 screen to evaluate genes that are protective against or cause susceptibility to APAP-induced liver injury. HuH7 human hepatocellular carcinoma cells containing CRISPR/Cas9 gene knockouts were treated with 15mM APAP for 30 minutes to 4 days. A gene expression profile was developed based on the 1) top screening hits, 2) overlap with gene expression data of APAP overdosed human patients, and 3) biological interpretation including assessment of known and suspected iii APAP-associated genes and their therapeutic potential, predicted affected biological pathways, and functionally validated candidate genes. -
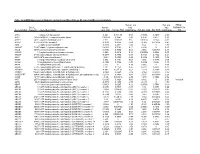
Mrna Expression in Human Leiomyoma and Eker Rats As Measured by Microarray Analysis
Table 3S: mRNA Expression in Human Leiomyoma and Eker Rats as Measured by Microarray Analysis Human_avg Rat_avg_ PENG_ Entrez. Human_ log2_ log2_ RAPAMYCIN Gene.Symbol Gene.ID Gene Description avg_tstat Human_FDR foldChange Rat_avg_tstat Rat_FDR foldChange _DN A1BG 1 alpha-1-B glycoprotein 4.982 9.52E-05 0.68 -0.8346 0.4639 -0.38 A1CF 29974 APOBEC1 complementation factor -0.08024 0.9541 -0.02 0.9141 0.421 0.10 A2BP1 54715 ataxin 2-binding protein 1 2.811 0.01093 0.65 0.07114 0.954 -0.01 A2LD1 87769 AIG2-like domain 1 -0.3033 0.8056 -0.09 -3.365 0.005704 -0.42 A2M 2 alpha-2-macroglobulin -0.8113 0.4691 -0.03 6.02 0 1.75 A4GALT 53947 alpha 1,4-galactosyltransferase 0.4383 0.7128 0.11 6.304 0 2.30 AACS 65985 acetoacetyl-CoA synthetase 0.3595 0.7664 0.03 3.534 0.00388 0.38 AADAC 13 arylacetamide deacetylase (esterase) 0.569 0.6216 0.16 0.005588 0.9968 0.00 AADAT 51166 aminoadipate aminotransferase -0.9577 0.3876 -0.11 0.8123 0.4752 0.24 AAK1 22848 AP2 associated kinase 1 -1.261 0.2505 -0.25 0.8232 0.4689 0.12 AAMP 14 angio-associated, migratory cell protein 0.873 0.4351 0.07 1.656 0.1476 0.06 AANAT 15 arylalkylamine N-acetyltransferase -0.3998 0.7394 -0.08 0.8486 0.456 0.18 AARS 16 alanyl-tRNA synthetase 5.517 0 0.34 8.616 0 0.69 AARS2 57505 alanyl-tRNA synthetase 2, mitochondrial (putative) 1.701 0.1158 0.35 0.5011 0.6622 0.07 AARSD1 80755 alanyl-tRNA synthetase domain containing 1 4.403 9.52E-05 0.52 1.279 0.2609 0.13 AASDH 132949 aminoadipate-semialdehyde dehydrogenase -0.8921 0.4247 -0.12 -2.564 0.02993 -0.32 AASDHPPT 60496 aminoadipate-semialdehyde -

Long-Term Oral Administration of LLHK, LHK, and HK Alters Gene Expression Profile and Restores Age-Dependent Atrophy and Dysfunction of Rat Salivary Glands
biomedicines Article Long-Term Oral Administration of LLHK, LHK, and HK Alters Gene Expression Profile and Restores Age-Dependent Atrophy and Dysfunction of Rat Salivary Glands 1, 1, 1, 2 Yasuko Ishikawa * , Tomasz D Pieczonka y, Aneta M Bragiel-Pieczonka z, Harumichi Seta , Tadahiro Ohkuri 2, Yumi Sasanuma 2 and Yuji Nonaka 2 1 Department of Medical Pharmacology, Institute of Biomedical Sciences, Tokushima University Graduate School, 3-18-15, Kuramoto-cho, Tokushima 770-8504, Japan; [email protected] (T.D.P.); [email protected] (A.M.B.-P.) 2 Suntory Global Innovation Center Ltd., Suntory World Research Center, 8-1-1 Seika-cho, Soraku-gun, Kyoto 619-0284, Japan; [email protected] (H.S.); [email protected] (T.O.); [email protected] (Y.S.); [email protected] (Y.N.) * Correspondence: [email protected] or [email protected]; Tel.: +80-3928-9628 Present address: Center of New Technologies, University of Warsaw, Banacha 2c, 02-097 Warsaw, Poland. y Present address: Department of Clinical Cytology, Centre of Medical Postgraduate Education, Marymoncka z 99/103, 01-813 Warsaw, Poland. Received: 26 January 2020; Accepted: 18 February 2020; Published: 20 February 2020 Abstract: Xerostomia, also known as dry mouth, is caused by a reduction in salivary secretion and by changes in the composition of saliva associated with the malfunction of salivary glands. Xerostomia decreases quality of life. In the present study, we investigated the effects of peptides derived from β-lactoglobulin C on age-dependent atrophy, gene expression profiles, and the dysfunction of salivary glands. -

Sac I Restriction Fragment Length Polymorphism (Rflp) Related to the Human Cst2 Gene
Bull. Tokyo dent. Coll., Vol. 43, No. 1, pp. 41ϳ44, February, 2002 41 Short Communication SAC I RESTRICTION FRAGMENT LENGTH POLYMORPHISM (RFLP) RELATED TO THE HUMAN CST2 GENE KIYOSHI MINAGUCHI, TATESHI KIRIYAMA, EIICHI SAITOH*, SATOKO ISEMURA** and KAZUO SANADA*** Department of Forensic Odontology, Tokyo Dental College, 1-2-2 Masago, Mihama-ku, Chiba 261-8502, Japan * Department of Oral Biochemistry, School of Dentistry at Niigata, The Nippon Dental University, 1-8 Hamaura-cho, Niigata 951-8580, Japan ** The Nippon Dental University Junior College at Niigata, 1-8 Hamaura-cho, Niigata 951-8580, Japan *** Department of Biochemistry, School of Dentistry at Tokyo, The Nippon Dental University, 1-9-20 Fujimi-cho, Chiyoda-ku, Tokyo 102-8159, Japan Received 7 January, 2002/Accepted for Publication 4 February, 2002 Abstract Restriction Fragment Length Polymorphism (RFLP) related to cystatin gene (CST) family was detected in the Japanese population by using restriction enzyme Sac I. A polymorphic site, located at 0.9kb from the 3Ј end of the CST2 gene, revealed a two allele polymorphism with band sizes of 3.5kb and 8.3kb by hybridization with probe including exon 2 of the CST1 gene. The gene frequencies in the Japanese population were 0.326 The phenotypes of the polymorphism .(86סfor 3.5kb allele and 0.674 for 8.3kb allele (n showed no association with the previously reported electrophoretic cystatin SA protein phenotypes22). Key words: Type 2 cystatin gene family—CST2—RFLP—SNP— Salivary cystatin SA—Polymorphism The human type 2 cystatin gene family is a nucleotide polymorphisms (SNPs) related to multigene family composed of seven members the type 2 cystatin gene family have been localized on chromosome 20p11.22,9,10,12,21,22,24) submitted in the JSNP database (the database and five of these genes (CST1-5) produce pro- of Japanese single nucleotide polymorphism) teins which are secreted in human saliva1,3,11,14–16). -

Hunting for Familial Parkinson's Disease Mutations in the Post
G C A T T A C G G C A T genes Article Hunting for Familial Parkinson’s Disease Mutations in the Post Genome Era Steven R. Bentley 1,* , Ilaria Guella 2, Holly E. Sherman 2, Hannah M. Neuendorf 1 , Alex M. Sykes 1, Javed Y. Fowdar 1, Peter A. Silburn 3, Stephen A. Wood 1, Matthew J. Farrer 2 and George D. Mellick 1 1 Griffith Institute for Drug Discovery, Griffith University, Don Young Road, Nathan, QLD 4111, Australia; [email protected] (H.M.N.); a.sykes@griffith.edu.au (A.M.S.); j.fowdar@griffith.edu.au (J.Y.F.); s.wood@griffith.edu.au (S.A.W.); g.mellick@griffith.edu.au (G.D.M.) 2 Djavad Mowafhagian Centre for Brain, University of British Columbia, Vancouver, BC V6T 1Z3, Canada; [email protected] (I.G.); [email protected] (H.E.S.); [email protected] (M.J.F.) 3 Centre for Clinical Research, University of Queensland, Brisbane, QLD 4029, Australia; [email protected] * Correspondence: s.bentley@griffith.edu.au Abstract: Parkinson’s disease (PD) is typically sporadic; however, multi-incident families provide a powerful platform to discover novel genetic forms of disease. Their identification supports de- ciphering molecular processes leading to disease and may inform of new therapeutic targets. The LRRK2 p.G2019S mutation causes PD in 42.5–68% of carriers by the age of 80 years. We hypothesise similarly intermediately penetrant mutations may present in multi-incident families with a generally strong family history of disease. We have analysed six multiplex families for missense variants using whole exome sequencing to find 32 rare heterozygous mutations shared amongst affected members.