Maternal Butalbital Use and Selected Defects in the National Birth Defects Prevention Study
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chronic Use of Opioid Medications Before and After Bariatric Surgery
Supplementary Online Content Raebel MA, Newcomer SR, Reifler LM, et al. Chronic use of opioid medications before and after bariatric surgery. JAMA. doi:10.1001/jama.2013.278344 eFigure. Opioid Dispensings in 60 Days Before and 60 Days After Bariatric Surgery eTable 1. Opioid Classification and Morphine Equivalents Conversion Factors for Opioids Administered by Tablet, Capsule, Liquid, Transdermal Patch, and Transmucosal Formulations eTable 2. ICD-9 Codes for Comorbid Diagnosis of Substance Abuse, Anxiety, Posttraumatic Stress Disorder, and/or Depression eTable 3. ICD-9 Codes for Inclusion and Exclusion of Chronic Pain Diagnoses eTable 4. Therapeutic Classes, Generic Names, and Selected Brand Names of Covariate Medications eTable 5. Types of Opioids Dispensed During the Year Before and the Year After Bariatric Surgery Among Presurgery Chronic Opioid Users eTable 6. Types of Opioids Used Before and After Bariatric Surgery Among 933 Individuals With Chronic Opioid Use Before Bariatric Surgery eTable 7. Unadjusted Characteristics of Chronic Opioid Users According to Whether or Not Both Presurgery and Postsurgery Body Mass Indexes (BMIs) Data Were Available eTable 8. Presurgery and Postsurgery Use of Opioids and Selected Other Analgesic and Adjunctive Pain Medication Classes Among Presurgery Chronic Opioid Users With Presurgery Chronic Pain Diagnoses This supplementary material has been provided by the authors to give readers additional information about their work. © 2013 American Medical Association. All rights reserved. Downloaded From: -

BUTALBITAL ANALGESICS Allzital (Butalbital-Acetaminophen
BUTALBITAL ANALGESICS Allzital (butalbital-acetaminophen), butalbital-aspirin-caffeine, butalbital-aspirin-caffeine- codeine, butalbital-acetaminophen, butalbital-acetaminophen-caffeine, butalbital- acetaminophen-caffeine-codeine, Vanatol LQ (butalbital-acetaminophen-caffeine liquid oral solution) RATIONALE FOR INCLUSION IN PA PROGRAM Background Butalbital containing products are non-opioid analgesics that contain a combination of different drug products indicated for the relief of the symptom complex of tension (or muscle contraction) headache pain. Butalbital is a short to intermediate-acting barbiturate that works in concert with acetaminophen, an antipyretic non-salicylate agent, aspirin, a pain-relieving NSAID, and caffeine, a stimulant that works in the CNS, to decrease pain via a mechanism that isn’t well understood. Butalbital is a habit-forming drug that potentiates the effects of other commonly abuse drugs or substances like alcohol. Caffeine might help increase vasodilation and smooth muscle relaxation, while butalbital is thought to help balance the CNS stimulation caused by caffeine and produces depressant effects (1). Regulatory Status FDA approved indication: Butalbital containing products are used in the relief of the symptom complex of tension or muscle contraction headaches (2-8). Frequent use of acute medications is generally thought to cause medication-overuse headache. To decrease the risk of medication-overuse headache (“rebound headache” or “drug-induced headache”) many experts limit acute therapy to two headache days per week on a regular basis. Based on concerns of overuse, medication-overuse headache, and withdrawal, the use of butalbital-containing analgesics should be limited and carefully monitored. The Allzital limit is set to the maximum of 12 doses per day for acute treatment of 8 headaches per month as this product contains less butalbital than other products. -

Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules
Medication Guide Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules, C III Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules are: • A strong prescription pain medicine that contains an opioid (narcotic) that is indicated for the relief of the symptom complex of tension (or muscle contraction) headache, when other pain treatments such as non-opioid pain medicines do not treat your pain well enough or you cannot tolerate them. • An opioid pain medicine that can put you at risk for overdose and death. Even if you take your dose correctly as prescribed you are at risk for opioid addiction, abuse, and misuse that can lead to death. Important information about Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules: • Get emergency help right away if you take too much Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules (overdose). When you first start taking Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules, when your dose is changed, or if you take too much (overdose), serious or life-threatening breathing problems that can lead to death may occur. • Taking Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules with other opioid medicines, benzodiazepines, alcohol, or other central nervous system depressants (including street drugs) can cause severe drowsiness, decreased awareness, breathing problems, coma, and death. • Never give anyone else your Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules. They could die from taking it. Store Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules away from children and in a safe place to prevent stealing or abuse. Selling or giving away Butalbital, Acetaminophen, Caffeine, and Codeine Phosphate Capsules is against the law. • Get emergency help right away if you take more than 4,000 mg of acetaminophen in 1 day. -
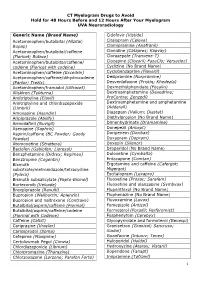
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology Generic Name (Brand Name) Cidofovir (Vistide) Acetaminophen/butalbital (Allzital; Citalopram (Celexa) Bupap) Clomipramine (Anafranil) Acetaminophen/butalbital/caffeine Clonidine (Catapres; Kapvay) (Fioricet; Butace) Clorazepate (Tranxene-T) Acetaminophen/butalbital/caffeine/ Clozapine (Clozaril; FazaClo; Versacloz) codeine (Fioricet with codeine) Cyclizine (No Brand Name) Acetaminophen/caffeine (Excedrin) Cyclobenzaprine (Flexeril) Acetaminophen/caffeine/dihydrocodeine Desipramine (Norpramine) (Panlor; Trezix) Desvenlafaxine (Pristiq; Khedezla) Acetaminophen/tramadol (Ultracet) Dexmethylphenidate (Focalin) Aliskiren (Tekturna) Dextroamphetamine (Dexedrine; Amitriptyline (Elavil) ProCentra; Zenzedi) Amitriptyline and chlordiazepoxide Dextroamphetamine and amphetamine (Limbril) (Adderall) Amoxapine (Asendin) Diazepam (Valium; Diastat) Aripiprazole (Abilify) Diethylpropion (No Brand Name) Armodafinil (Nuvigil) Dimenhydrinate (Dramamine) Asenapine (Saphris) Donepezil (Aricept) Aspirin/caffeine (BC Powder; Goody Doripenem (Doribax) Powder) Doxapram (Dopram) Atomoxetine (Strattera) Doxepin (Silenor) Baclofen (Gablofen; Lioresal) Droperidol (No Brand Name) Benzphetamine (Didrex; Regimex) Duloxetine (Cymbalta) Benztropine (Cogentin) Entacapone (Comtan) Bismuth Ergotamine and caffeine (Cafergot; subcitrate/metronidazole/tetracycline Migergot) (Pylera) Escitalopram (Lexapro) Bismuth subsalicylate (Pepto-Bismol) Fluoxetine (Prozac; Sarafem) -

Drug Plasma Half-Life and Urine Detection Window | January 2019
500 Chipeta Way | Salt Lake City, UT 84108-1221 Phone: (800) 522-2787 | Fax: (801) 583-2712 www.aruplab.com | www.arupconsult.com DRUG PLASMA HALF-LIFE AND URINE DETECTION WINDOW | JANUARY 2019 URINE- PLASMA DRUG, DRUG METABOLITE(S)* COMMON TRADE AND STREET NAMES, NOTES DETECTION HALF-LIFEt WINDOWt STIMULANTS Benzedrine, dexedrine, Adderall, Vyvanse, speed; could be methamphetamine Amphetamine 7–34 hours 1–5 days metabolite; if so, typically < 30 percent of parent Cocaine Coke, crack; parent drug rarely observed due to short half-life 0.7–1.5 hours < 1 day Benzoylecgonine Cocaine metabolite 5.5–7.5 hours 1–2 days Desoxyn, methedrine, Vicks inhaler (D- and L-isomers not resolved; low concentrations Methamphetamine expected if the source is Vicks); selegeline (Atapryl, Carbex, Eldepryl, Zelapar) 6–17 hours 1–5 days metabolite Methylenedioxyamphetamine (MDA) MDA 11–17 hours 1–3 days Methylenedioxyethylamphetamine (MDEA) MDEA, MDE, Eve 6–11 hours 1–3 days Methylenedioxymethamphetamine (MDMA) MDMA, XTC, ecstasy, Molly 6–10 hours 1–3 days Methylphenidate Ritalin, Concerta, Focalin, Metadate, Methylin 1.4–4.2 hours < 1 day Ritalinic acid Methylphenidate metabolite 1.8–2.5 hours < 1 day Phentermine Adipex-P, Lomaira, Qsymia 19–24 hours 1–5 days OPIOIDS Buprenorphine Belbuca, Buprenex, Butrans, Suboxone, Subutex, Sublocade, Zubsolv 26–42 hours 1–7 days Norbuprenorphine, Glucuronides Buprenorphine metabolites 15–150 hours 1–14 days Included in many preparations; morphine metabolite; may be a contaminant if < 2 Codeine 1.9–3.9 hours 1–3 days percent of morphine Fentanyl Actiq, Duragesic, Fentora, Lazanda, Sublimaze, Subsys, Ionsys 3–12 hours 1–3 days Norfentanyl Fentanyl metabolite 9–10 hours 1–3 days Heroin Diacetylmorphine, dope, smack, dust; parent drug not detected. -

CENTRAL NERVOUS SYSTEM DEPRESSANTS Opioid Pain Relievers Anxiolytics (Also Belong to Psychiatric Medication Category) • Codeine (In 222® Tablets, Tylenol® No
CENTRAL NERVOUS SYSTEM DEPRESSANTS Opioid Pain Relievers Anxiolytics (also belong to psychiatric medication category) • codeine (in 222® Tablets, Tylenol® No. 1/2/3/4, Fiorinal® C, Benzodiazepines Codeine Contin, etc.) • heroin • alprazolam (Xanax®) • hydrocodone (Hycodan®, etc.) • chlordiazepoxide (Librium®) • hydromorphone (Dilaudid®) • clonazepam (Rivotril®) • methadone • diazepam (Valium®) • morphine (MS Contin®, M-Eslon®, Kadian®, Statex®, etc.) • flurazepam (Dalmane®) • oxycodone (in Oxycocet®, Percocet®, Percodan®, OxyContin®, etc.) • lorazepam (Ativan®) • pentazocine (Talwin®) • nitrazepam (Mogadon®) • oxazepam ( Serax®) Alcohol • temazepam (Restoril®) Inhalants Barbiturates • gases (e.g. nitrous oxide, “laughing gas”, chloroform, halothane, • butalbital (in Fiorinal®) ether) • secobarbital (Seconal®) • volatile solvents (benzene, toluene, xylene, acetone, naptha and hexane) Buspirone (Buspar®) • nitrites (amyl nitrite, butyl nitrite and cyclohexyl nitrite – also known as “poppers”) Non-Benzodiazepine Hypnotics (also belong to psychiatric medication category) • chloral hydrate • zopiclone (Imovane®) Other • GHB (gamma-hydroxybutyrate) • Rohypnol (flunitrazepam) CENTRAL NERVOUS SYSTEM STIMULANTS Amphetamines Caffeine • dextroamphetamine (Dexadrine®) Methelynedioxyamphetamine (MDA) • methamphetamine (“Crystal meth”) (also has hallucinogenic actions) • methylphenidate (Biphentin®, Concerta®, Ritalin®) • mixed amphetamine salts (Adderall XR®) 3,4-Methelynedioxymethamphetamine (MDMA, Ecstasy) (also has hallucinogenic actions) Cocaine/Crack -
Headaches in Woman
11 Headaches in Women Randolph W. Evans Women have headaches more commonly than men. The preva- lence of migraine is 18% of women and 6% of men. This gender ratio increases from menarche, peaks at 42 years of age, and then declines. For young women the incidence of migraine with aura peaks between the ages of 12 and 13 (14.1/1,000 person- years), and migraine without aura peaks between the ages of 14 and 17 (18.9/1,000 person-years) (1). Table 11-1 provides the life- time prevalence of various headaches in women and men. Estrogen levels are a key factor in the increased prevalence of migraine in women. Evidence includes the following: migraine prevalence increases at menarche; estrogen withdrawal during menstruation is a common migraine trigger; estrogen administra- tion in oral contraceptives and hormone replacement therapy can trigger migraines; migraines typically decrease during the second and third trimesters of pregnancy, when estrogen levels are high; migraines are common immediately postpartum, with the precipi- tous drop in estrogen levels; and migraines generally improve with physiological menopause. Exactly how changes in estrogen levels influence migraine is not understood. Among numerous effects, fluctuations in estrogen levels can result in changes in prostaglandins and the uterus, prolactin release, opioid regula- tion, and melatonin secretion. These fluctuations can also cause changes in neurotransmitters, including the catecholamines, noradrenaline, serotonin, dopamine, and endorphins (2). This chapter reviews some important headache issues for women, including menstrual migraine, menopause and migraine, oral contraceptive use in migraineurs, and headaches during pregnancy and postpartum. MENSTRUAL MIGRAINE The reported prevalence of menstrual migraine varies from 4% to 73%, depending on the criteria used for the timing of the attack. -

Barbiturates
URINE DRUG TEST INFORMATION SHEET BARBITURATES Classification: Central nervous system depressants insomnia, anxiety or tension due to their very high (CNS Depressants) risk of physical dependence and fatal overdose. Due to the structural nature of barbiturates, the duration Background: Barbiturates are a group of drugs of action does not always correlate well with the that act as central nervous system depressants. biological half-life. Opiates, benzodiazepines and alcohol are also CNS depressants, and like their use, the effect seems to Physiological Effects: Lowered blood pressure, the user as an overall sense of calm. Barbiturates respiratory depression, fatigue, fever, impaired were introduced in 1903, dominating the sedative- coordination, nystagmus, slurred speech and ataxia hypnotic market for the first half of the twentieth century. Unfortunately, because barbiturates have Psychological Effects: Drowsiness, dizziness, unusual a relatively low therapeutic-to-toxic index and excitement, irritability, poor concentration, sedation, substantial potential for abuse, they quickly became confusion, impaired judgment, addiction, euphoria, a major health problem. Barbiturates are commonly decreased anxiety and a loss of inhibition abused for their sedative properties and widespread Toxicity: Barbiturates are especially more availability. The introduction of benzodiazepines in dangerous when abused with alcohol, opiates and the 1960s quickly supplanted the barbiturates due benzodiazepines because they act on the same to their higher safety -

Maternal Medication and Herbal Use and Risk for Hypospadias: Data from the National Birth Defects Prevention Study, 1997--2007
HHS Public Access Author manuscript Author Manuscript Author ManuscriptPharmacoepidemiol Author Manuscript Drug Author Manuscript Saf. Author manuscript; available in PMC 2014 July 01. Published in final edited form as: Pharmacoepidemiol Drug Saf. 2013 July ; 22(7): 783–793. doi:10.1002/pds.3448. Maternal Medication and Herbal Use and Risk for Hypospadias: Data from the National Birth Defects Prevention Study, 1997--2007 Jennifer N. Lind1, Sarah C. Tinker1, Cheryl S. Broussard1, Jennita Reefhuis1, Suzan L. Carmichael2, Margaret A. Honein1, Richard S. Olney1, Samantha E. Parker3, and Martha M. Werler3 for the National Birth Defects Prevention Study 1National Center on Birth Defects and Developmental Disabilities, Centers for Disease Control and Prevention, Atlanta, Georgia, USA 2Department of Pediatrics, Stanford University, Stanford, California, USA 3Slone Epidemiology Center at Boston University, Boston, Massachusetts, USA Abstract Purpose—Investigate associations between maternal use of common medications and herbals during early pregnancy and risk for hypospadias in male infants. Methods—We used data from the National Birth Defects Prevention Study, a multi-site, population-based, case-control study. We analyzed data from 1,537 infants with second-or third- degree isolated hypospadias and 4,314 liveborn male control infants without major birth defects, with estimated dates of delivery from 1997–2007. Exposure was reported use of prescription or over-the-counter medications or herbal products, from 1 month before to 4 months after conception. Adjusted odds ratios (aORs) and 95% confidence intervals (CI) were estimated using multivariable logistic regression, adjusting for maternal age, race/ethnicity, education, pre- pregnancy BMI, previous live births, maternal sub-fertility, study site, and year. -

List of Formulary Drug Removals
July 2021 Formulary Drug Removals Below is a list of medicines by drug class that have been removed from your plan’s formulary. If you continue using one of the drugs listed below and identified as a Formulary Drug Removal, you may be required to pay the full cost. If you are currently using one of the formulary drug removals, ask your doctor to choose one of the generic or brand formulary options listed below. Category Formulary Drug Formulary Options Drug Class Removals Acromegaly SANDOSTATIN LAR SOMATULINE DEPOT SIGNIFOR LAR SOMAVERT Allergies dexchlorpheniramine levocetirizine Antihistamines Diphen Elixir RyClora CARBINOXAMINE TABLET 6 MG Allergies BECONASE AQ flunisolide spray, fluticasone spray, mometasone spray, DYMISTA Nasal Steroids / Combinations OMNARIS QNASL ZETONNA Anticonvulsants topiramate ext-rel capsule carbamazepine, carbamazepine ext-rel, clobazam, divalproex sodium, (generics for QUDEXY XR only) divalproex sodium ext-rel, gabapentin, lamotrigine, lamotrigine ext-rel, levetiracetam, levetiracetam ext-rel, oxcarbazepine, phenobarbital, phenytoin, phenytoin sodium extended, primidone, rufinamide, tiagabine, topiramate, valproic acid, zonisamide, FYCOMPA, OXTELLAR XR, TROKENDI XR, VIMPAT, XCOPRI BANZEL SUSPENSION clobazam, lamotrigine, rufinamide, topiramate, TROKENDI XR ONFI SABRIL vigabatrin ZONEGRAN carbamazepine, carbamazepine ext-rel, divalproex sodium, divalproex sodium ext-rel, gabapentin, lamotrigine, lamotrigine ext-rel, levetiracetam, levetiracetam ext-rel, oxcarbazepine, phenobarbital, phenytoin, phenytoin sodium -

High-Risk Medications in the Elderly
High-Risk Medications in the Elderly The Centers for Medicare & Medicaid Services (CMS) contracted with the National Committee for Quality Assurance (NCQA) to develop clinical strategies to monitor and evaluate the quality of care provided to Medicare beneficiaries. The NCQA’s Geriatric Measurement Advisory Panel identified several categories of medications that have an increased risk of adverse effects to elderly patients. The enclosed chart identifies several key medication categories that CMS and NCQA are monitoring. In an effort to ensure patients’ safety, many of our clients have established pre-authorization protocols for those prescriptions for high risk medications in patients older than 65 years of age. Since pharmacists have a very important role in patient care, we want you to be part of this safety initiative. We strongly encourage that you contact the prescriber when your elderly patient is requesting a new or refilled prescription of a high-risk medication listed on the below chart. Category High Risk Medications Alternatives Analgesics butalbital/APAP Mild Pain: butalbital/APAP/caffeine (ESGIC, FIORICET) acetaminophen, codeine, short-term NSAIDs butalbital /APAP/caffeine/codeine Moderate/Severe Pain: butalbital/ASA/caffeine (FIORINAL) tramadol (ULTRAM), tramadol/APAP* (ULTRACET), butalbital/ASA/caffeine/codeine morphine sulfate (MS CONTIN), ketorolac (TORADOL) hydrocodone/APAP (VICODIN, etc.), oxycodone indomethacin (INDOCIN) (OXYIR), oxycodone/APAP (PERCOCET), fentanyl meperidina (DEMEROL) patch (DURAGESIC), OXYCONTIN -

Review Memorandum
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY ASSAY ONLY TEMPLATE A. 510(k) Number: k092268 B. Purpose for Submission: New assay C. Measurand: Barbiturates D. Type of Test: Homogeneous enzyme immunoassay – qualitative and semi- quantitative E. Applicant: Randox Laboratories Ltd. F. Proprietary and Established Names: Randox Barbiturates assay Randox Multidrug Calibrator Set Randox Multidrug Controls, Levels 1 and 2 G. Regulatory Information: 1. Regulation section: Product Code Classification Regulation Section Panel DIS Class II 21 CFR § 862.3200, 91-Toxicology Barbiturate test system DLJ Class II 21 CFR § 862.3200, 91-Toxicology Calibrators, Drug specific LAS Class I, reserved 21 CFR 862.3280 91-Toxicology Clinical Toxicology control material 1 H. Intended Use: 1. Intended use(s): See Indications for use below. 2. Indication(s) for use: Randox Barbiturates Assay: The Randox Laboratories Ltd. Barbiturates Assay is an in vitro diagnostic test for the detection of Barbiturates, in human urine on the Rx Imola and Rx Daytona. The cutoff for secobarbital is 200 ng/mL. This in vitro diagnostic device is intended for prescription use only. The semi-quantitative mode is for purpose of (1) enabling laboratories to determine an appropriate dilution of the specimen for confirmation by a confirmatory method such as GCMS Or (2) permitting laboratories to establish quality control procedures. This assay provides only a preliminary analytical result. A more specific alternative chemical method must be used in order to obtain a confirmed analytical result. Gas chromatograph/mass spectrometry (GC/MS) is the preferred confirmatory method. Clinical consideration and professional judgment should be exercised with any drug of abuse test result, particularly when the preliminary result is positive.