Characterization of Incubation Experiments and Development of an Enrichment Culture Capable of Ammonium Oxidation Under Iron
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Taxonomic and Chemical Assessment of Exceptionally Abundant Rock Mine Biofilm
Taxonomic and chemical assessment of exceptionally abundant rock mine biofilm Karolina Tomczyk-Żak1, Paweª Szczesny2,3, Robert Gromadka4 and Urszula Zielenkiewicz1 1 Department of Microbial Biochemistry, Institute of Biochemistry and Biophysics, PAS, Warsaw, Poland 2 Faculty of Biology, Institute of Experimental Plant Biology and Biotechnology, University of Warsaw, Warsaw, Poland 3 Department of Bioinformatics, Institute of Biochemistry and Biophysics Polish Academy of Sciences, Warsaw, Poland 4 Laboratory of DNA Sequencing and Oligonucleotides Synthesis, Institute of Biochemistry and Biophysics, PAS, Warsaw, Poland ABSTRACT Background. An exceptionally thick biofilm covers walls of ancient gold and arsenic Zªoty Stok mine (Poland) in the apparent absence of organic sources of energy. Methods and Results. We have characterized this microbial community using culture- dependent and independent methods. We sequenced amplicons of the 16S rRNA gene obtained using generic primers and additional primers targeted at Archaea and Actinobacteria separately. Also, we have cultured numerous isolates from the biofilm on different media under aerobic and anaerobic conditions. We discovered very high biodiversity, and no single taxonomic group was dominant. The majority of almost 4,000 OTUs were classified above genus level indicating presence of novel species. Elemental analysis, performed using SEM-EDS and X-ray, of biofilm samples showed that carbon, sulphur and oxygen were not evenly distributed in the biofilm and that their presence is highly correlated. However, the distribution of arsenic and iron was more flat, and numerous intrusions of elemental silver and platinum were noted, indicating that microorganisms play a key role in releasing these elements from the rock. Submitted 13 April 2017 Conclusions. -
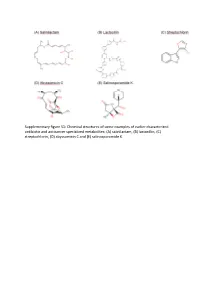
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

Download Download
http://wjst.wu.ac.th Natural Sciences Diversity Analysis of an Extremely Acidic Soil in a Layer of Coal Mine Detected the Occurrence of Rare Actinobacteria Megga Ratnasari PIKOLI1,*, Irawan SUGORO2 and Suharti3 1Department of Biology, Faculty of Science and Technology, Universitas Islam Negeri Syarif Hidayatullah Jakarta, Ciputat, Tangerang Selatan, Indonesia 2Center for Application of Technology of Isotope and Radiation, Badan Tenaga Nuklir Nasional, Jakarta Selatan, Indonesia 3Department of Chemistry, Faculty of Science and Computation, Universitas Pertamina, Simprug, Jakarta Selatan, Indonesia (*Corresponding author’s e-mail: [email protected], [email protected]) Received: 7 September 2017, Revised: 11 September 2018, Accepted: 29 October 2018 Abstract Studies that explore the diversity of microorganisms in unusual (extreme) environments have become more common. Our research aims to predict the diversity of bacteria that inhabit an extreme environment, a coal mine’s soil with pH of 2.93. Soil samples were collected from the soil at a depth of 12 meters from the surface, which is a clay layer adjacent to a coal seam in Tanjung Enim, South Sumatera, Indonesia. A culture-independent method, the polymerase chain reaction based denaturing gradient gel electrophoresis, was used to amplify the 16S rRNA gene to detect the viable-but-unculturable bacteria. Results showed that some OTUs that have never been found in the coal environment and which have phylogenetic relationships to the rare actinobacteria Actinomadura, Actinoallomurus, Actinospica, Streptacidiphilus, Aciditerrimonas, and Ferrimicrobium. Accordingly, the highly acidic soil in the coal mine is a source of rare actinobacteria that can be explored further to obtain bioactive compounds for the benefit of biotechnology. -

Methanogenic Activity in Río Tinto, a Terrestrial Mars Analogue R
Methanogenic activity in Río Tinto, a terrestrial Mars analogue R. Amils Centro de Biología Molecular Severo Ochoa (UAM-CSIC) y Centro de Astrobiología (INTA- CSIC) Frascati, noviembre 2009 new insides in the Mars exploration H2O on Mars methane (PFS) it can be concluded that on Mars there are sedimentary rocks that were formed in acidic conditions (acidic lakes or oceans) • possible terrestrial analogs: - submarine hydrothermalism - acidic environments to explore the deep sea requires expensive equipment (Alvin) natural acidic waters natural acidic environments: - areas with volcanic activity 0 SO2 + H2S ——> S + H2O - metal mining activities 3+ 2- + FeS2 + H2O —> Fe + SO4 + H in this case the extreme acidic conditions are promoted by biological activity geomicrobiology of metallic sulfides pyrite, molibdenite, tungstenite (thiosulfate mec.) 3+ 2- 2+ + FeS2+6Fe +3H2O → S2O3 +7Fe +6H 2- 3+ 2- 2+ + S2O3 +8Fe +5H2O → 2SO4 +8Fe +10H Rest of sulfides (polisulfide mec.) 3+ + 2+ 2+ 8MS+8Fe +8H → 8M +4H2Sn+8Fe (n≥2) 3+ o 2+ + 4H2Sn+8Fe → S8 +8Fe +8H o 2- + S8 +4H2O (S oxidizers) → SO4 +8H Bacterias come-meteoritos role of the microbial activity in the leaching of pyrite chemical 3+ reaction Fe Fe2+ microbial activity 2- + SO4 + H Rio Tinto rise at the heart of the Iberian Pyritic Belt Río Tinto is an acidic river, pH 2.3, 100 km long and with a high concentration of soluble metals the iron concentration at the origin is between 15-20 g/l and the sulfate is constant and around 15 g/l geoMICROBIOLOGY combination of conventional microbial ecology techniques and molecular ecology tools A B Phylogeny of acidophilic microorganisms detected in Rio Tinto Actinobacteria Cyanobacteria . -

Microbial Community Structure Dynamics in Ohio River Sediments During Reductive Dechlorination of Pcbs
University of Kentucky UKnowledge University of Kentucky Doctoral Dissertations Graduate School 2008 MICROBIAL COMMUNITY STRUCTURE DYNAMICS IN OHIO RIVER SEDIMENTS DURING REDUCTIVE DECHLORINATION OF PCBS Andres Enrique Nunez University of Kentucky Right click to open a feedback form in a new tab to let us know how this document benefits ou.y Recommended Citation Nunez, Andres Enrique, "MICROBIAL COMMUNITY STRUCTURE DYNAMICS IN OHIO RIVER SEDIMENTS DURING REDUCTIVE DECHLORINATION OF PCBS" (2008). University of Kentucky Doctoral Dissertations. 679. https://uknowledge.uky.edu/gradschool_diss/679 This Dissertation is brought to you for free and open access by the Graduate School at UKnowledge. It has been accepted for inclusion in University of Kentucky Doctoral Dissertations by an authorized administrator of UKnowledge. For more information, please contact [email protected]. ABSTRACT OF DISSERTATION Andres Enrique Nunez The Graduate School University of Kentucky 2008 MICROBIAL COMMUNITY STRUCTURE DYNAMICS IN OHIO RIVER SEDIMENTS DURING REDUCTIVE DECHLORINATION OF PCBS ABSTRACT OF DISSERTATION A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the College of Agriculture at the University of Kentucky By Andres Enrique Nunez Director: Dr. Elisa M. D’Angelo Lexington, KY 2008 Copyright © Andres Enrique Nunez 2008 ABSTRACT OF DISSERTATION MICROBIAL COMMUNITY STRUCTURE DYNAMICS IN OHIO RIVER SEDIMENTS DURING REDUCTIVE DECHLORINATION OF PCBS The entire stretch of the Ohio River is under fish consumption advisories due to contamination with polychlorinated biphenyls (PCBs). In this study, natural attenuation and biostimulation of PCBs and microbial communities responsible for PCB transformations were investigated in Ohio River sediments. Natural attenuation of PCBs was negligible in sediments, which was likely attributed to low temperature conditions during most of the year, as well as low amounts of available nitrogen, phosphorus, and organic carbon. -

Geomicrobiological Processes in Extreme Environments: a Review
202 Articles by Hailiang Dong1, 2 and Bingsong Yu1,3 Geomicrobiological processes in extreme environments: A review 1 Geomicrobiology Laboratory, China University of Geosciences, Beijing, 100083, China. 2 Department of Geology, Miami University, Oxford, OH, 45056, USA. Email: [email protected] 3 School of Earth Sciences, China University of Geosciences, Beijing, 100083, China. The last decade has seen an extraordinary growth of and Mancinelli, 2001). These unique conditions have selected Geomicrobiology. Microorganisms have been studied in unique microorganisms and novel metabolic functions. Readers are directed to recent review papers (Kieft and Phelps, 1997; Pedersen, numerous extreme environments on Earth, ranging from 1997; Krumholz, 2000; Pedersen, 2000; Rothschild and crystalline rocks from the deep subsurface, ancient Mancinelli, 2001; Amend and Teske, 2005; Fredrickson and Balk- sedimentary rocks and hypersaline lakes, to dry deserts will, 2006). A recent study suggests the importance of pressure in the origination of life and biomolecules (Sharma et al., 2002). In and deep-ocean hydrothermal vent systems. In light of this short review and in light of some most recent developments, this recent progress, we review several currently active we focus on two specific aspects: novel metabolic functions and research frontiers: deep continental subsurface micro- energy sources. biology, microbial ecology in saline lakes, microbial Some metabolic functions of continental subsurface formation of dolomite, geomicrobiology in dry deserts, microorganisms fossil DNA and its use in recovery of paleoenviron- Because of the unique geochemical, hydrological, and geological mental conditions, and geomicrobiology of oceans. conditions of the deep subsurface, microorganisms from these envi- Throughout this article we emphasize geomicrobiological ronments are different from surface organisms in their metabolic processes in these extreme environments. -

Alpine Soil Bacterial Community and Environmental Filters Bahar Shahnavaz
Alpine soil bacterial community and environmental filters Bahar Shahnavaz To cite this version: Bahar Shahnavaz. Alpine soil bacterial community and environmental filters. Other [q-bio.OT]. Université Joseph-Fourier - Grenoble I, 2009. English. tel-00515414 HAL Id: tel-00515414 https://tel.archives-ouvertes.fr/tel-00515414 Submitted on 6 Sep 2010 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. THÈSE Pour l’obtention du titre de l'Université Joseph-Fourier - Grenoble 1 École Doctorale : Chimie et Sciences du Vivant Spécialité : Biodiversité, Écologie, Environnement Communautés bactériennes de sols alpins et filtres environnementaux Par Bahar SHAHNAVAZ Soutenue devant jury le 25 Septembre 2009 Composition du jury Dr. Thierry HEULIN Rapporteur Dr. Christian JEANTHON Rapporteur Dr. Sylvie NAZARET Examinateur Dr. Jean MARTIN Examinateur Dr. Yves JOUANNEAU Président du jury Dr. Roberto GEREMIA Directeur de thèse Thèse préparée au sien du Laboratoire d’Ecologie Alpine (LECA, UMR UJF- CNRS 5553) THÈSE Pour l’obtention du titre de Docteur de l’Université de Grenoble École Doctorale : Chimie et Sciences du Vivant Spécialité : Biodiversité, Écologie, Environnement Communautés bactériennes de sols alpins et filtres environnementaux Bahar SHAHNAVAZ Directeur : Roberto GEREMIA Soutenue devant jury le 25 Septembre 2009 Composition du jury Dr. -

Composition of Free-Living, Aggregate-Associated and Sediment Surface-Associated Bacterial Communities in the German Wadden Sea
AQUATIC MICROBIAL ECOLOGY Vol. 38: 15–30, 2005 Published January 21 Aquat Microb Ecol Composition of free-living, aggregate-associated and sediment surface-associated bacterial communities in the German Wadden Sea Heike Stevens, Thorsten Brinkhoff, Meinhard Simon* Institute for Chemistry and Biology of the Marine Environment (ICBM), University of Oldenburg, PO Box 2503, 26111 Oldenburg, Germany ABSTRACT: The Wadden Sea is a tidal flat ecosystem at the land-sea interface of the southern North Sea with a high load of suspended matter and nutrients. Despite the general importance of microbial processes, the composition of bacterial communities in this ecosystem has been little studied. There- fore, we investigated the composition of the bacterial communities freely suspended (FL) in the bulk water, associated with suspended aggregates (AG) and with the oxic sediment surface (SE) over an annual cycle (April 1999 to June 2000) in the East Frisian Wadden Sea, by applying denaturing gra- dient gel electrophoresis (DGGE) of PCR-amplified 16S rRNA gene fragments. Wind strength, sus- pended particulate matter (SPM), particulate organic carbon (POC), chlorophyll, phaeopigments, and the composition of the phytoplankton community were also studied. The dynamics of these parame- ters covaried to various extents, but not systematically, with that of bacterial numbers and community composition. A cluster analysis revealed that the DGGE banding patterns of the 3 communities grouped separately even though band overlaps among the communities occurred. Some identical or highly similar phylotypes were found in the FL- and AG-associated communities, and in the AG- and SE-associated communities, as shown by sequence analysis of excised bands. -

Full Paper Ilumatobacter Fluminis Gen. Nov., Sp. Nov., a Novel Actinobacterium Isolated from the Sediment of an Estuary
J. Gen. Appl. Microbiol., 55, 201‒205 (2009) Full Paper Ilumatobacter fl uminis gen. nov., sp. nov., a novel actinobacterium isolated from the sediment of an estuary Atsuko Matsumoto,1 Hiroki Kasai,2 Yoshihide Matsuo,2 Satoshi Ōmura,1 Yoshikazu Shizuri,2 and Yōko Takahashi1,* 1 Kitasato Institute for Life Sciences, Kitasato University, Minato-ku, Tokyo 108‒8641, Japan 2 Marine Biotechnology Institute, Kitasato University, Kamaishi, Iwate 026‒0001, Japan (Received December 1, 2008; Accepted February 2, 2009) Bacterial strain YM22-133T was isolated from the sediment of an estuary and grew in media with an artifi cial seawater base. Strain YM22-133T was Gram-positive, aerobic, non-motile and rod shaped. The cell-wall peptidoglycan contained LL-DAP, glycine, alanine and hydroxyglutamate. The predominant menaquinone was MK-9 (H8), with MK-9 (H0), MK-9 (H2), MK-9 (H4) and MK-9 (H6) present as minor menaquinones. The G+C content of the genomic DNA from the strain was 68 mol%. Phylogenetic analysis of the 16S rRNA gene sequence showed that the strain is near- est to Acidimicrobium ferrooxidans DSM 10331T. However, the similarity is relatively low (87.1%) and the physiological characteristics are also different: Acidimicrobium ferrooxidans is thermo- tolerant and acidophilic. Therefore, strain YM22-133T can be classifi ed as a novel genus and species, Ilumatobacter fl uminis gen. nov., sp. nov. (type strain YM22-133T =DSM 18936T=MBIC 08263T). Key Words—Acidimicrobium; Actinobacteria; artifi cial sea water; Ilumatobacter fl uminis gen. nov., sp. nov. Introduction isolated as part of this study. Phylogenetic analysis on the basis of 16S rRNA gene sequence analysis showed Recently, bacteria isolated from marine environ- that the strain is most closely related to the genus Aci- ments have attracted attention due to the recognition dimicrobium (Clark and Norris, 1996). -

The Phylogenetic Composition and Structure of Soil Microbial Communities Shifts in Response to Elevated Carbon Dioxide
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by University of Minnesota Digital Conservancy The ISME Journal (2012) 6, 259–272 & 2012 International Society for Microbial Ecology All rights reserved 1751-7362/12 www.nature.com/ismej ORIGINAL ARTICLE The phylogenetic composition and structure of soil microbial communities shifts in response to elevated carbon dioxide Zhili He1, Yvette Piceno2, Ye Deng1, Meiying Xu1,3, Zhenmei Lu1,4, Todd DeSantis2, Gary Andersen2, Sarah E Hobbie5, Peter B Reich6 and Jizhong Zhou1,2 1Institute for Environmental Genomics, Department of Botany and Microbiology, University of Oklahoma, Norman, OK, USA; 2Ecology Department, Earth Sciences Division, Lawrence Berkeley National Laboratory, Berkeley, CA, USA; 3Guangdong Provincial Key Laboratory of Microbial Culture Collection and Application, Guangdong Institute of Microbiology, Guangzhou, China; 4College of Life Sciences, Zhejiang University, Hangzhou, China; 5Department of Ecology, Evolution, and Behavior, St Paul, MN, USA and 6Department of Forest Resources, University of Minnesota, St Paul, MN, USA One of the major factors associated with global change is the ever-increasing concentration of atmospheric CO2. Although the stimulating effects of elevated CO2 (eCO2) on plant growth and primary productivity have been established, its impacts on the diversity and function of soil microbial communities are poorly understood. In this study, phylogenetic microarrays (PhyloChip) were used to comprehensively survey the richness, composition and structure of soil microbial communities in a grassland experiment subjected to two CO2 conditions (ambient, 368 p.p.m., versus elevated, 560 p.p.m.) for 10 years. The richness based on the detected number of operational taxonomic units (OTUs) significantly decreased under eCO2. -

Altitudinal Patterns of Diversity and Functional Traits of Metabolically Active Microorganisms in Stream Biofilms
The ISME Journal (2015) 9, 2454–2464 © 2015 International Society for Microbial Ecology All rights reserved 1751-7362/15 www.nature.com/ismej ORIGINAL ARTICLE Altitudinal patterns of diversity and functional traits of metabolically active microorganisms in stream biofilms Linda Wilhelm1, Katharina Besemer2, Lena Fragner3, Hannes Peter4, Wolfram Weckwerth3 and Tom J Battin1,5 1Department of Limnology and Oceanography, Faculty of Life Sciences, University of Vienna, Vienna, Austria; 2School of Engineering, University of Glasgow, Glasgow, UK; 3Department of Ecogenomics and Systems Biology, University of Vienna, Vienna, Austria; 4Lake and Glacier Ecology Research Group, Institute of Ecology, University of Innsbruck, Innsbruck, Austria and 5Stream Biofilm and Ecosystem Research Laboratory, School of Architecture, Civil and Environmental Engineering, Ecole Polytechnique Fédérale de Lausanne, Lausanne, Switzerland Resources structure ecological communities and potentially link biodiversity to energy flow. It is commonly believed that functional traits (generalists versus specialists) involved in the exploitation of resources depend on resource availability and environmental fluctuations. The longitudinal nature of stream ecosystems provides changing resources to stream biota with yet unknown effects on microbial functional traits and community structure. We investigated the impact of autochthonous (algal extract) and allochthonous (spruce extract) resources, as they change along alpine streams from above to below the treeline, on microbial diversity, -

Microbial Community Composition in Acid Mine Drainage Lake of Xiang
This article was downloaded by: [Zhejiang University] On: 11 April 2013, At: 17:31 Publisher: Taylor & Francis Informa Ltd Registered in England and Wales Registered Number: 1072954 Registered office: Mortimer House, 37-41 Mortimer Street, London W1T 3JH, UK Geomicrobiology Journal Publication details, including instructions for authors and subscription information: http://www.tandfonline.com/loi/ugmb20 Microbial Community Composition in Acid Mine Drainage Lake of Xiang Mountain Sulfide Mine in Anhui Province, China Chunbo Hao a b , Lina Zhang a b , Lihua Wang a b , Siyuan Li a b & Hailiang Dong a c a Geomicrobiology Laboratory, State Key Laboratory of Geobiology and Environmental Geology, China University of Geosciences, Beijing, China b School of Water Resources and Environment, China University of Geosciences, Beijing, China c Department of Geology and Environmental Earth Science, Miami University, Oxford, Ohio, USA Accepted author version posted online: 18 Apr 2012.Version of record first published: 14 Sep 2012. To cite this article: Chunbo Hao , Lina Zhang , Lihua Wang , Siyuan Li & Hailiang Dong (2012): Microbial Community Composition in Acid Mine Drainage Lake of Xiang Mountain Sulfide Mine in Anhui Province, China, Geomicrobiology Journal, 29:10, 886-895 To link to this article: http://dx.doi.org/10.1080/01490451.2011.635762 PLEASE SCROLL DOWN FOR ARTICLE Full terms and conditions of use: http://www.tandfonline.com/page/terms-and-conditions This article may be used for research, teaching, and private study purposes. Any substantial or systematic reproduction, redistribution, reselling, loan, sub-licensing, systematic supply, or distribution in any form to anyone is expressly forbidden. The publisher does not give any warranty express or implied or make any representation that the contents will be complete or accurate or up to date.