Progesterone Receptors: Form and Function in Brain
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Risk of Breast Cancer After Stopping Menopausal Hormone Therapy In
Risk of breast cancer after stopping menopausal hormone therapy in the E3N cohort Agnès Fournier, Sylvie Mesrine, Laure Dossus, Marie-Christine Boutron-Ruault, Françoise Clavel-Chapelon, Nathalie Chabbert-Buffet To cite this version: Agnès Fournier, Sylvie Mesrine, Laure Dossus, Marie-Christine Boutron-Ruault, Françoise Clavel- Chapelon, et al.. Risk of breast cancer after stopping menopausal hormone therapy in the E3N cohort. Breast Cancer Research and Treatment, Springer Verlag, 2014, 145 (2), pp.535-43. 10.1007/s10549- 014-2934-6. inserm-01319982 HAL Id: inserm-01319982 https://www.hal.inserm.fr/inserm-01319982 Submitted on 23 May 2016 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. TITLE PAGE Risk of breast cancer after stopping menopausal hormone therapy in the E3N cohort Authors : Agnès Fournier1,2,3, Sylvie Mesrine1,2,3, Laure Dossus1,2,3, Marie-Christine Boutron- Ruault1,2,3, Françoise Clavel-Chapelon1,2,3, Nathalie Chabbert-Buffet4 Affiliations: 1. Inserm, Center for research in Epidemiology and Population Health, U1018, Nutrition, Hormones and Women’s Health team, F-94807, Villejuif, France 2. Univ Paris-Sud, UMRS 1018, F-94807, Villejuif, France 3. Institut Gustave Roussy, F-94805, Villejuif, France 4. -
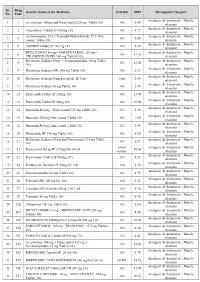
PMBJP Product.Pdf
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Megestrol Acetate/Melengestrol Acetate 2115 Adverse Effects and Precautions Megestrol Acetate Is Also Used in the Treatment of Ano- 16
Megestrol Acetate/Melengestrol Acetate 2115 Adverse Effects and Precautions Megestrol acetate is also used in the treatment of ano- 16. Mwamburi DM, et al. Comparing megestrol acetate therapy with oxandrolone therapy for HIV-related weight loss: similar As for progestogens in general (see Progesterone, rexia and cachexia (see below) in patients with cancer results in 2 months. Clin Infect Dis 2004; 38: 895–902. p.2125). The weight gain that may occur with meges- or AIDS. The usual dose is 400 to 800 mg daily, as tab- 17. Grunfeld C, et al. Oxandrolone in the treatment of HIV-associ- ated weight loss in men: a randomized, double-blind, placebo- trol acetate appears to be associated with an increased lets or oral suspension. A suspension of megestrol ace- controlled study. J Acquir Immune Defic Syndr 2006; 41: appetite and food intake rather than with fluid reten- tate that has an increased bioavailability is also availa- 304–14. tion. Megestrol acetate may have glucocorticoid ef- ble (Megace ES; Par Pharmaceutical, USA) and is Hot flushes. Megestrol has been used to treat hot flushes in fects when given long term. given in a dose of 625 mg in 5 mL daily for anorexia, women with breast cancer (to avoid the potentially tumour-stim- cachexia, or unexplained significant weight loss in pa- ulating effects of an oestrogen—see Malignant Neoplasms, un- Effects on carbohydrate metabolism. Megestrol therapy der Precautions of HRT, p.2075), as well as in men with hot 1-3 4 tients with AIDS. has been associated with hyperglycaemia or diabetes mellitus flushes after orchidectomy or anti-androgen therapy for prostate in AIDS patients being treated for cachexia. -

PHARMACEUTICAL APPENDIX to the TARIFF SCHEDULE 2 Table 1
Harmonized Tariff Schedule of the United States (2020) Revision 19 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2020) Revision 19 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names INN which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service CAS registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. -

U.S. Medical Eligibility Criteria for Contraceptive Use, 2016
Morbidity and Mortality Weekly Report Recommendations and Reports / Vol. 65 / No. 3 July 29, 2016 U.S. Medical Eligibility Criteria for Contraceptive Use, 2016 U.S. Department of Health and Human Services Centers for Disease Control and Prevention Recommendations and Reports CONTENTS Introduction ............................................................................................................1 Methods ....................................................................................................................2 How to Use This Document ...............................................................................3 Keeping Guidance Up to Date ..........................................................................5 References ................................................................................................................8 Abbreviations and Acronyms ............................................................................9 Appendix A: Summary of Changes from U.S. Medical Eligibility Criteria for Contraceptive Use, 2010 ...........................................................................10 Appendix B: Classifications for Intrauterine Devices ............................. 18 Appendix C: Classifications for Progestin-Only Contraceptives ........ 35 Appendix D: Classifications for Combined Hormonal Contraceptives .... 55 Appendix E: Classifications for Barrier Methods ..................................... 81 Appendix F: Classifications for Fertility Awareness–Based Methods ..... 88 Appendix G: Lactational -

Partial Agreement in the Social and Public Health Field
COUNCIL OF EUROPE COMMITTEE OF MINISTERS (PARTIAL AGREEMENT IN THE SOCIAL AND PUBLIC HEALTH FIELD) RESOLUTION AP (88) 2 ON THE CLASSIFICATION OF MEDICINES WHICH ARE OBTAINABLE ONLY ON MEDICAL PRESCRIPTION (Adopted by the Committee of Ministers on 22 September 1988 at the 419th meeting of the Ministers' Deputies, and superseding Resolution AP (82) 2) AND APPENDIX I Alphabetical list of medicines adopted by the Public Health Committee (Partial Agreement) updated to 1 July 1988 APPENDIX II Pharmaco-therapeutic classification of medicines appearing in the alphabetical list in Appendix I updated to 1 July 1988 RESOLUTION AP (88) 2 ON THE CLASSIFICATION OF MEDICINES WHICH ARE OBTAINABLE ONLY ON MEDICAL PRESCRIPTION (superseding Resolution AP (82) 2) (Adopted by the Committee of Ministers on 22 September 1988 at the 419th meeting of the Ministers' Deputies) The Representatives on the Committee of Ministers of Belgium, France, the Federal Republic of Germany, Italy, Luxembourg, the Netherlands and the United Kingdom of Great Britain and Northern Ireland, these states being parties to the Partial Agreement in the social and public health field, and the Representatives of Austria, Denmark, Ireland, Spain and Switzerland, states which have participated in the public health activities carried out within the above-mentioned Partial Agreement since 1 October 1974, 2 April 1968, 23 September 1969, 21 April 1988 and 5 May 1964, respectively, Considering that the aim of the Council of Europe is to achieve greater unity between its members and that this -

Combined Estrogen–Progestogen Menopausal Therapy
COMBINED ESTROGEN–PROGESTOGEN MENOPAUSAL THERAPY Combined estrogen–progestogen menopausal therapy was considered by previous IARC Working Groups in 1998 and 2005 (IARC, 1999, 2007). Since that time, new data have become available, these have been incorporated into the Monograph, and taken into consideration in the present evaluation. 1. Exposure Data 1.1.2 Progestogens (a) Chlormadinone acetate Combined estrogen–progestogen meno- Chem. Abstr. Serv. Reg. No.: 302-22-7 pausal therapy involves the co-administration Chem. Abstr. Name: 17-(Acetyloxy)-6-chlo- of an estrogen and a progestogen to peri- or ropregna-4,6-diene-3,20-dione menopausal women. The use of estrogens with IUPAC Systematic Name: 6-Chloro-17-hy- progestogens has been recommended to prevent droxypregna-4,6-diene-3,20-dione, acetate the estrogen-associated risk of endometrial Synonyms: 17α-Acetoxy-6-chloro-4,6- cancer. Evidence from the Women’s Health pregnadiene-3,20-dione; 6-chloro-Δ6-17- Initiative (WHI) of adverse effects from the use acetoxyprogesterone; 6-chloro-Δ6-[17α] of a continuous combined estrogen–progestogen acetoxyprogesterone has affected prescribing. Patterns of exposure Structural and molecular formulae, and relative are also changing rapidly as the use of hormonal molecular mass therapy declines, the indications are restricted, O CH and the duration of the therapy is reduced (IARC, 3 C 2007). CH3 CH3 O C 1.1 Identification of the agents CH3 H O 1.1.1 Estrogens HH For Estrogens, see the Monograph on O Estrogen-only Menopausal Therapy in this Cl volume. C23H29ClO4 Relative molecular mass: 404.9 249 IARC MONOGRAPHS – 100A (b) Cyproterone acetate Structural and molecular formulae, and relative Chem. -

Role of Androgens, Progestins and Tibolone in the Treatment of Menopausal Symptoms: a Review of the Clinical Evidence
REVIEW Role of androgens, progestins and tibolone in the treatment of menopausal symptoms: a review of the clinical evidence Maria Garefalakis Abstract: Estrogen-containing hormone therapy (HT) is the most widely prescribed and well- Martha Hickey established treatment for menopausal symptoms. High quality evidence confi rms that estrogen effectively treats hot fl ushes, night sweats and vaginal dryness. Progestins are combined with School of Women’s and Infants’ Health The University of Western Australia, estrogen to prevent endometrial hyperplasia and are sometimes used alone for hot fl ushes, King Edward Memorial Hospital, but are less effective than estrogen for this purpose. Data are confl icting regarding the role of Subiaco, Western Australia, Australia androgens for improving libido and well-being. The synthetic steroid tibolone is widely used in Europe and Australasia and effectively treats hot fl ushes and vaginal dryness. Tibolone may improve libido more effectively than estrogen containing HT in some women. We summarize the data from studies addressing the effi cacy, benefi ts, and risks of androgens, progestins and tibolone in the treatment of menopausal symptoms. Keywords: androgens, testosterone, progestins, tibolone, menopause, therapeutic Introduction Therapeutic estrogens include conjugated equine estrogens, synthetically derived piperazine estrone sulphate, estriol, dienoestrol, micronized estradiol and estradiol valerate. Estradiol may also be given transdermally as a patch or gel, as a slow release percutaneous implant, and more recently as an intranasal spray. Intravaginal estrogens include topical estradiol in the form of a ring or pessary, estriol in pessary or cream form, dienoestrol and conjugated estrogens in the form of creams. In some countries there is increasing prescribing of a combination of estradiol, estrone, and estriol as buccal lozenges or ‘troches’, which are formulated by private compounding pharmacists. -

WO 2016/180764 Al 17 November 2016 (17.11.2016) P O P CT
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2016/180764 Al 17 November 2016 (17.11.2016) P O P CT (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every A61K 31/565 (2006.01) A61K 31/567 (2006.01) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, (21) International Application Number: BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, PCT/EP20 16/060298 DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (22) International Filing Date: HN, HR, HU, ID, IL, IN, IR, IS, JP, KE, KG, KN, KP, KR, May 2016 (09.05.2016) KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, (25) Filing Language: English PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, SC, (26) Publication Language: English SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: 15 167521 .2 13 May 2015 (13.05.2015) (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (71) Applicant: BAYER OY [FI/FI]; Pansiontie 47, 20210 GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, Turku (FI). TZ, UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, (72) Inventors: HOLMBERG, Svante; Hirvikoirankatu 9 A 1, DK, EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, 20900 Turku (FI). -

Research Article Biomarkers & Prevention
Published OnlineFirst March 14, 2014; DOI: 10.1158/1055-9965.EPI-13-0944 Cancer Epidemiology, Research Article Biomarkers & Prevention Oral Contraceptives and Breast Cancer Risk Overall and by Molecular Subtype Among Young Women Elisabeth F. Beaber1,3, Kathleen E. Malone1,3, Mei-Tzu Chen Tang1, William E. Barlow1,4, Peggy L. Porter1,2,5, Janet R. Daling1,3, and Christopher I. Li1,3 Abstract Background: Evidence suggests that recent oral contraceptive (OC) use is associated with a small increased breast cancer risk; yet risks associated with contemporary OC preparations and by molecular subtype are not well characterized. Methods: We conducted a population-based case–control study of invasive breast cancer among women ages 20 to 44 residing in the Seattle–Puget Sound area from 2004 to 2010 (985 cases and 882 controls). We collected information on contraceptive use and participant characteristics via an in-person interview. Mul- tivariable-adjusted logistic regression was used to calculate odds ratios (OR) and 95% confidence intervals (CI). Results: Lifetime duration of OC use for 15 years was associated with an increased breast cancer risk (OR, 1.5; 95% CI, 1.1–2.2). Current OC use (within 1 year of reference date) for 5 years was associated with an increased risk (OR, 1.6; 95% CI, 1.1–2.5) and there were no statistically significant differences in risk by OC preparation. Risk magnitudes were generally greater among women ages 20 to 39, and for estrogen receptor– À À negative (ER ) and triple-negative breast cancer (current use for 5 years among ages 20–39: ER OR, 3.5; 95% CI, 1.3–9.0; triple-negative OR, 3.7; 95% CI, 1.2–11.8), although differences between groups were not statistically significant. -

Hormonal and Non-Hormonal Management of Vasomotor Symptoms: a Narrated Review
Central Journal of Endocrinology, Diabetes & Obesity Review Article Corresponding authors Orkun Tan, Department of Obstetrics and Gynecology, Division of Reproductive Endocrinology Hormonal and Non-Hormonal and Infertility, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd. Dallas, TX 75390 and ReproMed Fertility Center, 3800 San Management of Vasomotor Jacinto Dallas, TX 75204, USA, Tel: 214-648-4747; Fax: 214-648-8066; E-mail: [email protected] Submitted: 07 September 2013 Symptoms: A Narrated Review Accepted: 05 October 2013 Orkun Tan1,2*, Anil Pinto2 and Bruce R. Carr1 Published: 07 October 2013 1Department of Obstetrics and Gynecology, Division of Reproductive Endocrinology Copyright and Infertility, University of Texas Southwestern Medical Center, USA © 2013 Tan et al. 2Department of Obstetrics and Gynecology, Division of Reproductive Endocrinology and Infertility, ReproMed Fertility Center, USA OPEN ACCESS Abstract Background: Vasomotor symptoms (VMS; hot flashes, hot flushes) are the most common complaints of peri- and postmenopausal women. Therapies include various estrogens and estrogen-progestogen combinations. However, both physicians and patients became concerned about hormone-related therapies following publication of data by the Women’s Health Initiative (WHI) study and have turned to non-hormonal approaches of varying effectiveness and risks. Objective: Comparison of the efficacy of non-hormonal VMS therapies with estrogen replacement therapy (ERT) or ERT combined with progestogen (Menopausal Hormone Treatment; MHT) and the development of literature-based guidelines for the use of hormonal and non-hormonal VMS therapies. Methods: Pubmed, Cochrane Controlled Clinical Trials Register Database and Scopus were searched for relevant clinical trials that provided data on the treatment of VMS up to June 2013. -

Pharmaceutical Appendix to the Tariff Schedule 2
Harmonized Tariff Schedule of the United States (2006) – Supplement 1 (Rev. 1) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2006) – Supplement 1 (Rev. 1) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABACAVIR 136470-78-5 ACEXAMIC ACID 57-08-9 ABAFUNGIN 129639-79-8 ACICLOVIR 59277-89-3 ABAMECTIN 65195-55-3 ACIFRAN 72420-38-3 ABANOQUIL 90402-40-7 ACIPIMOX 51037-30-0 ABARELIX 183552-38-7 ACITAZANOLAST 114607-46-4 ABCIXIMAB 143653-53-6 ACITEMATE 101197-99-3 ABECARNIL 111841-85-1 ACITRETIN 55079-83-9 ABIRATERONE 154229-19-3 ACIVICIN 42228-92-2 ABITESARTAN 137882-98-5 ACLANTATE 39633-62-0 ABLUKAST 96566-25-5 ACLARUBICIN 57576-44-0 ABUNIDAZOLE 91017-58-2 ACLATONIUM NAPADISILATE 55077-30-0 ACADESINE 2627-69-2 ACODAZOLE 79152-85-5 ACAMPROSATE 77337-76-9 ACONIAZIDE 13410-86-1 ACAPRAZINE 55485-20-6 ACOXATRINE 748-44-7 ACARBOSE 56180-94-0 ACREOZAST 123548-56-1 ACEBROCHOL 514-50-1 ACRIDOREX 47487-22-9 ACEBURIC