The Effects of Lidocaine with Epinephrine on Bupivacaine-Induced
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

WO 2016/081643 Al 26 May 2016 (26.05.2016) W P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2016/081643 Al 26 May 2016 (26.05.2016) W P O P C T (51) International Patent Classification: (71) Applicant (for AL, AT, BE, BG, CH, CN, CY, CZ, DE, DK, C07K 16/28 (2006.01) C12N 15/13 (2006.01) EE, ES, FI, FR, GB, GR, HR, HU, IE, IN, IS, IT, LT, LU, C07K 16/40 (2006.01) A61K 39/395 (2006.01) LV, MC, MK, MT, NL, NO, PL, PT, RO, RS, SE, SI, SK, C07K 16/46 (2006.01) A61P 25/00 (2006.01) SM, TR only): F. HOFFMANN-LA ROCHE AG [CH/CH]; Grenzacherstrasse 124, 4070 Basel (CH). (21) International Application Number: PCT/US20 15/06 1405 (72) Inventors: ZHANG, Yin; 1 DNA Way, South San Fran cisco, California 94080 (US). ZUCHERO, Joy Yu; 1 (22) International Filing Date: DNA Way, South San Francisco, California 94080 (US). 18 November 2015 (18.1 1.2015) ATWAL, Jasvinder; 1 DNA Way, South San Francisco, (25) Filing Language: English California 94080 (US). COUCH, Jessica; 1 DNA Way, South San Francisco, California 94080 (US). DENNIS, (26) Publication Language: English Mark S.; 1 DNA Way, South San Francisco, California (30) Priority Data: 94080 (US). ERNST, James A.; 1 DNA Way, South San 62/081,827 19 November 2014 (19. 11.2014) US Francisco, California 94080 (US). WATTS, Ryan J.; 1 DNA Way, South San Francisco, California 94080 (US). (71) Applicant (for all designated States except AL, AT, BE, LAZAR, Gregory A.; 1 DNA Way, South San Francisco, BG, BN, CH, CN, CY, CZ, DE, DK, EE, ES, FI, FR, GB, California 94080 (US). -

761066Orig1s000
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 761066Orig1s000 CLINICAL REVIEW(S) Medical Officer’s Review of BLA 761066 Division of Dermatology and Dental Products Type: Biosimilar 351(k) Correspondence date: 25-MAY-2017 Serial Amendment: 000 CDER Stamp date: 25-MAY-2017 Supporting Document Number: 001 Review Date: 19-MAR-2018 Applicant: Samsung Bioepis Co., Ltd. 107 Cheomdan-daero, Yeonsu-gu Incheon, Republic of Korea 21987 Drug: SB4, a proposed biosimilar to U.S.-licensed Enbrel (etanercept) Route of Administration: Subcutaneous Dosage Form: Single-use pre-filled syringes: 50 mg/1.0 mL (50 mg/mL); 25 mg/0.5 mL (50 mg/mL) Pharmacologic Category: 1-235-tumor necrosis factor receptor (human) fusion protein with 236-467-immunoglobulin Gi (human γ1-chain Fc fragment), dimer Proposed Indications: 1) Rheumatoid Arthritis (RA) in combination with methotrexate: Reducing signs and symptoms, inhibiting the progression of structural damage, and improving physical function in patients with moderately to severely active disease. 2) Polyarticular Juvenile Idiopathic Arthritis Reducing signs and symptoms of moderately to severe active polyarticular juvenile idiopathic arthritis (JIA) in patients 2 years and older and weigh 63 kg (138 pounds) or more. 3) Ankylosing Spondylitis (AS): Reducing signs and symptoms in patients with active disease 4) Psoriatic Arthritis (PsA): Reducing signs and symptoms of active arthritis, inhibiting the progression of structural damage, and improving physical function. 5) Plaque Psoriasis (PsO): Treatment of patients 4 years and older with chronic moderate or severe plaque psoriasis who are candidates for systemic therapy or phototherapy. Project Manager: Barbara Gould Team Leader: David Kettl, MD Medical Officer: Gary Chiang, MD, MPH. -

N-Ethoxyethylpiperidine, Trimecaine and Piromecaine Based Ionic Compounds: Synthesis and Prediction of Biological Activity
International Journal of Chemical Engineering and Applications, Vol. 8, No. 3, June 2017 N-ethoxyethylpiperidine, Trimecaine and Piromecaine Based Ionic Compounds: Synthesis and Prediction of Biological Activity D. S. Zolotareva, A. A. Basharimova, S. Bayazit, V. K. Yu, and A. G. Zazybin anti-arrhythmic activity with no teratogenic, embriotoxic, Abstract—Herein we report on the synthesis of new ionic mutagenic and allergic effects [7]. Trimecaine which we used compounds based on N-ethoxyethylpiperidine, trimecaine and in our research as a parent compound nowadays has been piromecaine, obtained via N-alkylation with alkyl halides. The applied as a local anesthetic and antiarrhythmic and in cardio combination of piperidine ring, secondary amine group, trimecaine and piromecaine in form of a base with different surgery [8], [9]. Piromecaine also shows local anesthetic and alkyl radicals bring to molecule new biological activity and/or antiarrhythmic activity [10], [11]. To define the range of reduce toxic effects of parent compounds. The potential biological activity of new synthesized compounds we used pharmacological activity and potential toxic effects of the Pass online service (Prediction of Activity Spectra for obtained compounds were predicted with PASS online service Substances) [12], which helps to predict biological and (Prediction of Activity Spectra for Substances) showing pharmacological effects, action mechanisms and also toxicity cardiotonic, antiarrhythmic and spasmolytic potential activities. The dependence of the potential activity on the nature of and adverse impact of compound, for example as in [13] – substituent was compared. These novel ionic compounds can be [15]. In this manuscript we report some methods for the applied for synthesis of ionic compounds and drug candidates. -

Local Anaesthetic Infiltration for Peri-Operative Pain Control in Total
Marques et al. BMC Musculoskeletal Disorders 2014, 15:220 http://www.biomedcentral.com/1471-2474/15/220 RESEARCH ARTICLE Open Access Local anaesthetic infiltration for peri-operative pain control in total hip and knee replacement: systematic review and meta-analyses of short- and long-term effectiveness Elsa MR Marques1*, Hayley E Jones1, Karen T Elvers2, Mark Pyke3, Ashley W Blom2 and Andrew D Beswick2 Abstract Background: Surgical pain is managed with multi-modal anaesthesia in total hip replacement (THR) and total knee replacement (TKR). It is unclear whether including local anaesthetic infiltration before wound closure provides additional pain control. Methods: We performed a systematic review of randomised controlled trials of local anaesthetic infiltration in patients receiving THR or TKR. We searched MEDLINE, Embase and Cochrane CENTRAL to December 2012. Two reviewers screened abstracts, extracted data, and contacted authors for unpublished outcomes and data. Outcomes collected were post-operative pain at rest and during activity after 24 and 48 hours, opioid requirement, mobilisation, hospital stay and complications. When feasible, we estimated pooled treatment effects using random effects meta-analyses. Results: In 13 studies including 909 patients undergoing THR, patients receiving local anaesthetic infiltration experienced a greater reduction in pain at 24 hours at rest by standardised mean difference (SMD) −0.61 (95% CI −1.05, −0.16; p = 0.008) and by SMD −0.43 (95% CI −0.78 −0.09; p = 0.014) at 48 hours during activity. In TKR, diverse multi-modal regimens were reported. In 23 studies including 1439 patients undergoing TKR, local anaesthetic infiltration reduced pain on average by SMD −0.40 (95% CI −0.58, −0.22; p < 0.001) at 24 hours at rest and by SMD −0.27 (95% CI −0.50, −0.05; p = 0.018) at 48 hours during activity, compared with patients receiving no infiltration or placebo. -

This Is the Author's Version of a Work That Was Accepted for Publication In
NOTICE: this is the author’s version of a work that was accepted for publication in the European Journal of Medicinal Chemistry. Changes resulting from the publishing process, such as peer review, editing, corrections, structural formatting, and other quality control mechanisms may not be reflected in this document. Changes may have been made to this work since it was submitted for publication. A definitive version was subsequently published in the European Journal of Medicinal Chemistry, Vol. 63 (2013). DOI: 10.1016/j.ejmech.2013.02.003 Synthesis, local anaesthetic and antiarrhythmic activities of N-alkyl derivatives of proline anilides Dmitrii V. Kalinin,a Vladimir I. Pantsurkin,a Boris Ya. Syropyatov,a Svetlana A. Kalinina,a Irina P. Rudakova,a Mikhail I. Vakhrina and Anton V. Dolzhenkob,c* a Perm State Pharmaceutical Academy, 2 Polevaya Street, Perm 614990, Russian Federation b Jeffrey Cheah School of Medicine and Health Sciences, Monash University Sunway Campus, Jalan Lagoon Selatan, Bandar Sunway, Selangor 46150, Malaysia c School of Pharmacy, Curtin Health Innovation Research Institute, Curtin University, GPO Box U1987 Perth, Western Australia 6845, Australia Corresponding author. Tel.: +61-8-9266-3747; fax: +61-8-9266-2769. E-mail addresses: [email protected]; [email protected]; [email protected] (A. V. Dolzhenko) Abstract We describe here the design, synthesis and evaluation of in vivo local anaesthetic and antiarrhythmic activities of a series of N-alkylproline anilides. Most of the compounds demonstrated surface anaesthetic activity higher than that of lidocaine, ropivacaine and bupivacaine. We established that the local anaesthetic activity was sensitive to structural variations in the substitution pattern at the aromatic ring and the type of alkyl group at the proline nitrogen atom. -

Pharmacology
STATE ESTABLISHMENT «DNIPROPETROVSK MEDICAL ACADEMY OF HEALTH MINISTRY OF UKRAINE» V.I. MAMCHUR, V.I. OPRYSHKO, А.А. NEFEDOV, A.E. LIEVYKH, E.V.KHOMIAK PHARMACOLOGY WORKBOOK FOR PRACTICAL CLASSES FOR FOREIGN STUDENTS STOMATOLOGY DEPARTMENT DNEPROPETROVSK - 2016 2 UDC: 378.180.6:61:615(075.5) Pharmacology. Workbook for practical classes for foreign stomatology students / V.Y. Mamchur, V.I. Opryshko, A.A. Nefedov. - Dnepropetrovsk, 2016. – 186 p. Reviewed by: N.I. Voloshchuk - MD, Professor of Pharmacology "Vinnitsa N.I. Pirogov National Medical University.‖ L.V. Savchenkova – Doctor of Medicine, Professor, Head of the Department of Clinical Pharmacology, State Establishment ―Lugansk state medical university‖ E.A. Podpletnyaya – Doctor of Pharmacy, Professor, Head of the Department of General and Clinical Pharmacy, State Establishment ―Dnipropetrovsk medical academy of Health Ministry of Ukraine‖ Approved and recommended for publication by the CMC of State Establishment ―Dnipropetrovsk medical academy of Health Ministry of Ukraine‖ (protocol №3 from 25.12.2012). The educational tutorial contains materials for practical classes and final module control on Pharmacology. The tutorial was prepared to improve self-learning of Pharmacology and optimization of practical classes. It contains questions for self-study for practical classes and final module control, prescription tasks, pharmacological terms that students must know in a particular topic, medical forms of main drugs, multiple choice questions (tests) for self- control, basic and additional references. This tutorial is also a student workbook that provides the entire scope of student’s work during Pharmacology course according to the credit-modular system. The tutorial was drawn up in accordance with the working program on Pharmacology approved by CMC of SE ―Dnipropetrovsk medical academy of Health Ministry of Ukraine‖ on the basis of the standard program on Pharmacology for stomatology students of III - IV levels of accreditation in the specialties Stomatology – 7.110105, Kiev 2011. -

United States Patent (19) 11 Patent Number: 5,993,836 Castillo (45) Date of Patent: Nov.30, 1999
USOO5993836A United States Patent (19) 11 Patent Number: 5,993,836 Castillo (45) Date of Patent: Nov.30, 1999 54 TOPICAL ANESTHETIC FORMULATION 5,002,974 3/1991 Geria ....................................... 514/782 5,332,576 7/1994. Mantelle . ... 424/443 76 Inventor: James G. Castillo, 15412-15'St., Lutz, 5,585,398 12/1996 Ernst ....................................... 514/537 Fla. 33549 Primary Examiner Thurman K. Page ASSistant Examiner William E. Benston, Jr. 21 Appl. No.: 09/067,948 Attorney, Agent, or Firm-Pendorf & Cutliff 22 Filed: Apr. 28, 1998 57 ABSTRACT 51 Int. Cl. ................................ A61K 6/00; A61K 7/00 A topical, transdermal anesthetic comprising a eutectic mix 52 U.S. Cl. .......................... 424/401; 424/443; 424/435; ture of, preferably, lidocaine and prilocaine in a ratio of 424/485 about 3:1 by weight, incorporated within a lipophilic base. 58 Field of Search ..................................... 424/401, 443, In addition to the property of rapid-onset, the formulation of 424/435, 485, 484, 450, 486 the present invention has high Storage Stability and is leSS restricted by dosage limitations of other prilocaine 56) References Cited containing transdermal anesthetics and is, advantageously, U.S. PATENT DOCUMENTS not dependent upon occlusive dressing for optimal transder mal absorption. 4,529,601 7/1985 Broberg et al. ......................... 514/626 4.562,060 12/1985 Broberg et al. ........................... 424/28 20 Claims, No Drawings 5,993,836 1 2 TOPCAL ANESTHETIC FORMULATION of risks associated with parenteral treatment, elimination of the inconveniences of parenteral treatment; and elimination BACKGROUND OF THE INVENTION of gastrointestinal irritation resulting from exposing the 1. -

Percutaneous Absorption of Disopyramide, Lidocaine and Trimecaine
Volume 141, 1998 31 PERCUTANEOUS ABSORPTION OF DISOPYRAMIDE, LIDOCAINE AND TRIMECAINE Jan Příborskýa,b,*, Katsuya Kikuchic, Kozo Takayamac, Tsuneji Nagaic a Glaxo W ellcome s.r.o., K Žižkovu 4, 190 00 Prague, Czech Republic b Institute of Pharmacology, Faculty of Medicine, Palacký University, Hněvotínská 3, 77 515 Olomouc, Czech Republic c Department of Pharmaceutics, Hoshi University, Ebara 2-4-41, Shinagawa-ku, Tokyo 142, Japan Received December 10,1997 Key words: Antiarrhytmic drugs / Penetration enhancer / Cyclic monoterpenes Promoting effect of cyclic monoterpenes on percutaneous absorption of antiarrhytmic drugs disopyramide, lidocaine and trimecaine was investigated in the rats. Laurocapram (Azone) was used as a standard comparator of penetration enhance- ment. The absorption of trimecaine was significantly enhanced by addition of limonene, trans-p-menthane and Azone in 1% concentration. Lidocaine and disopyramide penetrated across the skin only when 1% of limonene was used. Other cyclic monoterpenes showed no effect on percutaneous absorption of examined drugs. INTRODUCTION ride which was generous gift from Sanitas (Říčany u Pra- hy, Czech Republic). Following cyclic monoterpenes we- Previous studies performed in our laboratory proved re used: hydrocarbons – limonene and trans-p-menthane, that several antirheumatics are suitable candidates for alcohols – menthol, ketones – menthone and ethers – percutaneous drug formulations1,2. Percutaneous absorp- cineole, all by Tokyo Chemical Industries Co., Ltd. (To- tion of drugs can be enhanced by means of cyclohexanone kyo, Japan). Laurocapram (Azone) was supplied by Su- derivatives1, cyclic monoterpenes 2 and other penetration misho-Nelson Co., Ltd. (Osaka Japan). Carboxyvinyl enhancers like Azone, DMSO, pyrrolidones3 or dioxola- polymer, marketed as „hiviswako 105“ was supplied from nes4. -
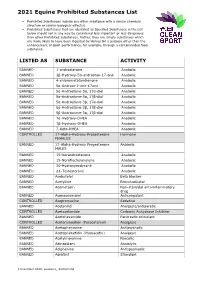
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

Synthesis and Biological Activities of Local Anesthetics Cite This: RSC Adv.,2019,9, 41173 Shiyang Zhou,A Gangliang Huang *B and Guangying Chen*A
RSC Advances View Article Online REVIEW View Journal | View Issue Synthesis and biological activities of local anesthetics Cite this: RSC Adv.,2019,9, 41173 Shiyang Zhou,a Gangliang Huang *b and Guangying Chen*a Local anesthetics are mainly used in stomatology, ophthalmology, gynecology and surgery to temporarily relieve pain. Local anesthetics act on nerve endings or around nerve trunks, and are combined with specific sodium ion (Na+) channel sites on the nerve membrane. They can affect the membrane potential by reducing Na+ passage through sodium ion channels, thus blocking the generation and conduction of Received 8th November 2019 nerve impulses, reversibly blocking the generation and conduction of sensory nerve impulses. Local Accepted 27th November 2019 anesthetics are used for convenience in local surgical operations and treatments. Herein, we mainly DOI: 10.1039/c9ra09287k review the research progress on local anesthetics and discuss the important aspects of design, synthesis rsc.li/rsc-advances and biological activity of various new compounds. – Creative Commons Attribution-NonCommercial 3.0 Unported Licence. 1. Introduction which is mainly used before surgery.8 11 In contrast, local anesthetics act on nerve endings or around nerve trunks, Anesthetics are a type of drug that act on the nervous system, reversibly blocking the generation and conduction of sensory causing it to be inhibited and resulting in the loss of the sense nerve impulses, and temporarily eliminating local sensation of pain.1–4 According to the different scopes of action of drugs, (mainly pain sensation) under the condition of consciousness. the department of anesthetics is divided into general anes- Moreover, it is convenient for local surgical operations and – thetics and local anesthetics.5–7 General anesthetics act on the treatments.12 14 Local anesthetics are mainly used in stomatol- central nervous system, causing it to be reversibly inhibited, ogy, ophthalmology, gynecology and surgery to relieve pain thereby resulting in a loss of consciousness and sensation. -

Rational Design, Synthesis, and In-Silico Evaluation of Homologous Local Anesthetic Compounds As TASK-1 Channel Blockers †
Proceeding Paper Rational Design, Synthesis, and In-Silico Evaluation of Homologous Local Anesthetic Compounds as TASK-1 Channel Blockers † Lorena Camargo-Ayala 1, Luis Prent-Peñaloza 2, Mauricio Bedoya 3, Margarita Gutiérrez 2,* and Wendy González 3,4,* 1 Doctorate in Sciences Mention in Research and Development of Bioactive Products, Institute of Chemistry of Natural Resources, Organic Synthesis Laboratory and Biological Activity (LSO-Act-Bio), University of Talca, Casilla 747, Talca 3460000, Chile; [email protected] 2 Organic Synthesis Laboratory and Biological Activity (LSO-Act-Bio), Institute of Chemistry of Natural Resources, University of Talca, Casilla 747, Talca 3460000, Chile; [email protected] 3 Center for Bioinformatics and Molecular Simulations (CBSM), Universidad de Talca, Casilla 747, Talca 3460000, Chile; [email protected] 4 Millennium Nucleus of Ion Channels-Associated Diseases (MiNICAD). Universidad de Talca, Casilla 747, Talca 3460000, Chile * Correspondence: [email protected] (M.G.); [email protected] (W.G.) † Presented at the 24th International Electronic Conference on Synthetic Organic Chemistry, 15 November–15 December 2020; Available online: https://ecsoc-24.sciforum.net/. Abstract: Advances in different technological and scientific fields have led to the development of tools that allow the design of drugs in a rational way, using defined therapeutic targets, and through Citation: Camargo-Ayala, L.; simulations that offer a molecular view of the ligand–receptor interactions, giving precise infor- -

(12) United States Patent (10) Patent No.: US 9,056,044 B2 MO Et Al
USOO9056.04.4B2 (12) United States Patent (10) Patent No.: US 9,056,044 B2 MO et al. (45) Date of Patent: *Jun. 16, 2015 (54) ALPROSTADIL AND LIDOCAINE Y10S 514/947 (2013.01); A61K 47/10 TREATMENT OF PREMATURE (2013.01); A61 K47/14 (2013.01); A61K47/26 EUACULATION (2013.01); A61 K3I/20 (2013.01); A61 K 3 1/4453 (2013.01) (71) Applicant: NexMed Holdings, Inc., San Diego, CA (58) Field of Classification Search (US) CPC ............ A61 K31/5575; A61 K31/445; A61 K (72) Inventors: Y. Joseph Mo, Princeton, NJ (US); 9/0014; A61 K9/0034; A61 K31/167 Minggi Lu, Lawrenceville, NJ (US); USPC .......... 514/573, 330, 331, 947, 626; 424/400, Qin Wang, Plainsboro, NJ (US); James 424/434:562/503 L. Yeager, Lake Forest, IL (US) See application file for complete search history. (73) Assignee: NEXMED HOLDINGS, INC., San (56) References Cited Diego, CA (US) U.S. PATENT DOCUMENTS (*) Notice: Subject to any disclaimer, the term of this 4.954,487 A 9/1990 Cooper et al. patent is extended or adjusted under 35 5,151,448 A 9, 1992 Crenshaw et al. U.S.C. 154(b) by 0 days. 5,234,957 A 8, 1993 Mantelle 5,314.915 A 5/1994 Rencher This patent is Subject to a terminal dis 5,332,576 A 7, 1994 Mantelle claimer. 5,654,337 A 8, 1997 Roentsch et al. 5,708,031 A 1, 1998 Scott 5,922,341 A 7/1999 Smith et al. (21) Appl. No.: 14/463,966 5,942.545 A 8, 1999 Samour et al.