Intensive Induction Chemotherapy Followed by Early High-Dose
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Summary of Product Characteristics
Health Products Regulatory Authority Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT NIPENT 10 mg powder for solution for injection, powder for solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION One vial contains 10 mg Pentostatin. When reconstituted (see Section 6.6), the resulting solution contains pentostatin 2 mg/ml. For a full list of excipients see section 6.1. 3 PHARMACEUTICAL FORM Powder for solution for injection, powder for solution for infusion. The vials contain a solid white to off-white cake or powder. The pH of reconstituted solution is 7.0 – 8.2. 4 CLINICAL PARTICULARS 4.1 Therapeutic Indications Pentostatin is indicated as single agent therapy in the treatment of adult patients with hairy cell leukaemia. 4.2 Posology and method of administration Pentostatin is indicated for adult patients. Administration to patient It is recommended that patients receive hydration with 500 to 1,000 ml of 5% glucose only or 5% glucose in 0.18% or 0.9% saline or glucose 3.3% in 0.3% saline or 2.5% glucose in 0.45% saline or equivalent before pentostatin administration. An additional 500 ml of 5% glucose only or 5% glucose in 0.18% or 0.9% saline or 2.5% glucose in 0.45% saline or equivalent should be administered after pentostatin is given. The recommended dosage of pentostatin for the treatment of hairy cell leukaemia is 4 mg/m2 in a single administration every other week. Pentostatin may be given intravenously by bolus injection or diluted in a larger volume and given over 20 to 30 minutes. -

Cancer Drug Pharmacology Table
CANCER DRUG PHARMACOLOGY TABLE Cytotoxic Chemotherapy Drugs are classified according to the BC Cancer Drug Manual Monographs, unless otherwise specified (see asterisks). Subclassifications are in brackets where applicable. Alkylating Agents have reactive groups (usually alkyl) that attach to Antimetabolites are structural analogues of naturally occurring molecules DNA or RNA, leading to interruption in synthesis of DNA, RNA, or required for DNA and RNA synthesis. When substituted for the natural body proteins. substances, they disrupt DNA and RNA synthesis. bendamustine (nitrogen mustard) azacitidine (pyrimidine analogue) busulfan (alkyl sulfonate) capecitabine (pyrimidine analogue) carboplatin (platinum) cladribine (adenosine analogue) carmustine (nitrosurea) cytarabine (pyrimidine analogue) chlorambucil (nitrogen mustard) fludarabine (purine analogue) cisplatin (platinum) fluorouracil (pyrimidine analogue) cyclophosphamide (nitrogen mustard) gemcitabine (pyrimidine analogue) dacarbazine (triazine) mercaptopurine (purine analogue) estramustine (nitrogen mustard with 17-beta-estradiol) methotrexate (folate analogue) hydroxyurea pralatrexate (folate analogue) ifosfamide (nitrogen mustard) pemetrexed (folate analogue) lomustine (nitrosurea) pentostatin (purine analogue) mechlorethamine (nitrogen mustard) raltitrexed (folate analogue) melphalan (nitrogen mustard) thioguanine (purine analogue) oxaliplatin (platinum) trifluridine-tipiracil (pyrimidine analogue/thymidine phosphorylase procarbazine (triazine) inhibitor) -

Program Slides
Living with Hairy Cell Leukemia Michael Grever, MD Professor Emeritus, Internal Medicine/Hematology The Ohio State University Columbus, OH Living with Hairy Cell Leukemia Introduction to Hairy Cell Leukemia Michael Grever, MD Professor Emeritus, Internal Medicine/Hematology The Ohio State University Columbus, OH Financial Disclosures M. Grever served on a data safety monitoring board for Ascerta Inc., as a consultant for Pharmacyclics Inc., and as a consultant for AstraZeneca. First Descriptions of Hairy Cell Leukemia Gosselin GR, Hanlon DG, Pease GL. Leukaemic Reticuloendotheliosis. Can Med Assoc J. 74(11):886- 91, 1956 Bouroncle BA, Wiseman BC, Doan CA. Leukemic Reticuloendotheliosis. Blood 13: 609-630, 1958 Dr. Bertha Bouroncle Typical hairy cells among red blood cells (Dr. Gerard Lozanski, OSU) Clinical Features of Hairy Cell Leukemia • Remarkable male predominance 4:1 • Median age 55 years • Symptoms related to fatigue and infection • Physical exam shows enlarged spleen • Low blood cell counts (“cytopenias”) • Diagnosis made by examination of blood and bone marrow • Flow cytometry is critical for the diagnosis • Characteristic markers: CD11c, CD25; CD103; CD123 Manifestations of Hairy Cell Leukemia • Fatigue and symptoms of anemia • Easy bruising or bleeding • Infection • Herpes zoster (shingles) • Autoimmune disorders such as vasculitis, rheumatoid- like arthritis, immune thrombocytopenia • Bone lesions Clinical Judgement in Treatment Decisions • Determine if diagnosis is correct (e.g., classic HCL is different disease than HCL variant). • While 10% patients with HCL do not require immediate treatment, they require close follow-up. • Patients with active infection should not receive cladribine, and require special treatment planning • Need to assess kidney function and history of hepatitis exposure before treatment • Bone marrow biopsies at initiation and following completion of therapy have value. -

Association of Oral Anticoagulants and Proton Pump Inhibitor Cotherapy with Hospitalization for Upper Gastrointestinal Tract Bleeding
Supplementary Online Content Ray WA, Chung CP, Murray KT, et al. Association of oral anticoagulants and proton pump inhibitor cotherapy with hospitalization for upper gastrointestinal tract bleeding. JAMA. doi:10.1001/jama.2018.17242 eAppendix. PPI Co-therapy and Anticoagulant-Related UGI Bleeds This supplementary material has been provided by the authors to give readers additional information about their work. Downloaded From: https://jamanetwork.com/ on 10/02/2021 Appendix: PPI Co-therapy and Anticoagulant-Related UGI Bleeds Table 1A Exclusions: end-stage renal disease Diagnosis or procedure code for dialysis or end-stage renal disease outside of the hospital 28521 – anemia in ckd 5855 – Stage V , ckd 5856 – end stage renal disease V451 – Renal dialysis status V560 – Extracorporeal dialysis V561 – fitting & adjustment of extracorporeal dialysis catheter 99673 – complications due to renal dialysis CPT-4 Procedure Codes 36825 arteriovenous fistula autogenous gr 36830 creation of arteriovenous fistula; 36831 thrombectomy, arteriovenous fistula without revision, autogenous or 36832 revision of an arteriovenous fistula, with or without thrombectomy, 36833 revision, arteriovenous fistula; with thrombectomy, autogenous or nonaut 36834 plastic repair of arteriovenous aneurysm (separate procedure) 36835 insertion of thomas shunt 36838 distal revascularization & interval ligation, upper extremity 36840 insertion mandril 36845 anastomosis mandril 36860 cannula declotting; 36861 cannula declotting; 36870 thrombectomy, percutaneous, arteriovenous -

2015 Antiemesis.Pdf
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Antiemesis Version 2.2015 NCCN.org Continue Version 2.2015, 09/22/15 © National Comprehensive Cancer Network, Inc. 2015, All rights reserved. The NCCN Guidelines® and this illustration may not be reproduced in any form without the express written permission of NCCN®. Printed by Alexandre Ferreira on 10/25/2015 6:12:07 AM. For personal use only. Not approved for distribution. Copyright © 2015 National Comprehensive Cancer Network, Inc., All Rights Reserved. NCCN Guidelines Version 2.2015 Panel Members NCCN Guidelines Index Antiemesis Table of Contents Antiemesis Discussion David S. Ettinger, MD/Chair † Steve Kirkegaard, PharmD Σ Eric Roeland, MD The Sidney Kimmel Comprehensive Huntsman Cancer Institute UC San Diego Moores Cancer Cancer Center at Johns Hopkins at the University of Utah Center Michael J. Berger, PharmD/Vice Chair, BCOP Σ Dwight D. Kloth, PharmD, BCOP Σ Hope S. Rugo, MD † ‡ The Ohio State University Comprehensive Fox Chase Cancer Center UCSF Helen Diller Family Cancer Center - James Cancer Hospital Comprehensive Cancer Center and Solove Research Institute Ruth Lagman, MD £ Mayo Clinic Cancer Center Bridget Scullion, PharmD, BCOP Jonathan Aston, PharmD, BCOP, BCPS Σ Dana-Farber/Brigham and Women’s Vanderbilt-Ingram Cancer Center Dean Lim, MD † Cancer Center | Massachusetts City of Hope Comprehensive Cancer Center General Hospital Cancer Center Sally Barbour, PharmD, BCOP, CCP Σ Duke Cancer Institute Cynthia Ma, MD, PhD † John Timoney, PharmD, BCOP † Siteman Cancer Center at Barnes-Jewish Memorial Sloan Kettering Cancer Philip J. Bierman, MD † ‡ Hospital and Washington University School Center Fred & Pamela Buffet Cancer Center of Medicine Barbara Todaro, PharmD Σ Debra Brandt, DO Belinda Mandrell, PhD, RN † Roswell Park Cancer Institute Yale Cancer Center/Smilow Cancer Hospital St. -

Systemic Therapy Update
Systemic Therapy Update Volume 23 Issue 11 November 2020 For Health Professionals Who Care for Cancer Patients Inside This Issue: Editor’s Choice New Programs: Olaparib Maintenance Therapy in Newly Reformatted PPPOs: BRAJACTG, BRAJACTTG, BRAJACTW, Diagnosed Ovarian Cancer (UGOOVFOLAM) BRAJDCARBT, BRAJFECD, BRAJFECDT, BRAJTTW, Provincial Systemic Therapy Program BRAJZOL2, BRAVGEMP, BRAVPTRAT, BRLATACG, Interim Processes for Managing COVID-19 Pandemic | BRLATWAC, CNAJTZRT, CNBEV, CNCAB, CNTEMOZ, Revised Policy: Parenteral Drug Therapy [III-90] CNTEMOZMD, GIAJFFOX, GIAVCAP, GIAVPG, GICIRB, Drug Shortages GIENACTRT, GIFFOXB, GIFIRINOX, GIFOLFIRI, GIGAVCC, New: Bleomycin, Leuprolide | Resolved: Anagrelide, GIGAVFFOX, GIGFLODOC, GIGFOLFIRI, GIPAJFIROX, GIPE, GIPGEM, GIPGEMABR, GIRAJFFOX, GIRCRT, GOCXCATB, Bromocriptine © GOENDAI, GOENDCAT, GOOVBEVG, GOOVCARB, Cancer Drug Manual GOOVCATM, GOOVCATX, GOOVDDCAT, GOOVGEM, New: Gilteritinib, Pentostatin Revised: Arsenic Trioxide, GOOVPLDC, GUAJPG, GUAVPG, GUBEP, GUNAJPG, Doxorubicin Pegylated Liposomal, Idelalisib UGUPABI, GUSCPE, GUSUNI, HNNAVPG, LKCMLI, Benefit Drug List ULKMFRUX, LUAJNP, LUAVPG, LULACATRT, LULAPERT, New: UGOOVFOLAM LUSCPE, LUSCPERT, LUSCPI, LYABVD, LYBENDR, LYCHOP, New Protocols, PPPOs and Patient Handouts LYCHOPR, LYCLLFBR, ULYFIBRU, LYGDP, LYGDPR, LYIBRU, GO: UGOOVFOLAM MYBORPRE, MYBORREL, UMYCARDEX, UMYCARLD, Revised Protocols, PPPOs and Patient Handouts UMYDARBD, UMYDARLD, UMYLENMTN, MYMPBOR, GI: GIAVCRT, GIAVPANI, GIFIRINOX, GIGAVCC, GIIR, MYPAM, MYZOL, SAAVGI, SAIME, SMAVDT, -

Standard Oncology Criteria C16154-A
Prior Authorization Criteria Standard Oncology Criteria Policy Number: C16154-A CRITERIA EFFECTIVE DATES: ORIGINAL EFFECTIVE DATE LAST REVIEWED DATE NEXT REVIEW DATE DUE BEFORE 03/2016 12/2/2020 1/26/2022 HCPCS CODING TYPE OF CRITERIA LAST P&T APPROVAL/VERSION N/A RxPA Q1 2021 20210127C16154-A PRODUCTS AFFECTED: See dosage forms DRUG CLASS: Antineoplastic ROUTE OF ADMINISTRATION: Variable per drug PLACE OF SERVICE: Retail Pharmacy, Specialty Pharmacy, Buy and Bill- please refer to specialty pharmacy list by drug AVAILABLE DOSAGE FORMS: Abraxane (paclitaxel protein-bound) Cabometyx (cabozantinib) Erwinaze (asparaginase) Actimmune (interferon gamma-1b) Calquence (acalbrutinib) Erwinia (chrysantemi) Adriamycin (doxorubicin) Campath (alemtuzumab) Ethyol (amifostine) Adrucil (fluorouracil) Camptosar (irinotecan) Etopophos (etoposide phosphate) Afinitor (everolimus) Caprelsa (vandetanib) Evomela (melphalan) Alecensa (alectinib) Casodex (bicalutamide) Fareston (toremifene) Alimta (pemetrexed disodium) Cerubidine (danorubicin) Farydak (panbinostat) Aliqopa (copanlisib) Clolar (clofarabine) Faslodex (fulvestrant) Alkeran (melphalan) Cometriq (cabozantinib) Femara (letrozole) Alunbrig (brigatinib) Copiktra (duvelisib) Firmagon (degarelix) Arimidex (anastrozole) Cosmegen (dactinomycin) Floxuridine Aromasin (exemestane) Cotellic (cobimetinib) Fludara (fludarbine) Arranon (nelarabine) Cyramza (ramucirumab) Folotyn (pralatrexate) Arzerra (ofatumumab) Cytosar-U (cytarabine) Fusilev (levoleucovorin) Asparlas (calaspargase pegol-mknl Cytoxan (cyclophosphamide) -
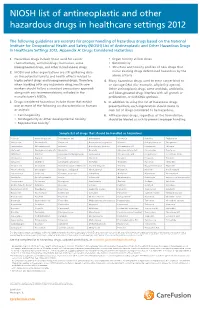
NIOSH List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings 2012
NIOSH list of antineoplastic and other hazardous drugs in healthcare settings 2012 The following guidelines are excerpts for proper handling of hazardous drugs based on the National Institute for Occupational Health and Safety (NIOSH) List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings 2012, Appendix A: Drugs Considered Hazardous 1. Hazardous drugs include those used for cancer • Organ toxicity at low doses* chemotherapy, antiviral drugs, hormones, some • Genotoxicity** bioengineered drugs, and other miscellaneous drugs. • Structure and toxicity profiles of new drugs that 2. NIOSH and other organizations are still gathering data mimic existing drugs determined hazardous by the on the potential toxicity and health effects related to above criteria highly potent drugs and bioengineered drugs. Therefore, 4. Many hazardous drugs used to treat cancer bind to when working with any hazardous drug, health care or damage DNA (for example, alkylating agents). workers should follow a standard precautions approach Other antineoplastic drugs, some antivirals, antibiotics, along with any recommendations included in the and bioengineered drugs interfere with cell growth or manufacturer’s MSDSs. proliferation, or with DNA synthesis. 3. Drugs considered hazardous include those that exhibit 5. In addition to using the list of hazardous drugs one or more of the following six characteristics in humans presented here, each organization should create its or animals: own list of drugs considered to be hazardous. • Carcinogenicity 6. All hazardous -

CYTOTOXIC and NON-CYTOTOXIC HAZARDOUS MEDICATIONS
CYTOTOXIC and NON-CYTOTOXIC HAZARDOUS MEDICATIONS1 CYTOTOXIC HAZARDOUS MEDICATIONS NON-CYTOTOXIC HAZARDOUS MEDICATIONS Altretamine IDArubicin Acitretin Iloprost Amsacrine Ifosfamide Aldesleukin Imatinib 3 Arsenic Irinotecan Alitretinoin Interferons Asparaginase Lenalidomide Anastrazole 3 ISOtretinoin azaCITIDine Lomustine Ambrisentan Leflunomide 3 azaTHIOprine 3 Mechlorethamine Bacillus Calmette Guerin 2 Letrozole 3 Bleomycin Melphalan (bladder instillation only) Leuprolide Bortezomib Mercaptopurine Bexarotene Megestrol 3 Busulfan 3 Methotrexate Bicalutamide 3 Methacholine Capecitabine 3 MitoMYcin Bosentan MethylTESTOSTERone CARBOplatin MitoXANtrone Buserelin Mifepristone Carmustine Nelarabine Cetrorelix Misoprostol Chlorambucil Oxaliplatin Choriogonadotropin alfa Mitotane CISplatin PACLitaxel Cidofovir Mycophenolate mofetil Cladribine Pegasparaginase ClomiPHENE Nafarelin Clofarabine PEMEtrexed Colchicine 3 Nilutamide 3 Cyclophosphamide Pentostatin cycloSPORINE Oxandrolone 3 Cytarabine Procarbazine3 Cyproterone Pentamidine (Aerosol only) Dacarbazine Raltitrexed Dienestrol Podofilox DACTINomycin SORAfenib Dinoprostone 3 Podophyllum resin DAUNOrubicin Streptozocin Dutasteride Raloxifene 3 Dexrazoxane SUNItinib Erlotinib 3 Ribavirin DOCEtaxel Temozolomide Everolimus Sirolimus DOXOrubicin Temsirolimus Exemestane 3 Tacrolimus Epirubicin Teniposide Finasteride 3 Tamoxifen 3 Estramustine Thalidomide Fluoxymesterone 3 Testosterone Etoposide Thioguanine Flutamide 3 Tretinoin Floxuridine Thiotepa Foscarnet Trifluridine Flucytosine Topotecan Fulvestrant -

Characterization of the Antiyeast Compound and Probiotic Properties
Electronic Journal of Biotechnology ISSN: 0717-3458 http://www.ejbiotechnology.info DOI: 10.2225/vol13-issue5-fulltext-7 Anticancer and apoptosis-inducing activities of microbial metabolites Sirinet Phonnok1 · Wanlaya Uthaisang Tanechpongtamb1 Benjamas Thanomsub Wongsatayanon1 1 Department of Microbiology, Faculty of Medicine, Srinakharinwirot University, Bangkok, Thailand 2 Department of Biochemistry, Faculty of Medicine, Srinakharinwirot University, Bangkok, Thailand 10110 Corresponding author: [email protected] Received March 2, 2010 / Accepted May 11, 2010 Published online: September 15, 2010 © 2010 by Pontificia Universidad Católica de Valparaíso, Chile Abstract The problems of systemic toxicity and drug resistance in cancer chemotherapy urge the continuing discovery of new anticancer agents. We explored the specific anticancer activity from microbial metabolites to find new lead compound. 394 microbial extracts were evaluated on anti-proliferative activity against 4 cancer cell lines using MTT assay. Of these, 20 samples showed varying degree of cytotoxicity but specifically to the cancer cell lines since the growth of normal cells was not significantly inhibited by 1 mg/ml of each cell extracts. The 4 most potent extracts exhibited strongest growth inhibition to each cancer cell type were selected for further studied. Cell morphological changes such as cell shrinkage, lose of surface contact and blebbing were observed in all treated cancer cells. DNA-binding dye staining demonstrated nuclear condensation and fragmentation. Chromosomal DNA cleavage detected as DNA ladder pattern by gel electrophoresis including activation of cellular caspase-3 activity, a hallmark of apoptosis, were observed in all treated cancer cell lines. These characteristics suggested the mechanism of apoptosis cell death induced by the extracts. -

The Cost Burden of Blood Cancer Care a Longitudinal Analysis of Commercially Insured Patients Diagnosed with Blood Cancer
MILLIMAN RESEARCH REPORT The cost burden of blood cancer care A longitudinal analysis of commercially insured patients diagnosed with blood cancer October 2018 Gabriela Dieguez, FSA, MAAA Christine Ferro, CHFP David Rotter, PhD Commissioned by The Leukemia & Lymphoma Society Table of Contents EXECUTIVE SUMMARY ............................................................................................................................................... 2 BACKGROUND ............................................................................................................................................................. 1 FINDINGS ...................................................................................................................................................................... 2 PREVALENCE AND COST OF BLOOD CANCER BY AGE GROUP ....................................................................... 2 INCIDENCE OF BLOOD CANCER ........................................................................................................................... 4 BLOOD CANCER CARE SPENDING FOLLOWING INITIAL DIAGNOSIS ............................................................... 5 PATIENT OUT-OF-POCKET COSTS FOLLOWING A BLOOD CANCER DIAGNOSIS ........................................... 9 The impact of insurance plan design on patient OOP costs .......................................................................... 10 CONSIDERATIONS FOR PAYERS ........................................................................................................................... -

Cancer Drug Costs for a Month of Treatment at Initial Food
Cancer drug costs for a month of treatment at initial Food and Drug Administration approval Year of FDA Monthly Cost Monthly cost (2013 Generic name Brand name(s) approval (actual $'s) $'s) Vinblastine Velban 1965 $78 $575 Thioguanine, 6-TG Thioguanine Tabloid 1966 $17 $122 Hydroxyurea Hydrea 1967 $14 $97 Cytarabine Cytosar-U, Tarabine PFS 1969 $13 $82 Procarbazine Matulane 1969 $2 $13 Testolactone Teslac 1969 $179 $1,136 Mitotane Lysodren 1970 $134 $801 Plicamycin Mithracin 1970 $50 $299 Mitomycin C Mutamycin 1974 $5 $22 Dacarbazine DTIC-Dome 1975 $29 $125 Lomustine CeeNU 1976 $10 $41 Carmustine BiCNU, BCNU 1977 $33 $127 Tamoxifen citrate Nolvadex 1977 $44 $167 Cisplatin Platinol 1978 $125 $445 Estramustine Emcyt 1981 $420 $1,074 Streptozocin Zanosar 1982 $61 $147 Etoposide, VP-16 Vepesid 1983 $181 $422 Interferon alfa 2a Roferon A 1986 $742 $1,573 Daunorubicin, Daunomycin Cerubidine 1987 $533 $1,090 Doxorubicin Adriamycin 1987 $521 $1,066 Mitoxantrone Novantrone 1987 $477 $976 Ifosfamide IFEX 1988 $1,667 $3,274 Flutamide Eulexin 1989 $213 $399 Altretamine Hexalen 1990 $341 $606 Idarubicin Idamycin 1990 $227 $404 Levamisole Ergamisol 1990 $105 $187 Carboplatin Paraplatin 1991 $860 $1,467 Fludarabine phosphate Fludara 1991 $662 $1,129 Pamidronate Aredia 1991 $507 $865 Pentostatin Nipent 1991 $1,767 $3,015 Aldesleukin Proleukin 1992 $13,503 $22,364 Melphalan Alkeran 1992 $35 $58 Cladribine Leustatin, 2-CdA 1993 $764 $1,229 Asparaginase Elspar 1994 $694 $1,088 Paclitaxel Taxol 1994 $2,614 $4,099 Pegaspargase Oncaspar 1994 $3,006 $4,713