Chiari Type II Malformation: Past, Present, and Future
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
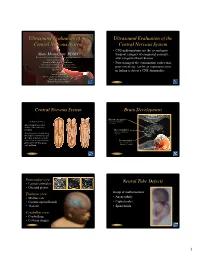
Ultrasound Evaluation of the Central Nervous System
Ultrasound Evaluation of the Ultrasound Evaluation of the Central Nervous System Central Nervous System ••CNSCNS malformations are the second most Mani Montazemi, RDMS frequent category of congenital anomaly, Director of Ultrasound Education & Quality Assurancee after congenital heart disease Baylor College of Medicine Division of Maternal-Fetal Medicine ••PoorPoor timing of the examination, rather than Department of Obstetrics and Gynecology Texas Children’s Hospital, Pavilion for Women poor sensitivity, can be an important factor Houston Texas & in failing to detect a CNS abnormality Clinical Instructor Thomas Jefferson University Hospital Radiology Department Fetal Head Philadelphia, Pennsylvania Fetal Head Central Nervous System Brain Development 9 -13 weeks Rhombencephalon 5th Menstrual Week •Gives rise to hindbrain •4th ventricle Arises from the posterior surface of the embryonic ectoderm Mesencephalon •Gives rise to midbrain A small groove is found along •Aqueduct the midline of the embryo and the edges of this groove fold over to form a neuro tube that Prosencephalon gives rise to the fetal spinal •Gives rise to forebrain rd cord and brain •Lateral & 3 ventricles Fetal Head Fetal Head Ventricular view Neural Tube Defects ••LateralLateral ventricles ••ChoroidChoroid plexus Group of malformations: Thalamic view • Anencephaly ••MidlineMidline falx •Anencephaly ••CavumCavum septiseptipellucidi pellucidi ••CephalocelesCephaloceles ••ThalamiThalami ••SpinaSpina bifida Cerebellar view ••CerebellumCerebellum ••CisternaCisterna magna Fetal -

Reorganisation of the Visual Cortex in Callosal Agenesis and Colpocephaly
Journal of Clinical Neuroscience (2000) 7(1), 13–15 © 2000 Harcourt Publishers Ltd DOI: 10.1054/ jocn.1998.0105, available online at http://www.idealibrary.com on Clinical study Reorganisation of the visual cortex in callosal agenesis and colpocephaly Richard G. Bittar1,2 MB BS, Alain Ptito1 PHD, Serge O. Dumoulin1, Frederick Andermann1 MD FRCP(C), David C. Reutens1 MD FRACP 1Montreal Neurological Institute and Hospital and Department of Neurology and Neurosurgery, McGill University, Montreal, Canada 2Department of Anatomy and Histology, University of Sydney, Australia Summary Structural defects involving eloquent regions of the cerebral cortex may be accompanied by abnormal localisation of function. Using functional magnetic resonance imaging (fMRI), we studied the organisation of the visual cortex in a patient with callosal agenesis and colpocephaly, whose visual acuity and binocular visual fields were normal. The stimulus used was a moving grating confined to one hemifield, on a background of moving dots. In addition to activation patterns elicited by stimulation of each hemifield in the patient, the activation pattern was compared to that seen in six normal volunteers. fMRI demonstrated large scale reorganisation of visual cortical areas in the left hemisphere, and fewer activation foci were observed in both occipital lobes when compared with normal subjects. © 2000 Harcourt Publishers Ltd Keywords: functional magnetic resonance imaging, vision, colpocephaly, callosal agenesis, reorganisation INTRODUCTION showed interictal slow wave activity throughout the right hemi- sphere and seizures with onset in the right temporo-frontal region. An exciting application of non-invasive functional brain mapping Ictal Tc99m HMPAO single photon emission computed tomography techniques such as functional magnetic resonance imaging (fMRI) (SPECT) revealed a diffuse increase in perfusion within the right is the study of cortical sensory and motor reorganisation following cerebral hemisphere. -

The Chiari Malformations *
J Neurol Neurosurg Psychiatry: first published as 10.1136/jnnp.72.suppl_2.ii38 on 1 June 2002. Downloaded from THE CHIARI MALFORMATIONS Donald M Hadley ii38* J Neurol Neurosurg Psychiatry 2002;72(Suppl II):ii38–ii40 r Hans Chiari1 first described three hindbrain disorders associated with hydrocephalus in 1891. They have neither an anatomical nor embryological correlation with each other, but Dthey all involve the cerebellum and spinal cord and are thought to belong to the group of abnormalities that result from failure of normal dorsal induction. These include neural tube defects, cephaloceles, and spinal dysraphic abnormalities. Symptoms range from headache, sensory changes, vertigo, limb weakness, ataxia and imbalance to hearing loss. Only those with a type I Chiari malformation may be born grossly normal. The abnormalities are best shown on midline sagittal T1 weighted magnetic resonance imaging (MRI),2 but suspicious features on routine axial computed tomographic brain scans (an abnormal IVth ventricle, a “full” foramen magnum, and absent cisterna magna) should be recognised and followed up with MRI. c CHIARI I This is the mildest of the hindbrain malformations and is characterised by displacement of deformed cerebellar tonsils more than 5 mm caudally through the foramen magnum.3 The brain- stem and IVth ventricle retain a relatively normal position although the IVth ventricle may be small copyright. and slightly distorted (fig 1). A number of subgroups have been defined. c In the first group, intrauterine hydrocephalus causes tonsillar herniation. Once myelinated the tonsils retain this pointed configuration and ectopic position. Patients tend to present in child- hood with hydrocephalus and usually with syringomyelia. -

Unusual Presentation of Congenital Dermal Sinus: Tethered Spinal Cord with Intradural Epidermoid and Dual Paramedian Cutaneous Ostia
Neurosurg Focus 33 (4):E5, 2012 Unusual presentation of congenital dermal sinus: tethered spinal cord with intradural epidermoid and dual paramedian cutaneous ostia Case report EFREM M. COX, M.D., KATHLeeN E. KNUDSON, M.D., SUNIL MANJILA, M.D., AND ALAN R. COHEN, M.D. Division of Pediatric Neurosurgery, Rainbow Babies and Children’s Hospital; and Department of Neurological Surgery, The Neurological Institute, University Hospitals Case Medical Center, Cleveland, Ohio The authors present the first report of spinal congenital dermal sinus with paramedian dual ostia leading to 2 intradural epidermoid cysts. This 7-year-old girl had a history of recurrent left paramedian lumbosacral subcutaneous abscesses, with no chemical or pyogenic meningitis. Admission MRI studies demonstrated bilateral lumbar dermal sinus tracts and a tethered spinal cord. At surgery to release the tethered spinal cord the authors encountered para- median dermal sinus tracts with dual ostia, as well as 2 intradural epidermoid cysts that were not readily apparent on MRI studies. Congenital dermal sinus should be considered in the differential diagnosis of lumbar subcutaneous abscesses, even if the neurocutaneous signatures are located off the midline. (http://thejns.org/doi/abs/10.3171/2012.8.FOCUS12226) KEY WORDS • tethered spinal cord • epidermoid cyst • neural tube defect • congenital dermal sinus • dual ostia ONGENITAL dermal sinus tracts of the spine are a Spinal congenital epidermoid cysts arise from epi- rare form of spinal dysraphism, and are hypoth- thelial inclusion -

Approach to Brain Malformations
Approach to Brain Malformations A General Imaging Approach to Brain CSF spaces. This is the basis for development of the Dandy- Malformations Walker malformation; it requires abnormal development of the cerebellum itself and of the overlying leptomeninges. Whenever an infant or child is referred for imaging because of Looking at the midline image also gives an idea of the relative either seizures or delayed development, the possibility of a head size through assessment of the craniofacial ratio. In the brain malformation should be carefully investigated. If the normal neonate, the ratio of the cranial vault to the face on child appears dysmorphic in any way (low-set ears, abnormal midline images is 5:1 or 6:1. By 2 years, it should be 2.5:1, and facies, hypotelorism), the likelihood of an underlying brain by 10 years, it should be about 1.5:1. malformation is even higher, but a normal appearance is no guarantee of a normal brain. In all such cases, imaging should After looking at the midline, evaluate the brain from outside be geared toward showing a structural abnormality. The to inside. Start with the cerebral cortex. Is the thickness imaging sequences should maximize contrast between gray normal (2-3 mm)? If it is too thick, think of pachygyria or matter and white matter, have high spatial resolution, and be polymicrogyria. Is the cortical white matter junction smooth or acquired as volumetric data whenever possible so that images irregular? If it is irregular, think of polymicrogyria or Brain: Pathology-Based Diagnoses can be reformatted in any plane or as a surface rendering. -

Facts About Spina Bifida 1995-2009 Bifida 1995-2009
Facts about Spina Facts about Spina Bifida 1995-2009 Bifida 1995-2009 January 9, 2012 Definition and Types United States Estimates Spina Bifida is a type of neural tube defect where the Each year, about 1,500 babies are born with Spina Bifida in spine does not form properly within the first month of the U.S. The lifetime medical cost associated with caring for pregnancy. There are three types of Spina Bifida: Oc- a child that has been diagnosed with Spina Bifida is estimated 4 culta, Meningocele, and Myelomeningocele. at $460,923 in 2009. Occulta, the mildest form, occurs when there is a In 1992, the Centers for Disease Control and Prevention division between the vertebrae. However, the spi- (CDC) recommended that women of childbearing age con- nal cord does not protrude through the back. The sume 400 micrograms of synthetic folic acid daily. Subse- spinal cord and the nerve usually are normal. This quently, the Food and Drug Administration (FDA) required type of spina bifida usually does not cause any dis- the addition of folate to enriched cereal-grain products by abilities. January 1998. Since then, the incident rate for Spina Bifida of . Meningocele, the least common form, occurs when post-fortification (1998-2006) was 3.68 cases per 10,000 live the covering for the spinal cord but not the spinal births, declined 31% from the pre-fortification (1995-1996) cord protrudes through the back. There is usually rate of 5.04 cases per 10,000 live births.4 little or no nerve damage. This type of spina bifida can cause minor disabilities. -

Colpocephaly Diagnosed in a Neurologically Normal Adult in the Emergency Department
CASE REPORT Colpocephaly Diagnosed in a Neurologically Normal Adult in the Emergency Department Christopher Parker, DO University of Illinois College of Medicine, Department of Emergency Medicine, Wesley Eilbert, MD Chicago, Illinois Timothy Meehan, MD Christopher Colbert, DO Section Editor: Rick A. McPheeters, DO Submission history: Submitted July 25, 2019; Revision received September 18, 2019; Accepted September 26, 2019 Electronically published October 21, 2019 Full text available through open access at http://escholarship.org/uc/uciem_cpcem DOI: 10.5811/cpcem.2019.9.44646 Colpocephaly is a form of congenital ventriculomegaly characterized by enlarged occipital horns of the lateral ventricles with associated neurologic abnormalities. The diagnosis of colpocephaly is typically made in infancy. Its diagnosis in adulthood without associated clinical symptoms is exceptionally rare. We report a case of colpocephaly diagnosed incidentally in an adult without neurologic abnormalities in the emergency department. To our knowledge, this is only the ninth reported case in an asymptomatic adult and the first to be described in the emergency medicine literature. [Clin Pract Cases Emerg Med. 2019;3(4):421–424.] INTRODUCTION been treated at four different EDs in the two weeks prior to Colpocephaly is a rare form of congenital presentation for the headaches, but no imaging studies had ventriculomegaly often associated with partial or been performed. The patient had no psychiatric history. His complete agenesis of the corpus callosum. Diagnosis is highest level of education was a high school diploma, and typically made in infancy due to associated neurological he was unemployed. and neurodevelopmental disorders.1,2 Initial discovery On arrival, the patient was afebrile with pulse, blood in adulthood is exceedingly rare.3-9 When identified pressure, and respiratory rate all within the normal range. -

Maternal Vitamin B12 Status and Risk of Neural Tube Defects in a Population with High Neural Tube Defect Prevalence and No Folic Acid Fortification Anne M
Maternal Vitamin B12 Status and Risk of Neural Tube Defects in a Population With High Neural Tube Defect Prevalence and No Folic Acid Fortification Anne M. Molloy, Peadar N. Kirke, James F. Troendle, Helen Burke, Marie Sutton, Lawrence C. Brody, John M. Scott and James L. Mills Pediatrics 2009;123;917-923 DOI: 10.1542/peds.2008-1173 The online version of this article, along with updated information and services, is located on the World Wide Web at: http://www.pediatrics.org/cgi/content/full/123/3/917 PEDIATRICS is the official journal of the American Academy of Pediatrics. A monthly publication, it has been published continuously since 1948. PEDIATRICS is owned, published, and trademarked by the American Academy of Pediatrics, 141 Northwest Point Boulevard, Elk Grove Village, Illinois, 60007. Copyright © 2009 by the American Academy of Pediatrics. All rights reserved. Print ISSN: 0031-4005. Online ISSN: 1098-4275. Downloaded from www.pediatrics.org. Provided by Trinity Health Sciences Centre on November 4, 2009 ARTICLE Maternal Vitamin B12 Status and Risk of Neural Tube Defects in a Population With High Neural Tube Defect Prevalence and No Folic Acid Fortification Anne M. Molloy, PhDa, Peadar N. Kirke, FFPHMIb, James F. Troendle, PhDc, Helen Burke, BSocScb, Marie Sutton, MB, MPHb, Lawrence C. Brody, PhDd, John M. Scott, ScDe, James L. Mills, MD, MSc Schools of aMedicine and eImmunology and Biochemistry and Immunology, Trinity College, Dublin, Ireland; bChild Health Epidemiology Unit, Health Research Board, Dublin, Ireland; cDivision of Epidemiology, Statistics, and Prevention Research, Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, Maryland; dMolecular Pathogenesis Section, Genome Technology Branch, National Human Genome Research Institute, Bethesda, Maryland The authors have indicated they have no financial relationships relevant to this article to disclose. -

Congenital Disorders of Glycosylation from a Neurological Perspective
brain sciences Review Congenital Disorders of Glycosylation from a Neurological Perspective Justyna Paprocka 1,* , Aleksandra Jezela-Stanek 2 , Anna Tylki-Szyma´nska 3 and Stephanie Grunewald 4 1 Department of Pediatric Neurology, Faculty of Medical Science in Katowice, Medical University of Silesia, 40-752 Katowice, Poland 2 Department of Genetics and Clinical Immunology, National Institute of Tuberculosis and Lung Diseases, 01-138 Warsaw, Poland; [email protected] 3 Department of Pediatrics, Nutrition and Metabolic Diseases, The Children’s Memorial Health Institute, W 04-730 Warsaw, Poland; [email protected] 4 NIHR Biomedical Research Center (BRC), Metabolic Unit, Great Ormond Street Hospital and Institute of Child Health, University College London, London SE1 9RT, UK; [email protected] * Correspondence: [email protected]; Tel.: +48-606-415-888 Abstract: Most plasma proteins, cell membrane proteins and other proteins are glycoproteins with sugar chains attached to the polypeptide-glycans. Glycosylation is the main element of the post- translational transformation of most human proteins. Since glycosylation processes are necessary for many different biological processes, patients present a diverse spectrum of phenotypes and severity of symptoms. The most frequently observed neurological symptoms in congenital disorders of glycosylation (CDG) are: epilepsy, intellectual disability, myopathies, neuropathies and stroke-like episodes. Epilepsy is seen in many CDG subtypes and particularly present in the case of mutations -

Bilateral Posterior Periventricular Nodular Heterotopia: a Recognizable Cortical Malformation with a Spectrum of Associated Brain Abnormalities
ORIGINAL RESEARCH PEDIATRICS Bilateral Posterior Periventricular Nodular Heterotopia: A Recognizable Cortical Malformation with a Spectrum of Associated Brain Abnormalities S.A. Mandelstam, R.J. Leventer, A. Sandow, G. McGillivray, M. van Kogelenberg, R. Guerrini, S. Robertson, S.F. Berkovic, G.D. Jackson, and I.E. Scheffer ABSTRACT BACKGROUND AND PURPOSE: Bilateral posterior PNH is a distinctive complex malformation with imaging features distinguishing it from classic bilateral PNH associated with FLNA mutations. The purpose of this study was to define the imaging features of posterior bilateral periventricular nodular heterotopia and to determine whether associated brain malformations suggest specific subcategories. MATERIALS AND METHODS: We identified a cohort of 50 patients (31 females; mean age, 13 years) with bilateral posterior PNH and systematically reviewed and documented associated MR imaging abnormalities. Patients were negative for mutations of FLNA. RESULTS: Nodules were often noncontiguous (n ϭ 28) and asymmetric (n ϭ 31). All except 1 patient showed associated developmental brain abnormalities involving a spectrum of posterior structures. A range of posterior fossa abnormalities affected the cerebellum, including cerebellar malformations and posterior fossa cysts (n ϭ 38). Corpus callosum abnormalities (n ϭ 40) ranged from mild dysplasia to agenesis. Posterior white matter volume was decreased (n ϭ 22), and colpocephaly was frequent (n ϭ 26). Most (n ϭ 40) had associated cortical abnormalities ranging from minor to major (polymicrogyria), typically located in the cortex overlying the PNH. Abnormal Sylvian fissure morphology was common (n ϭ 27), and hippocampal abnormalities were frequent (n ϭ 37). Four family cases were identified—2 with concordant malformation patterns and 2 with discordant malformation patterns. -

Chiari Malformation by Ryan W Y Lee MD (Dr
Chiari malformation By Ryan W Y Lee MD (Dr. Lee of Shriners Hospitals for Children in Honolulu and the John A Burns School of Medicine at the University of Hawaii has no relevant financial relationships to disclose.) Originally released August 8, 1994; last updated March 9, 2017; expires March 9, 2020 Introduction This article includes discussion of Chiari malformation, Arnold-Chiari deformity, and Arnold-Chiari malformation. The foregoing terms may include synonyms, similar disorders, variations in usage, and abbreviations. Overview Chiari malformation describes a group of structural defects of the cerebellum, characterized by brain tissue protruding into the spinal canal. Chiari malformations are often associated with myelomeningocele, hydrocephalus, syringomyelia, and tethered cord syndrome. Although studies of etiology are few, an increasing number of specific genetic syndromes are found to be associated with Chiari malformations. Management primarily targets supportive care and neurosurgical intervention when necessary. Renewed effort to address current deficits in Chiari research involves work groups targeted at pathophysiology, symptoms and diagnosis, engineering and imaging analysis, treatment, pediatric issues, and related conditions. In this article, the author discusses the many aspects of diagnosis and management of Chiari malformation. Key points • Chiari malformation describes a group of structural defects of the cerebellum, characterized by brain tissue protruding into the spinal canal. • Chiari malformations are often associated -

Pushing the Limits of Prenatal Ultrasound: a Case of Dorsal Dermal Sinus Associated with an Overt Arnold–Chiari Malformation and a 3Q Duplication
reproductive medicine Case Report Pushing the Limits of Prenatal Ultrasound: A Case of Dorsal Dermal Sinus Associated with an Overt Arnold–Chiari Malformation and a 3q Duplication Olivier Leroij 1, Lennart Van der Veeken 2,*, Bettina Blaumeiser 3 and Katrien Janssens 3 1 Faculty of Medicine, University of Antwerp, 2610 Wilrijk, Belgium; [email protected] 2 Department of Obstetrics and Gynaecology, University Hospital Antwerp, 2650 Edegem, Belgium 3 Department of Medical Genetics, University Hospital and University of Antwerp, 2650 Edegem, Belgium; [email protected] (B.B.); [email protected] (K.J.) * Correspondence: [email protected] Abstract: We present a case of a fetus with cranial abnormalities typical of open spina bifida but with an intact spine shown on both ultrasound and fetal MRI. Expert ultrasound examination revealed a very small tract between the spine and the skin, and a postmortem examination confirmed the diagnosis of a dorsal dermal sinus. Genetic analysis found a mosaic 3q23q27 duplication in the form of a marker chromosome. This case emphasizes that meticulous prenatal ultrasound examination has the potential to diagnose even closed subtypes of neural tube defects. Furthermore, with cerebral anomalies suggesting a spina bifida, other imaging techniques together with genetic tests and measurement of alpha-fetoprotein in the amniotic fluid should be performed. Citation: Leroij, O.; Van der Veeken, Keywords: dorsal dermal sinus; Arnold–Chiari anomaly; 3q23q27 duplication; mosaic; marker chro- L.; Blaumeiser, B.; Janssens, K. mosome Pushing the Limits of Prenatal Ultrasound: A Case of Dorsal Dermal Sinus Associated with an Overt Arnold–Chiari Malformation and a 3q 1.