S1 Supplement S1 Mathematical Theory Motivating This Work We Have
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Solutions for Practice Problems for Molecular Biology, Session 5
Solutions to Practice Problems for Molecular Biology, Session 5: Gene Regulation and the Lac Operon Question 1 a) How does lactose (allolactose) promote transcription of LacZ? 1) Lactose binds to the polymerase and increases efficiency. 2) Lactose binds to a repressor protein, and alters its conformation to prevent it from binding to the DNA and interfering with the binding of RNA polymerase. 3) Lactose binds to an activator protein, which can then help the RNA polymerase bind to the promoter and begin transcription. 4) Lactose prevents premature termination of transcription by directly binding to and bending the DNA. Solution: 2) Lactose binds to a repressor protein, and alters its conformation to prevent it from binding to the DNA and interfering with the binding of RNA polymerase. b) What molecule is used to signal low glucose levels to the Lac operon regulatory system? 1) Cyclic AMP 2) Calcium 3) Lactose 4) Pyruvate Solution: 1) Cyclic AMP. Question 2 You design a summer class where you recreate experiments studying the lac operon in E. coli (see schematic below). In your experiments, the activity of the enzyme b-galactosidase (β -gal) is measured by including X-gal and IPTG in the growth media. X-gal is a lactose analog that turns blue when metabolisize by b-gal, but it does not induce the lac operon. IPTG is an inducer of the lac operon but is not metabolized by b-gal. I O lacZ Plac Binding site for CAP Pi Gene encoding β-gal Promoter for activator protein Repressor (I) a) Which of the following would you expect to bind to β-galactosidase? Circle all that apply. -

The Lactose Operon from Lactobacillus Casei Is Involved in the Transport
www.nature.com/scientificreports OPEN The lactose operon from Lactobacillus casei is involved in the transport and metabolism of the Received: 4 October 2017 Accepted: 26 April 2018 human milk oligosaccharide core-2 Published: xx xx xxxx N-acetyllactosamine Gonzalo N. Bidart1, Jesús Rodríguez-Díaz 2, Gaspar Pérez-Martínez1 & María J. Yebra1 The lactose operon (lacTEGF) from Lactobacillus casei strain BL23 has been previously studied. The lacT gene codes for a transcriptional antiterminator, lacE and lacF for the lactose-specifc phosphoenolpyruvate: phosphotransferase system (PTSLac) EIICB and EIIA domains, respectively, and lacG for the phospho-β-galactosidase. In this work, we have shown that L. casei is able to metabolize N-acetyllactosamine (LacNAc), a disaccharide present at human milk and intestinal mucosa. The mutant strains BL153 (lacE) and BL155 (lacF) were defective in LacNAc utilization, indicating that the EIICB and EIIA of the PTSLac are involved in the uptake of LacNAc in addition to lactose. Inactivation of lacG abolishes the growth of L. casei in both disaccharides and analysis of LacG activity showed a high selectivity toward phosphorylated compounds, suggesting that LacG is necessary for the hydrolysis of the intracellular phosphorylated lactose and LacNAc. L. casei (lacAB) strain defcient in galactose-6P isomerase showed a growth rate in lactose (0.0293 ± 0.0014 h−1) and in LacNAc (0.0307 ± 0.0009 h−1) signifcantly lower than the wild-type (0.1010 ± 0.0006 h−1 and 0.0522 ± 0.0005 h−1, respectively), indicating that their galactose moiety is catabolized through the tagatose-6P pathway. Transcriptional analysis showed induction levels of the lac genes ranged from 130 to 320–fold in LacNAc and from 100 to 200–fold in lactose, compared to cells growing in glucose. -

Deconstructing Glucose-Mediated Catabolite Repression of the Lac Operon Of
bioRxiv preprint doi: https://doi.org/10.1101/2020.06.23.166959; this version posted June 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Deconstructing glucose-mediated catabolite repression of the lac operon of Escherichia coli: II. Positive feedback exists and drives the repression. Ritesh K. Aggarwala,b and Atul Naranga# aDepartment of Biochemical Engineering and Biotechnology, Indian Institute of Technology, Hauz Khas, New Delhi 110016, India bCancer Center, Albert Einstein College of Medicine, 1300 Morris Park Avenue, Bronx, New York 10461, USA Running Title: Positive feedback exists and drives lac repression #Email: [email protected] ORCID https://orcid.org/0000-0002-5693-1758 (RKA); https://orcid.org/0000-0002-6190- 8894 (AN) 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.06.23.166959; this version posted June 24, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Abstract The expression of the lac operon of E. coli is subject to positive feedback during growth in the presence of gratuitous inducers, but its existence in the presence of lactose remains controversial. The key question in this debate is: Do the lactose enzymes, Lac permease and β-galactosidase, promote accumulation of allolactose? If so, positive feedback exists since allolactose does stimulate synthesis of the lactose enzymes. -

Allostery and Applications of the Lac Repressor
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 2014 Allostery and Applications of the Lac Repressor Matthew Almond Sochor University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Biochemistry Commons, and the Biophysics Commons Recommended Citation Sochor, Matthew Almond, "Allostery and Applications of the Lac Repressor" (2014). Publicly Accessible Penn Dissertations. 1448. https://repository.upenn.edu/edissertations/1448 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/1448 For more information, please contact [email protected]. Allostery and Applications of the Lac Repressor Abstract The lac repressor has been extensively studied for nearly half a century; this long and complicated experimental history leaves many subtle connections unexplored. This thesis sought to forge those connections from isolated and purified components up ot functioning lac genetic switches in cells and even organisms. We first connected the genetics and structure of the lac repressor to in vivo gene regulation in Escherichia coli. We found that point mutations of amino acids that structurally make specific contacts with DNA can alter repressor-operator DNA affinity and even the conformational equilibrium of the repressor. We then found that point mutations of amino acids that structurally make specific contacts with effector molecules can alter repressor-effector affinity and the conformational equilibrium. Allesults r are well explained by a Monod, Wyman, and Changeux model of allostery. We next connected purified in vitro components with in vivo gene regulation in E. coli. We used an in vitro transcription assay to measure repressor-operator DNA binding affinity, repressor-effector binding affinity, and conformational equilibrium. -

A Primer on Gene Regulation
12 A Primer on Gene Regulation To understand the principles Genes are the units of nucleotide sequence in DNA that specify a protein or Goal of gene regulation. non-coding RNA. The full complement of genes in the genome ofEscherichia coli is about 5,000, and that in the human genome is about 20,000. These genes Objectives are expressed via their transcription into RNA and subsequent (in the case After this chapter, you should be able to of protein-coding genes) translation into protein. Importantly, not all genes are expressed at the same time. In bacteria, some genes are expressed at a • distinguish between negative and constant rate, but others are turned ON (transcribed) or OFF in response to positive control. cues from the environment. In multicellular organisms, such as the embryo • calculate K for repressor binding to eq of an animal, genes are turned ON in a cell- or tissue-specific manner at the DNA. right time and in the right place in response to developmental cues. Gene • explain the lac operon AND gate. regulation is a vastly complicated and fascinating subject encompassing an extraordinary range of molecular mechanisms. This chapter is intended as a primer for introducing the classic example of the lactose operon in bacteria and the concepts of negative and positive control. Genes involved in lactose metabolism are grouped in a single transcription unit, the lactose operon The subject of gene regulation derives from the seminal discoveries on the lactose (lac) operon of E. coli made by François Jacob and Jacque Monod while they were working at the Institut Pasteur in Paris and for which they shared the Nobel Prize in Physiology and Medicine in 1965. -

Impact of Glycerol As Carbon Source Onto Specific Sugar and Inducer
bioengineering Article Impact of Glycerol as Carbon Source onto Specific Sugar and Inducer Uptake Rates and Inclusion Body Productivity in E. coli BL21(DE3) Julian Kopp 1, Christoph Slouka 1, Sophia Ulonska 2, Julian Kager 2, Jens Fricke 1, Oliver Spadiut 2 and Christoph Herwig 1,2,* ID 1 Christian Doppler Laboratory for Mechanistic and Physiological Methods for Improved Bioprocesses, Institute of Chemical, Environmental and Biological Engineering, Vienna University of Technology, 1060 Vienna, Austria; [email protected] (J.K.); [email protected] (C.S.); [email protected] (J.F.) 2 Research Division Biochemical Engineering, Institute of Chemical, Environmental and Biological Engineering, Vienna University of Technology, 1060 Vienna , Austria; [email protected] (S.U.); [email protected] (J.K.); [email protected] (O.S.) * Correspondence: [email protected]; Tel.: +43-(1)-58801-166084 Academic Editor: Mark Blenner Received: 24 October 2017; Accepted: 19 December 2017; Published: 21 December 2017 Abstract: The Gram-negative bacterium E. coli is the host of choice for a multitude of used recombinant proteins. Generally, cultivation is easy, media are cheap, and a high product titer can be obtained. However, harsh induction procedures using isopropyl β-D-1 thiogalactopyranoside as inducer are often referred to cause stress reactions, leading to a phenomenon known as “metabolic” or “product burden”. These high expressions of recombinant proteins mainly result in decreased growth rates and cell lysis at elevated induction times. Therefore, approaches tend to use “soft” or “tunable” induction with lactose and reduce the stress level of the production host. -
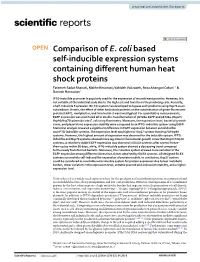
Comparison of E. Coli Based Self-Inducible Expression Systems
www.nature.com/scientificreports OPEN Comparison of E. coli based self‑inducible expression systems containing diferent human heat shock proteins Fatemeh Sadat Shariati, Malihe Keramati, Vahideh Valizadeh, Reza Ahangari Cohan* & Dariush Norouzian* IPTG‑inducible promoter is popularly used for the expression of recombinant proteins. However, it is not suitable at the industrial scale due to the high cost and toxicity on the producing cells. Recently, a Self‑Inducible Expression (SILEX) system has developed to bypass such problems using Hsp70 as an autoinducer. Herein, the efect of other heat shock proteins on the autoinduction of green fuorescent protein (EGFP), romiplostim, and interleukin‑2 was investigated. For quantitative measurements, EGFP expression was monitored after double‑transformation of pET28a‑EGFP and pET21a‑(Hsp27/ Hsp40/Hsp70) plasmids into E. coli using fuorimetry. Moreover, the expression level, bacterial growth curve, and plasmid and expression stability were compared to an IPTG‑ inducible system using EGFP. Statistical analysis revealed a signifcant diference in EGFP expression between autoinducible and IPTG‑inducible systems. The expression level was higher in Hsp27 system than Hsp70/Hsp40 systems. However, the highest amount of expression was observed for the inducible system. IPTG‑ inducible and Hsp70 systems showed more lag‑time in the bacterial growth curve than Hsp27/Hsp40 systems. A relatively stable EGFP expression was observed in SILEX systems after several freeze– thaw cycles within 90 days, while, IPTG‑inducible system showed a decreasing trend compared to the newly transformed bacteria. Moreover, the inducible system showed more variation in the EGFP expression among diferent clones than clones obtained by SILEX systems. All designed SILEX systems successfully self‑induced the expression of protein models. -

An-72632-Ic-Hpae-Pad-Lactose-Free
APPLICATION NOTE 72632 Fast and sensitive determination of lactose in lactose-free products using HPAE-PAD Authors Introduction Manali Aggrawal and Jeffrey Rohrer Lactose is the major disaccharide found in milk and is metabolized to glucose Thermo Fisher Scientific and galactose by the enzyme lactase. Lactose-intolerant individuals have a Sunnyvale, CA lactase deficiency; therefore, lactose is not completely metabolized. These individuals have difficulty digesting milk products, resulting in uncomfortable intestinal symptoms such as diarrhea and bloating. The demand for low- Keywords lactose dairy products is increasing and more lactose-free products are Dionex ICS-5000+, Dionex now available. These lactose-free products are produced by enzymatically CarboPac PA210 4µm column, breaking down lactose into glucose and galactose. However, the enzymatic dairy products, milk, baked goods, hydrolysis process is not 100% efficient and the resulting products contain cookies, chocolate, ICS-6000 varying amounts of residual lactose. Currently there are no legally defined lactose concentration limits or regulations governing lactose-free products either in USA or in EU legislation, except for infant formula in which lactose Goal should be 610 mg/100 kcal (Commission Directive, 2006).1 However, lactose To develop an HPAE-PAD method determinations are needed to meet ingredient labeling requirements. Some for the determination of allolactose, EU states have set threshold levels for the use of the terms ‘‘lactose-free”, lactose, and lactulose in lactose-free ‘‘very low lactose”, and ‘‘low lactose” for various foodstuffs. These threshold dairy products as well as lactose-free levels vary from 0.01 to 0.1 g/100 g of final product (EFSA, 2010).2 baked goods Milk products undergo heat treatment procedures columns are packed with a hydrophobic, polymeric, to eliminate microbes that can cause food spoilage. -
ABSTRACT AFROZ, TALIMAN. Understanding and Engineering
ABSTRACT AFROZ, TALIMAN. Understanding and Engineering Individuality in Bacterial Sugar Utilization. (Under the direction of Dr. Chase L. Beisel). Bacteria, like humans, can exhibit ‘individuality’ when faced with similar choices. The presence of positive and negative feedback in regulatory systems plays a crucial role in dictating ‘individuality’ in bacteria. Interestingly, positive and negative feedback loops are also present in sugar utilization pathways ubiquitous to virtually all microorganisms. The bacterial sugar utilization pathways encode regulators, transporters and catabolic enzymes that are induced by their cognate sugars. The regulators control the expression of the pathway components; transporters import more sugar conferring positive feedback and the enzymes break down the sugar imparting negative feedback. Because of scientific and biotechnological implications, sugar utilization pathways in the model bacterium Escherichia coli have been studied for many decades. However, most of the work done so far involved bulk characterization techniques that fail to capture unique behaviors observable only at the single-cell level. The very few single-cell studies available were focused on only two pathways. More importantly, the studies neglected sugar catabolism, which is an integral part of the natural sugar utilization pathways. Therefore, it remains unclear how the myriad of natural sugar utilization pathways respond to sugars at the single-cell level. This thesis work addresses this gap by investigating the single-cell response of E. coli to eight different sugars using a combination of experimental and computational approaches. The response of E. coli to sugars was remarkably diverse, with negative feedback due to catabolism playing a crucial role. Building on these insights, we engineered cells to convert sharp bimodal responses of natural sugar utilization pathways into linear, titratable responses. -

Determinants of Bistability in Induction of the Escherichia Coli Lac Operon
Los Alamos National Laboratory LA-UR-08-0753 Determinants of bistability in induction of the Escherichia coli lac operon David W. Dreisigmeyer,1 Jelena Stajic,2, ∗ Ilya Nemenman,3, 2 William S. Hlavacek,4, 2 and Michael E. Wall3, 5, 2, † 1High Performance Computing Division 2Center for Nonlinear Studies 3Computer, Computational, and Statistical Sciences Division 4Theoretical Division 5Bioscience Division, Los Alamos National Laboratory, Los Alamos, NM 87545, USA (Dated: October 23, 2018) Abstract We have developed a mathematical model of regulation of expression of the Escherichia coli lac operon, and have investigated bistability in its steady-state induction behavior in the absence of external glucose. Numerical analysis of equations describing regulation by artificial inducers revealed two natural bistability parameters that can be used to control the range of inducer concentrations over which the model exhibits bistability. By tuning these bistability parameters, we found a family of biophysically reasonable systems that are consistent with an experimentally determined bistable region for induction by thio-methylgalactoside (Ozbudak et al. Nature 427:737, 2004). The model predicts that bistability can be abolished when passive transport or permease export becomes sufficiently large; the former case is especially relevant to induction by isopropyl-β, D-thiogalactopyranoside. To model regulation by lactose, we developed similar equations arXiv:0802.1223v1 [q-bio.CB] 8 Feb 2008 in which allolactose, a metabolic intermediate in lactose metabolism and a natural inducer of lac, is the inducer. For biophysically reasonable parameter values, these equations yield no bistability in response to induction by lactose; however, systems with an unphysically small permease-dependent export effect can exhibit small amounts of bistability for limited ranges of parameter values. -

Origin of Bistability in the Lac Operon
3830 Biophysical Journal Volume 92 June 2007 3830–3842 Origin of Bistability in the lac Operon M. Santilla´n,*y M. C. Mackey,yz and E. S. Zeron§ *Unidad Monterrey, Centro de Investigacio´n y Estudios Avanzados del Instituto Polite´cnico Nacional, Monterrey, Me´xico; Escuela Superior de Fı´sica y Matema´ticas, Instituto Polite´cnico Nacional, Me´xico DF, Me´xico; yCentre for Nonlinear Dynamics in Physiology and Medicine, zDepartments of Physiology, Physics, and Mathematics, McGill University, Montreal, Canada; and §Departamento de Matema´ticas, Centro de Investigacio´n y Estudios Avanzados del Instituto Polite´cnico Nacional, Me´xico DF, Me´xico ABSTRACT Multistability is an emergent dynamic property that has been invoked to explain multiple coexisting biological states. In this work, we investigate the origin of bistability in the lac operon. To do this, we develop a mathematical model for the regulatory pathway in this system and compare the model predictions with other experimental results in which a nonmetabolizable inducer was employed. We investigate the effect of lactose metabolism using this model, and show that it greatly modifies the bistable region in the external lactose (Le) versus external glucose (Ge) parameter space. The model also predicts that lactose metabolism can cause bistability to disappear for very low Ge. We have also carried out stochastic numerical simulations of the model for several values of Ge and Le. Our results indicate that bistability can help guarantee that Escherichia coli consumes glucose and lactose in the most efficient possible way. Namely, the lac operon is induced only when there is almost no glucose in the growing medium, but if Le is high, the operon induction level increases abruptly when the levels of glucose in the environment decrease to very low values. -

Determination of Lactose in Low-Lactose Products
Determination of lactose in low-lactose products Manali Aggrawal and Jeff Rohrer, Thermo Fisher Scientific, Sunnyvale CA 94085, USA ABSTRACT RESULTS SAMPLE ANALYSIS RECOVERY CONCLUSIONS Purpose: To develop a High Performance Anion Exchange chromatography coupled with Pulsed Method accuracy was evaluated by measuring recoveries of sugar standards containing allolactose, Separation Figure 3 shows the separation of carbohydrates in lactose-free dairy samples S#1,S# 3, and S#5 . An HPAE-PAD method was successfully developed and validated for lactose analysis in 11 Amperometric Detection (HPAE-PAD) method for the determination of allolactose, lactose, and lactose, and lactulose spiked into lactose-free samples. Figure 5a, 5b shows the comparison of the along with a 1 ppm sugar standard mix. S#1 contains large amounts of glucose, galactose and small commercial lactose-free products including dairy as well as baked products. lactulose in lactose-free products. chromatogram of unspiked and spiked S#1 run under two eluent concentration; 14 mM and 23 mM To obtain a fast separation with baseline resolution of lactose from other structurally similar sugars, amounts of allolactose and lactose. S#3 is a 99% lactose-free low fat milk smoothie, it was diluted 40 Methods: We used the recently introduced Thermo Scientific™ Dionex™ CarboPac™ PA210-4μm KOH. As shown here, lactose is very well separated from similar disaccharides using both eluent the method was evaluated at different isocratic eluent conditions ranging from 12–25 mM KOH. times to bring the lactose concentration into the calibration range. S#5 (aged cheese) contains a very . The Dionex CarboPac PA210-4µm column allows the separation of lactose from structurally similar column to separate lactose from other structurally similar milk sugars.