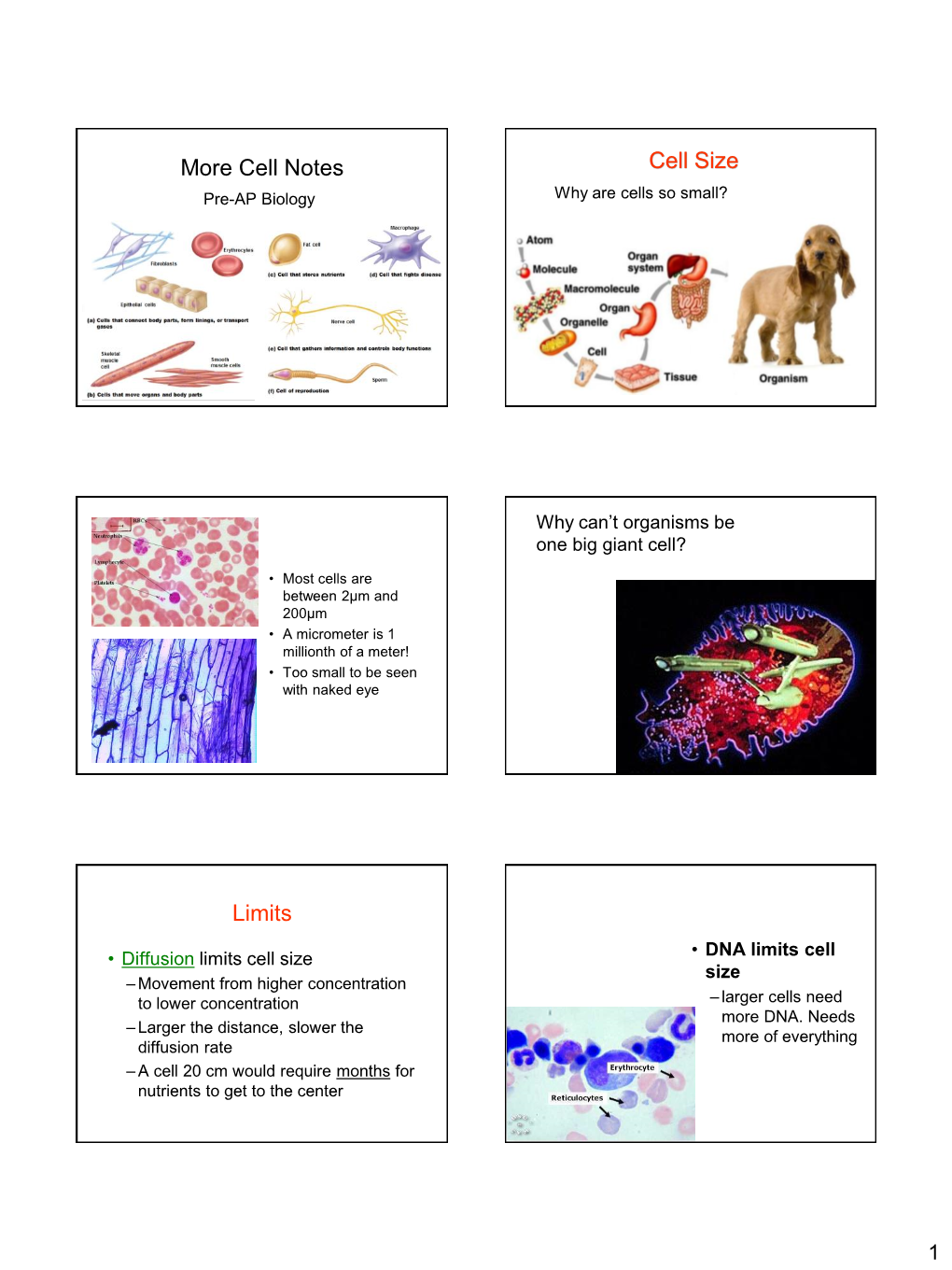
More Cell Notes Cell Size Limits
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Spring 2013 Lecture 23
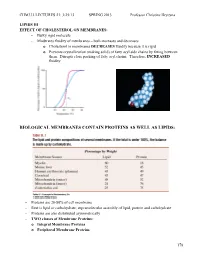
CHM333 LECTURES 23: 3/25/13 SPRING 2013 Professor Christine Hrycyna LIPIDS III EFFECT OF CHOLESTEROL ON MEMBRANES: - Bulky rigid molecule - Moderates fluidity of membranes – both increases and decreases o Cholesterol in membranes DECREASES fluidity because it is rigid o Prevents crystallization (making solid) of fatty acyl side chains by fitting between them. Disrupts close packing of fatty acyl chains. Therefore, INCREASED fluidity BIOLOGICAL MEMBRANES CONTAIN PROTEINS AS WELL AS LIPIDS: - Proteins are 20-80% of cell membrane - Rest is lipid or carbohydrate; supramolecular assembly of lipid, protein and carbohydrate - Proteins are also distributed asymmetrically - TWO classes of Membrane Proteins: o Integral Membrane Proteins o Peripheral Membrane Proteins 178 CHM333 LECTURES 23: 3/25/13 SPRING 2013 Professor Christine Hrycyna - INTEGRAL MEMBRANE PROTEINS o Located WITHIN the lipid bilayer o Usually span the bilayer one or more times – called transmembrane (TM) proteins o Hydrophobic amino acids interact with fatty acid chains in the hydrophobic core of the membrane o Can be removed from the membrane with detergents like SDS – need to disrupt the hydrophobic interactions § Membrane Disruption Animation: o http://www.youtube.com/watch?v=AHT37pvcjc0 o Function: § Transporters – moving molecules into or out of cells or cell membranes § Receptors – transmitting signals from outside of the cell to the inside - β Barrel Integral Membrane Proteins § Barrel-shaped membrane protein that is made up of antiparallel β-strands with hydrophilic (interior) and hydrophobic (facing lipid tails). § So far found only in outer membranes of Gram-negative bacteria, cell wall of Gram-positive bacteria, and outer membranes of mitochondria and chloroplasts. 179 CHM333 LECTURES 23: 3/25/13 SPRING 2013 Professor Christine Hrycyna - α-Helical Membrane Proteins - Can cross the membrane once or many times and have multiple transmembrane segments. -

Osmosis, Diffusion, and Membrane Transport Bio 219 Napa Valley College Dr
Osmosis, Diffusion, and Membrane Transport Bio 219 Napa Valley College Dr. Adam Ross Overview In order to understand how cells regulate themselves, we must first understand how things move into and out of cells Diffusion • Diffusion is the movement of particles from an area of high charge or concentration to an area of lower charge or concentration • Referred to as moving “down” a charge or concentration gradient • Ex. H+ ions in mitochondria moving through ATP synthase • Result of random molecular motion • Fick’s Law of Diffusion gives rate of diffusion: • Rate = P A (Cout – Cin) / (x) • Rate is proportional to permeability (P), surface area (A), concentration gradient (Cout – Cin); inversely proportional to diffusion distance or membrane thickness (x) Gradients • Concentration • Caused by unequal distribution of a substance on either side of the membrane • If the inside of a cell is negative, it will attract positively charged things • Electrical (charge) • Caused by unequal distribution of charge on either side of the membrane Diffusion Osmosis • Osmosis is the movement of solvent through a semi permeable membrane in order to balance the solute concentration on either side of the membrane. • In cells the solvent is water • Water can cross membranes Osmosis Osmolarity • Total concentration of all solutes in a solution • 1 Osm = 1 mole solute/ L • Have to account for both atoms in salts • 1M NaCl +1 L H2O → 1M Na+ + 1M Cl ≈ 2 Osm • Plasma = 290 mOsm Osmotic pressure • This is the actual driving force for net water movement • Depends on -

Pharmacokinetics, Pharmacodynamics and Drug
pharmaceutics Review Pharmacokinetics, Pharmacodynamics and Drug–Drug Interactions of New Anti-Migraine Drugs—Lasmiditan, Gepants, and Calcitonin-Gene-Related Peptide (CGRP) Receptor Monoclonal Antibodies Danuta Szkutnik-Fiedler Department of Clinical Pharmacy and Biopharmacy, Pozna´nUniversity of Medical Sciences, Sw.´ Marii Magdaleny 14 St., 61-861 Pozna´n,Poland; [email protected] Received: 28 October 2020; Accepted: 30 November 2020; Published: 3 December 2020 Abstract: In the last few years, there have been significant advances in migraine management and prevention. Lasmiditan, ubrogepant, rimegepant and monoclonal antibodies (erenumab, fremanezumab, galcanezumab, and eptinezumab) are new drugs that were launched on the US pharmaceutical market; some of them also in Europe. This publication reviews the available worldwide references on the safety of these anti-migraine drugs with a focus on the possible drug–drug (DDI) or drug–food interactions. As is known, bioavailability of a drug and, hence, its pharmacological efficacy depend on its pharmacokinetics and pharmacodynamics, which may be altered by drug interactions. This paper discusses the interactions of gepants and lasmiditan with, i.a., serotonergic drugs, CYP3A4 inhibitors, and inducers or breast cancer resistant protein (BCRP) and P-glycoprotein (P-gp) inhibitors. In the case of monoclonal antibodies, the issue of pharmacodynamic interactions related to the modulation of the immune system functions was addressed. It also focuses on the effect of monoclonal antibodies on expression of class Fc gamma receptors (FcγR). Keywords: migraine; lasmiditan; gepants; monoclonal antibodies; drug–drug interactions 1. Introduction Migraine is a chronic neurological disorder characterized by a repetitive, usually unilateral, pulsating headache with attacks typically lasting from 4 to 72 h. -

Membrane Transport, Absorption and Distribution of Drugs
Chapter 2 1 Pharmacokinetics: Membrane Transport, Absorption and Distribution of Drugs Pharmacokinetics is the quantitative study of drug movement in, through and out of the body. The overall scheme of pharmacokinetic processes is depicted in Fig. 2.1. The intensity of response is related to concentration of the drug at the site of action, which in turn is dependent on its pharmacokinetic properties. Pharmacokinetic considerations, therefore, determine the route(s) of administration, dose, and latency of onset, time of peak action, duration of action and frequency of administration of a drug. Fig. 2.1: Schematic depiction of pharmacokinetic processes All pharmacokinetic processes involve transport of the drug across biological membranes. Biological membrane This is a bilayer (about 100 Å thick) of phospholipid and cholesterol molecules, the polar groups (glyceryl phosphate attached to ethanolamine/choline or hydroxyl group of cholesterol) of these are oriented at the two surfaces and the nonpolar hydrocarbon chains are embedded in the matrix to form a continuous sheet. This imparts high electrical resistance and relative impermeability to the membrane. Extrinsic and intrinsic protein molecules are adsorbed on the lipid bilayer (Fig. 2.2). Glyco- proteins or glycolipids are formed on the surface by attachment to polymeric sugars, 2 aminosugars or sialic acids. The specific lipid and protein composition of different membranes differs according to the cell or the organelle type. The proteins are able to freely float through the membrane: associate and organize or vice versa. Some of the intrinsic ones, which extend through the full thickness of the membrane, surround fine aqueous pores. CHAPTER2 Fig. -

Cellular Transport Notes About Cell Membranes
Cellular Transport Notes @ 2011 Center for Pre-College Programs, New Jersey Institute of Technology, Newark, New Jersey About Cell Membranes • All cells have a cell membrane • Functions: – Controls what enters and exits the cell to maintain an internal balance called homeostasis TEM picture of a – Provides protection and real cell membrane. support for the cell @ 2011 Center for Pre-College Programs, New Jersey Institute of Technology, Newark, New Jersey 1 About Cell Membranes (continued) 1.Structure of cell membrane Lipid Bilayer -2 layers of phospholipids • Phosphate head is polar (water loving) Phospholipid • Fatty acid tails non-polar (water fearing) • Proteins embedded in membrane Lipid Bilayer @ 2011 Center for Pre-College Programs, New Jersey Institute of Technology, Newark, New Jersey Polar heads Fluid Mosaic love water Model of the & dissolve. cell membrane Non-polar tails hide from water. Carbohydrate cell markers Proteins @ 2011 Center for Pre-College Programs, New Jersey Institute of Technology, Newark, New Jersey 2 About Cell Membranes (continued) • 4. Cell membranes have pores (holes) in it • Selectively permeable: Allows some molecules in and keeps other molecules out • The structure helps it be selective! Pores @ 2011 Center for Pre-College Programs, New Jersey Institute of Technology, Newark, New Jersey Structure of the Cell Membrane Outside of cell Carbohydrate Proteins chains Lipid Bilayer Transport Protein Phospholipids Inside of cell (cytoplasm) @ 2011 Center for Pre-College Programs, New Jersey Institute of Technology, Newark, New Jersey 3 Types of Cellular Transport • Passive Transport celldoesn’tuseenergy 1. Diffusion 2. Facilitated Diffusion 3. Osmosis • Active Transport cell does use energy 1. -

An Introduction to the Viruses
Chapter 1 An introduction to the viruses Viruses are obligate intracellular parasites of man, other animals, plants and bacteria. They can only multiply within cells, and thus differ from bacteria, which can multiply in tissues in an extracellular position, and also in artificial culture media in the laboratory. It has always been recognized that viruses are distinct from the bacteria, but originally other groups of infective agents were included among the viruses which are now known to be organisms of a different and more complex nature. These included the Chlamydiae, organisms causing diseases such as lymphogranuloma venereum and trachoma, and the Rickettsiae, the causative agents of typhus and related infections. The specific treatment of infections caused by these two groups of agents will not be considered in this work, which is restricted to the chemotherapy of infections caused by the true viruses. The Chlamydiae and Rickettsiae are complete organisms, with cell walls, which possess both types of nucleic acid, enzyme systems which function within their cytoplasm, and a number of cell organelles. The viruses, on the other hand, possess either DNA or RNA as their genetic material, but not both. In some cases they contain enzymes, but these exert their functions within the host cell after the virus particle has under gone a process of dissolution during the early stage of infection. Except in the case of the arenaviruses, a group which contains the virus of Lassa fever, the virus particles do not contain organelles. A virus is thus much more rudimentary in structure, and relies upon the host cell to provide the systems for synthesizing its components which it does not possess itself. -

Phagocytosis References
9025 Technology Dr. Fishers, IN 46038 • www.bangslabs.com • [email protected] • 800.387.0672 PHAGOCYTOSIS REFERENCES GENERAL BEAD SELECTION & PHAGOCYTOSIS RATES Thiele L, Diederichs JE, Reszka R, Merkle HP, Walter E. (2003) Competitive adsorption of serum proteins at microparticles affects phagocytosis by dendritic cells. Biomaterials; 24(8):1409-18. (1µm Polybead® and Fluoresbrite® Carboxylate microspheres) Ahsan F, Rivas IP, Khan MA, Torres Suarez AI. (2002) Targeting to macrophages: role of physicochemical properties of particulate carriers-- liposomes and microspheres--on the phagocytosis by macrophages. J Controlled Release; 79:29-40. Thiele L, Rothen-Rutishauser B, Jilek S, Wunderli-Allenspach H, Merkle HP, Walter E. (2001) Evaluation of particle uptake in human blood monocyte-derived cells in vitro. Does phagocytosis activity of dendritic cells measure up with macrophages? J Controlled Release; 76:59-71. Koval M, Preiter K, Adles C, Stahl PD, Steinberg TH. (1998) Size of IgG-opsonized particles determines macrophage response during internalization. Exp Cell Res; 242(1):265-73. (0.2-0.3µm Polybead® microspheres; trypan blue quenching) Tabata Y, Ikada Y. (1988) Effect of the size and surface charge of polymer microspheres on their phagocytosis by macrophage. Biomaterials; 9(4):356-62. MONOCYTES Gu BJ, Duce JA, Valova VA, Wong B, Bush AI, Petrou S, Wiley JS. (2012) P2X7 receptor-mediated scavenger activity of mononuclear phagocytes toward non-opsonized particles and apoptotic cells is inhibited by serum glycoproteins but remains active in cerebrospinal fluid.Journal of Biological Chemistry. May 18;287:17318-30. (1µm Fluoresbrite® YG microspheres) Dumrese C, Slomianka L, Ziegler U, Choi SS, Kalia A, Fulurija A, Lu W, Berg DE, Benghezal M, Marshall B, Mittl PR. -

The Need for Mathematical Modelling of Spatial Drug Distribution Within the Brain Esmée Vendel1, Vivi Rottschäfer1 and Elizabeth C
Vendel et al. Fluids Barriers CNS (2019) 16:12 https://doi.org/10.1186/s12987-019-0133-x Fluids and Barriers of the CNS REVIEW Open Access The need for mathematical modelling of spatial drug distribution within the brain Esmée Vendel1, Vivi Rottschäfer1 and Elizabeth C. M. de Lange2* Abstract The blood brain barrier (BBB) is the main barrier that separates the blood from the brain. Because of the BBB, the drug concentration-time profle in the brain may be substantially diferent from that in the blood. Within the brain, the drug is subject to distributional and elimination processes: difusion, bulk fow of the brain extracellular fuid (ECF), extra-intracellular exchange, bulk fow of the cerebrospinal fuid (CSF), binding and metabolism. Drug efects are driven by the concentration of a drug at the site of its target and by drug-target interactions. Therefore, a quantita- tive understanding is needed of the distribution of a drug within the brain in order to predict its efect. Mathemati- cal models can help in the understanding of drug distribution within the brain. The aim of this review is to provide a comprehensive overview of system-specifc and drug-specifc properties that afect the local distribution of drugs in the brain and of currently existing mathematical models that describe local drug distribution within the brain. Furthermore, we provide an overview on which processes have been addressed in these models and which have not. Altogether, we conclude that there is a need for a more comprehensive and integrated model that flls the current gaps in predicting the local drug distribution within the brain. -

Identification of Caveolin and Caveolin-Related Proteins in the Brain
The Journal of Neuroscience, December 15, 1997, 17(24):9520–9535 Identification of Caveolin and Caveolin-Related Proteins in the Brain Patricia L. Cameron, Johnna W. Ruffin, Roni Bollag, Howard Rasmussen, and Richard S. Cameron Institute of Molecular Medicine and Genetics, Medical College of Georgia, Augusta, Georgia 30912-3175 Caveolae are 50–100 nm, nonclathrin-coated, flask-shaped brane. Immunoblot analyses demonstrate that detergent- plasma membrane microdomains that have been identified in insoluble complexes isolated from astrocytes are composed of most mammalian cell types, except lymphocytes and neurons. caveolin-1a, an identification verified by Northern blot analyses To date, multiple functions have been ascribed to caveolae, and by the cloning of a cDNA using reverse transcriptase-PCR including the compartmentalization of lipid and protein compo- amplification from total astrocyte RNA. Using a full-length nents that function in transmembrane signaling events, biosyn- caveolin-1 probe, Northern blot analyses suggest that the ex- thetic transport functions, endocytosis, potocytosis, and trans- pression of caveolin-1 may be regulated during brain develop- cytosis. Caveolin, a 21–24 kDa integral membrane protein, is ment. Immunoblot analyses of detergent-insoluble complexes the principal structural component of caveolae. We have initi- isolated from cerebral cortex and cerebellum identify two im- ated studies to examine the relationship of detergent-insoluble munoreactive polypeptides with apparent molecular weight and complexes identified -

The Membrane
The Membrane Natalie Gugala1*, Stephana J Cherak1 and Raymond J Turner1 1Department of Biological Sciences, University of Calgary, Canada *Corresponding author: RJ Turner, Department of Biological Sciences, University of Calgary, Alberta, Canada, Tel: 1-403-220-4308; Fax: 1-403-289-9311; Email: [email protected] Published Date: February 10, 2016 ABSTRACT and continues to be studied. The biological membrane is comprised of numerous amphiphilic The characterization of the cell membrane has significantly extended over the past century lipids, sterols, proteins, carbohydrates, ions and water molecules that result in two asymmetric polar leaflets, in which the interior is hydrophobic due to the hydrocarbon tails of the lipids. generated a dynamic heterogonous image of the membrane that includes lateral domains and The extension of the Fluid Mosaic Model, first proposed by Singer and Nicolson in 1972, has clusters perpetrated by lipid-lipid, protein-lipid and protein-protein interactions. Proteins found within the membrane, which are generally characterized as either intrinsic or extrinsic, have an array of biological functions vital for cell activity. The primary role of the membrane, among many, is to provide a barrier that conveys both separation and protection, thus maintaining the integrity of the cell. However, depending on the permeability of the membrane several ions are able to move down their concentration gradients. In turn this generates a membrane potential difference between the cytosol, which is found to have an excess negative charge, and surrounding extracellular fluid. Across a biological cell membrane, several potentials can be found. These include the Nernst or equilibrium potential, in which there is no overall flow of a Basicparticular Biochemistry ion and | www.austinpublishinggroup.com/ebooks the Donnan potential, created by an unequal distribution of ions. -

Internalization of Lectins in Neuronal Gerl
INTERNALIZATION OF LECTINS IN NEURONAL GERL NICHOLAS K. GONATAS, SEUNG U. KIM, ANNA STIEBER, and STRATIS AVRAMEAS From the Division of Neuropathology, Department of Pathology, University of PennsylvaniaSchool of Medicine, Philadelphia, Pennsylvania 19174, and the Laboratory of Immunocytochemistry,Pasteur Institute, Paris, France ABSTRACT Conjugates of ricin agglutinin and phytohemagglutinin with horseradish peroxi- dase (HRP) were used for a cytochemical study of internalization of their plasma membrane "receptors" in cultured isolated mouse dorsal root ganglion neurons. Labeling of cells with lectin-HRP was done at 4~ and internalization was performed at 37~ in a culture medium free of lectin-HRP. 15-30 rain after incubation at 37~ lectin-HRP-receptor complexes were seen in vesicles or tubules located near the plasma membrane. After 1-3 h at 37~ lectin-HRP- receptor complexes accumulated in vesicles and tubules corresponding to acid phosphatase-rich vesicles and tubules (GERL) at the trans aspect of the Golgi apparatus. A few coated vesicles and probably some dense bodies contained HRP after 3-6 h of incubation at 37~ Soluble HRP was not endocytosed under the conditions of this experiment or when it was present in the incubation medium at 37~ Internalization of lectin-HRP-receptor conjugates was decreased or in- hibited by mitochondrial respiration inhibitors but not by cytochalasin B or colchicine. These studies indicate that lectin-labeled plasma membrane moieties of neurons are endocytosed primarily in elements of GERL. Considerable information concerning the mobility tors" for ricin (Ric) and phytohemagglutinin and distribution of plasma membrane proteins has (PHA) labeled with horseradish peroxidase been gained with the use of various ligands. -

Passive and Active Transport
Passive and Active Transport 1. Thermodynamics of transport 2. Passive-mediated transport 3. Active transport neuron, membrane potential, ion transport Membranes • Provide barrier function – Extracellular – Organelles • Barrier can be overcome by „transport proteins“ – To mediate transmembrane movements of ions, Na+, K+ – Nutrients, glucose, amino acids etc. – Water (aquaporins) 1) Thermodynamics of Transport • Aout <-> Ain (ressembles a chemical equilibration) o‘ • GA - G A = RT ln [A] • ∆GA = GA(in) - GA(out) = RT ln ([A]in/[A]out) • GA: chemical potential of A o‘ • G A: chemical potential of standard state of A • If membrane has a potential, i.e., plasma membrane: -100mV (inside negative) then GA is termed the electrochemical potential of A Two types of transport across a membrane: o Nonmediated transport occurs by passive diffusion, i.e., O2, CO2 driven by chemical potential gradient, i.e. cannot occur against a concentration gradient o Mediated transport occurs by dedicated transport proteins 1. Passive-mediated transport/facilitated diffusion: [high] -> [low] 2. Active transport: [low] -> [high] May require energy in form of ATP or in form of a membrane potential 2) Passive-mediated transport Substances that are too large or too polar to diffuse across the bilayer must be transported by proteins: carriers, permeases, channels and transporters A) Ionophores B) Porins C) Ion Channels D) Aquaporins E) Transport Proteins A) Ionophores Organic molecules of divers types, often of bacterial origin => Increase the permeability of a target membrane for ions, frequently antibiotic, result in collapse of target membrane potential by ion equilibration 1. Carrier Ionophore, make ion soluble in membrane, i.e. valinomycin, 104 K+/sec 2.