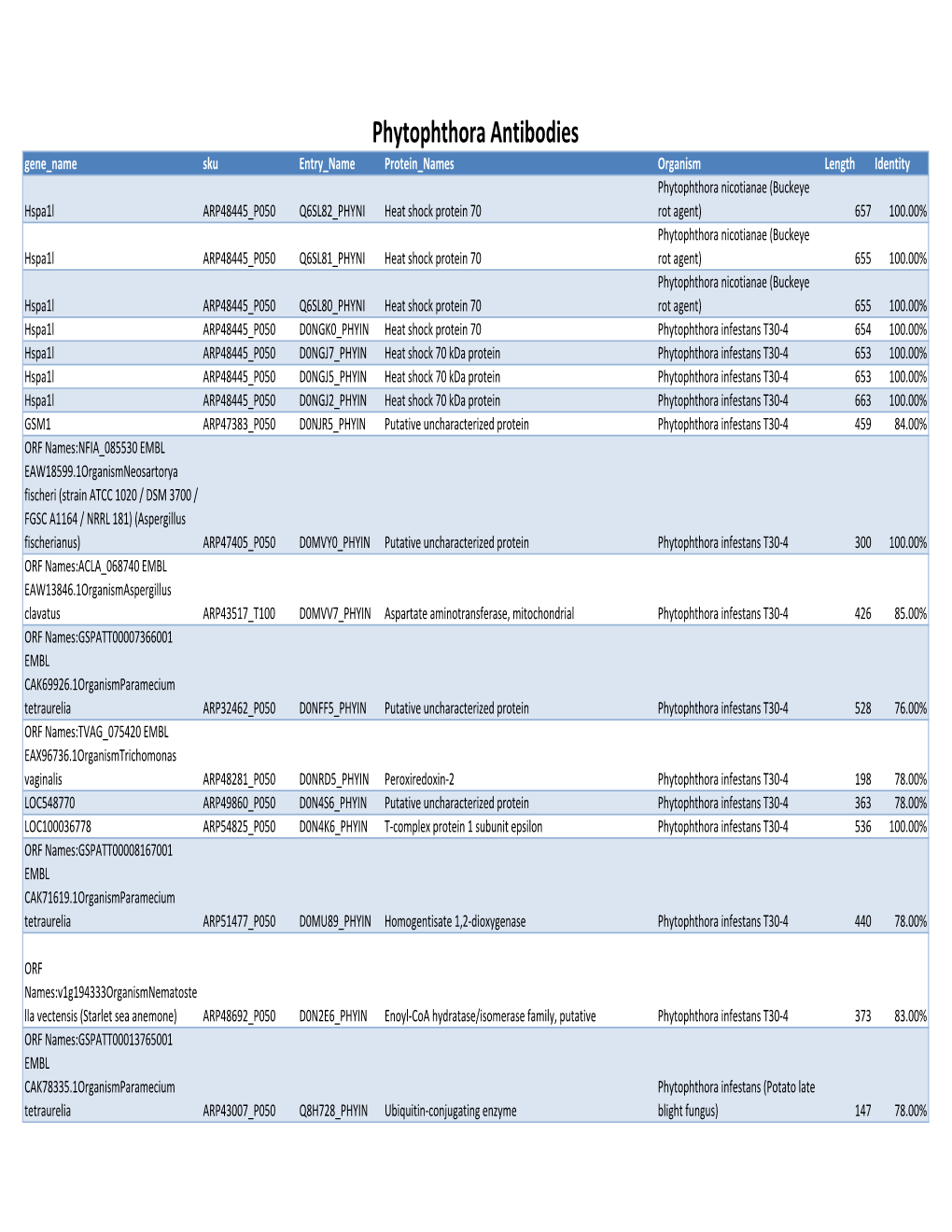
Phytophthora Antibodies
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Prokaryotic Ubiquitin-Like Protein Remains Intrinsically Disordered When Covalently Attached to Proteasomal Target Proteins Jonas Barandun1,2, Fred F
Barandun et al. BMC Structural Biology (2017) 17:1 DOI 10.1186/s12900-017-0072-1 RESEARCH ARTICLE Open Access Prokaryotic ubiquitin-like protein remains intrinsically disordered when covalently attached to proteasomal target proteins Jonas Barandun1,2, Fred F. Damberger1, Cyrille L. Delley1, Juerg Laederach1, Frédéric H. T. Allain1 and Eilika Weber-Ban1* Abstract Background: The post-translational modification pathway referred to as pupylation marks proteins for proteasomal degradation in Mycobacterium tuberculosis and other actinobacteria by covalently attaching the small protein Pup (prokaryotic ubiquitin-like protein) to target lysine residues. In contrast to the functionally analogous eukaryotic ubiquitin, Pup is intrinsically disordered in its free form. Its unfolded state allows Pup to adopt different structures upon interaction with different binding partners like the Pup ligase PafA and the proteasomal ATPase Mpa. While the disordered behavior of free Pup has been well characterized, it remained unknown whether Pup adopts a distinct structure when attached to a substrate. Results: Using a combination of NMR experiments and biochemical analysis we demonstrate that Pup remains unstructured when ligated to two well-established pupylation substrates targeted for proteasomal degradation in Mycobacterium tuberculosis, malonyl transacylase (FabD) and ketopantoyl hydroxylmethyltransferase (PanB). Isotopically labeled Pup was linked to FabD and PanB by in vitro pupylation to generate homogeneously pupylated substrates suitable for NMR analysis. The single target lysine of PanB was identified by a combination of mass spectroscopy and mutational analysis. Chemical shift comparison between Pup in its free form and ligated to substrate reveals intrinsic disorder of Pup in the conjugate. Conclusion: When linked to the proteasomal substrates FabD and PanB, Pup is unstructured and retains the ability to interact with its different binding partners. -

Yichen – Structure and Allostery of the Chaperonin Groel. Allosteric
Literature Lunch 5-1-13 Yichen – J Mol Biol. 2013 May 13;425(9):1476-87. doi: 10.1016/j.jmb.2012.11.028. Epub 2012 Nov 24. Structure and Allostery of the Chaperonin GroEL. Saibil HR, Fenton WA, Clare DK, Horwich AL. Source Crystallography and Institute of Structural and Molecular Biology, Birkbeck College London, Malet Street, London WC1E 7HX, UK. Abstract Chaperonins are intricate allosteric machines formed of two back-to-back, stacked rings of subunits presenting end cavities lined with hydrophobic binding sites for nonnative polypeptides. Once bound, substrates are subjected to forceful, concerted movements that result in their ejection from the binding surface and simultaneous encapsulation inside a hydrophilic chamber that favors their folding. Here, we review the allosteric machine movements that are choreographed by ATP binding, which triggers concerted tilting and twisting of subunit domains. These movements distort the ring of hydrophobic binding sites and split it apart, potentially unfolding the multiply bound substrate. Then, GroES binding is accompanied by a 100° twist of the binding domains that removes the hydrophobic sites from the cavity lining and forms the folding chamber. ATP hydrolysis is not needed for a single round of binding and encapsulation but is necessary to allow the next round of ATP binding in the opposite ring. It is this remote ATP binding that triggers dismantling of the folding chamber and release of the encapsulated substrate, whether folded or not. The basis for these ordered actions is an elegant system of nested cooperativity of the ATPase machinery. ATP binds to a ring with positive cooperativity, and movements of the interlinked subunit domains are concerted. -

SUMO and Ubiquitin in the Nucleus: Different Functions, Similar Mechanisms?
Downloaded from genesdev.cshlp.org on September 28, 2021 - Published by Cold Spring Harbor Laboratory Press REVIEW SUMO and ubiquitin in the nucleus: different functions, similar mechanisms? Grace Gill1 Department of Pathology, Harvard Medical School, Boston, Massachusetts 02115, USA The small ubiquitin-related modifier SUMO posttrans- tin, SUMO modification regulates protein localization lationally modifies many proteins with roles in diverse and activity. processes including regulation of transcription, chroma- This review focuses on recent advances in our under- tin structure, and DNA repair. Similar to nonproteolytic standing of SUMO function and regulation, drawing on a roles of ubiquitin, SUMO modification regulates protein limited set of examples relating to gene expression, chro- localization and activity. Some proteins can be modified matin structure, and DNA repair. Comparison of SUMO by SUMO and ubiquitin, but with distinct functional and ubiquitin activities in the nucleus reveals interest- consequences. It is possible that the effects of ubiquiti- ing differences in function and suggests surprising simi- nation and SUMOylation are both largely due to binding larities in mechanism. Thus, for example, modification of proteins bearing specific interaction domains. Both of transcription factors and histones by ubiquitin is gen- modifications are reversible, and in some cases dynamic erally associated with increased gene expression whereas cycles of modification may be required for activity. Stud- modification of transcription factors and histones by ies of SUMO and ubiquitin in the nucleus are yielding SUMO is generally associated with decreased gene ex- new insights into regulation of gene expression, genome pression. In some cases, SUMO and ubiquitin may di- maintenance, and signal transduction. -

Loss of Conserved Ubiquitylation Sites in Conserved Proteins During Human Evolution
INTERNATIONAL JOURNAL OF MOleCular meDICine 42: 2203-2212, 2018 Loss of conserved ubiquitylation sites in conserved proteins during human evolution DONGBIN PARK, CHUL JUN GOH, HYEIN KIM, JI SEOK LEE and YOONSOO HAHN Department of Life Science, Chung‑Ang University, Seoul 06974, Republic of Korea Received January 30, 2018; Accepted July 6, 2018 DOI: 10.3892/ijmm.2018.3772 Abstract. Ubiquitylation of lysine residues in proteins serves Introduction a pivotal role in the efficient removal of misfolded or unused proteins and in the control of various regulatory pathways Ubiquitylation, in which the highly conserved 76‑residue poly- by monitoring protein activity that may lead to protein peptide ubiquitin is covalently attached to a lysine residue of degradation. The loss of ubiquitylated lysines may affect substrate proteins, mediates the targeted destruction of ubiq- the ubiquitin‑mediated regulatory network and result in the uitylated proteins by the ubiquitin‑proteasome system (1‑4). emergence of novel phenotypes. The present study analyzed The ubiquitin‑mediated protein degradation pathway serves a mouse ubiquitylation data and orthologous proteins from crucial role in the efficient and specific removal of misfolded 62 mammals to identify 193 conserved ubiquitylation sites from proteins and certain key regulatory proteins (5,6). Ubiquitin 169 proteins that were lost in the Euarchonta lineage leading and other ubiquitin‑like proteins, including autophagy‑related to humans. A total of 8 proteins, including betaine homo- protein 8, Ubiquitin‑like -

Mdm2-Mediated Ubiquitylation: P53 and Beyond
Cell Death and Differentiation (2010) 17, 93–102 & 2010 Macmillan Publishers Limited All rights reserved 1350-9047/10 $32.00 www.nature.com/cdd Review Mdm2-mediated ubiquitylation: p53 and beyond J-C Marine*,1 and G Lozano2 The really interesting genes (RING)-finger-containing oncoprotein, Mdm2, is a promising drug target for cancer therapy. A key Mdm2 function is to promote ubiquitylation and proteasomal-dependent degradation of the tumor suppressor protein p53. Recent reports provide novel important insights into Mdm2-mediated regulation of p53 and how the physical and functional interactions between these two proteins are regulated. Moreover, a p53-independent role of Mdm2 has recently been confirmed by genetic data. These advances and their potential implications for the development of new cancer therapeutic strategies form the focus of this review. Cell Death and Differentiation (2010) 17, 93–102; doi:10.1038/cdd.2009.68; published online 5 June 2009 Mdm2 is a key regulator of a variety of fundamental cellular has also emerged from recent genetic studies. These processes and a very promising drug target for cancer advances and their potential implications for the development therapy. It belongs to a large family of (really interesting of new cancer therapeutic strategies form the focus of this gene) RING-finger-containing proteins and, as most of its review. For a more detailed discussion of Mdm2 and its other members, Mdm2 functions mainly, if not exclusively, as various functions an interested reader should also consult an E3 ligase.1 It targets various substrates for mono- and/or references9–12. poly-ubiquitylation thereby regulating their activities; for instance by controlling their localization, and/or levels by The p53–Mdm2 Regulatory Feedback Loop proteasome-dependent degradation. -

Ubiquitin Modifications
npg Cell Research (2016) 26:399-422. REVIEW www.nature.com/cr Ubiquitin modifications Kirby N Swatek1, David Komander1 1Medical Research Council Laboratory of Molecular Biology, Francis Crick Avenue, Cambridge, CB2 0QH, UK Protein ubiquitination is a dynamic multifaceted post-translational modification involved in nearly all aspects of eukaryotic biology. Once attached to a substrate, the 76-amino acid protein ubiquitin is subjected to further modi- fications, creating a multitude of distinct signals with distinct cellular outcomes, referred to as the ‘ubiquitin code’. Ubiquitin can be ubiquitinated on seven lysine (Lys) residues or on the N-terminus, leading to polyubiquitin chains that can encompass complex topologies. Alternatively or in addition, ubiquitin Lys residues can be modified by ubiq- uitin-like molecules (such as SUMO or NEDD8). Finally, ubiquitin can also be acetylated on Lys, or phosphorylated on Ser, Thr or Tyr residues, and each modification has the potential to dramatically alter the signaling outcome. While the number of distinctly modified ubiquitin species in cells is mind-boggling, much progress has been made to characterize the roles of distinct ubiquitin modifications, and many enzymes and receptors have been identified that create, recognize or remove these ubiquitin modifications. We here provide an overview of the various ubiquitin modifications present in cells, and highlight recent progress on ubiquitin chain biology. We then discuss the recent findings in the field of ubiquitin acetylation and phosphorylation, with a focus on Ser65-phosphorylation and its role in mitophagy and Parkin activation. Keywords: ubiquitin; proteasomal degradation; phosphorylation; post-translational modification; Parkin Cell Research (2016) 26:399-422. -

Protein Folding and Quality Control in the ER
Downloaded from http://cshperspectives.cshlp.org/ on September 25, 2021 - Published by Cold Spring Harbor Laboratory Press Protein Folding and Quality Control in the ER Kazutaka Araki and Kazuhiro Nagata Laboratory of Molecular and Cellular Biology, Faculty of Life Sciences, Kyoto Sangyo University, Kamigamo, Kita-ku, Kyoto 803-8555, Japan Correspondence: [email protected] The endoplasmic reticulum (ER) uses an elaborate surveillance system called the ER quality control (ERQC) system. The ERQC facilitates folding and modification of secretory and mem- brane proteins and eliminates terminally misfolded polypeptides through ER-associated degradation (ERAD) or autophagic degradation. This mechanism of ER protein surveillance is closely linked to redox and calcium homeostasis in the ER, whose balance is presumed to be regulated by a specific cellular compartment. The potential to modulate proteostasis and metabolism with chemical compounds or targeted siRNAs may offer an ideal option for the treatment of disease. he endoplasmic reticulum (ER) serves as a complex in the ER membrane (Johnson and Tprotein-folding factory where elaborate Van Waes 1999; Saraogi and Shan 2011). After quality and quantity control systems monitor arriving at the translocon, translation resumes an efficient and accurate production of secretory in a process called cotranslational translocation and membrane proteins, and constantly main- (Hegde and Kang 2008; Zimmermann et al. tain proper physiological homeostasis in the 2010). Numerous ER-resident chaperones and ER including redox state and calcium balance. enzymes aid in structural and conformational In this article, we present an overview the recent maturation necessary for proper protein fold- progress on the ER quality control system, ing, including signal-peptide cleavage, N-linked mainly focusing on the mammalian system. -

Shared and Unique Properties of Ubiquitin and SUMO Interaction Networks in DNA Repair
Downloaded from genesdev.cshlp.org on October 1, 2021 - Published by Cold Spring Harbor Laboratory Press PERSPECTIVE Shared and unique properties of ubiquitin and SUMO interaction networks in DNA repair Sjoerd J.L. van Wijk,1 Stefan Mu¨ ller,1 and Ivan Dikic1,2,3 1Institute of Biochemistry II, Goethe University School of Medicine, 60590 Frankfurt am Main, Germany; 2Frankfurt Institute for Molecular Life Sciences, Goethe University, 60438 Frankfurt am Main, Germany In this issue of Genes & Development, Yang and col- residues of target proteins and can be conjugated either as leagues (pp. 1847–1858) identify new components of a monomers or as polymeric chains that are generally small ubiquitin-like modifier (SUMO)-like interaction net- linked through internal lysine residues (Ikeda and Dikic work that orchestrates and fine-tunes the Fanconi anemia 2008). Conjugation of Ub and SUMO typically relies on (FA) pathway and replication-coupled repair. This new the coordinated activity of the catalytic E1–E2–E3 triad, pathway emphasizes the intricate interplay of ubiquitin but compared with Ub, the SUMO conjugation machinery (Ub) and SUMO networks in the DNA damage response. is less complex. SUMO becomes activated by the dimeric UBA2 (Ub-associated domain 2)/AOS1 complex and is subsequently transferred to Ubc9, the only known SUMO E2 (Kerscher et al. 2006; Gareau and Lima 2010). Although Ubiquitin (Ub) and the related small ubiquitin-like mod- Ubc9 is able to transfer SUMO directly to substrates, it ifier (SUMO) (hereafter commonly referred to as ubiqui- typically interacts with SUMO E3 ligases that mediate an tin-like proteins ½UBLs) are part of sophisticated and optimal positioning of the SUMO-loaded E2 and the sub- complex post-translational modification systems (Deribe strate to allow for efficient substrate SUMOylation (Gareau et al. -

Ubiquitin-Interacting Motifs of Ataxin-3 Regulate Its Polyglutamine Toxicity Through Hsc70-4-Dependent Aggregation
RESEARCH ARTICLE Ubiquitin-interacting motifs of ataxin-3 regulate its polyglutamine toxicity through Hsc70-4-dependent aggregation Sean L Johnson1, Bedri Ranxhi1, Kozeta Libohova1, Wei-Ling Tsou1*, Sokol V Todi1,2* 1Department of Pharmacology, Wayne State University, Detroit, United States; 2Department of Neurology, Wayne State University, Detroit, United States Abstract Spinocerebellar ataxia type 3 (SCA3) belongs to the family of polyglutamine neurodegenerations. Each disorder stems from the abnormal lengthening of a glutamine repeat in a different protein. Although caused by a similar mutation, polyglutamine disorders are distinct, implicating non-polyglutamine regions of disease proteins as regulators of pathogenesis. SCA3 is caused by polyglutamine expansion in ataxin-3. To determine the role of ataxin-3’s non- polyglutamine domains in disease, we utilized a new, allelic series of Drosophila melanogaster. We found that ataxin-3 pathogenicity is saliently controlled by polyglutamine-adjacent ubiquitin- interacting motifs (UIMs) that enhance aggregation and toxicity. UIMs function by interacting with the heat shock protein, Hsc70-4, whose reduction diminishes ataxin-3 toxicity in a UIM-dependent manner. Hsc70-4 also enhances pathogenicity of other polyglutamine proteins. Our studies provide a unique insight into the impact of ataxin-3 domains in SCA3, identify Hsc70-4 as a SCA3 enhancer, and indicate pleiotropic effects from HSP70 chaperones, which are generally thought to suppress polyglutamine degeneration. *For correspondence: [email protected] (W-LT); [email protected] (SVT) Introduction Competing interests: The Spinocerebellar ataxia type 3 (SCA3; also known as Machado-Joseph disease) is the most frequent authors declare that no dominant ataxia worldwide. SCA3 is caused by CAG repeat expansion in the gene ATXN3 that is competing interests exist. -

The Function of FK506-Binding Protein 13 in Protein Quality Control Protects Plasma Cells from Endoplasmic Reticulum Stress- Edited By: Associated Apoptosis Harry W
ORIGINAL RESEARCH published: 02 March 2017 doi: 10.3389/fimmu.2017.00222 The Function of FK506-Binding Protein 13 in Protein Quality Control Protects Plasma Cells from Endoplasmic Reticulum Stress- Edited by: Associated Apoptosis Harry W. Schroeder, University of Alabama at Birmingham, 1† 1 1 2 3 USA Mini Jeong , Eunkyeong Jang , Suk San Choi , Changhoon Ji , Kyungho Lee and Jeehee Youn1* Reviewed by: Michael A. Lynes, 1 Laboratory of Autoimmunology, Department of Anatomy and Cell Biology, College of Medicine, Hanyang University, Seoul, University of Connecticut, USA South Korea, 2 Protein Metabolism Medical Research Center, Department of Biomedical Sciences, College of Medicine, Thierry Defrance, Seoul National University, Seoul, South Korea, 3 Department of Biological Sciences, Konkuk University, Seoul, South Korea Institut national de la santé et de la recherche médicale (INSERM), France Plasma cells (PCs) are exposed to intense endoplasmic reticulum (ER) stress imposed *Correspondence: by enormous rates of immunoglobulin (Ig) synthesis and secretion. Therefore, protein Jeehee Youn [email protected] homeostasis is crucial for the survival of PCs, but its molecular mechanism remains largely unknown. Here, we found marked overexpression of FK506-binding protein 13 †Present address: Mini Jeong, (FKBP13) in long-lived PCs from autoimmune mice and investigated its function using Department of Biochemistry and a plasmacytoma cell line secreting IgA. FKBP13 expression was induced largely in the Molecular Biology, Korean Institute of Molecular Medicine and Nutrition, lumen of ER in response to treatment with an ER stressor tunicamycin or overexpression Korea University College of Medicine, of an adaptive unfolded protein response (UPR) protein X-box binding protein 1 (XBP1). -

Urmylation and Trna Thiolation Functions of Ubiquitinlike Uba4urm1
FEBS Letters 589 (2015) 904–909 journal homepage: www.FEBSLetters.org Urmylation and tRNA thiolation functions of ubiquitin-like Uba4ÁUrm1 systems are conserved from yeast to manq André Jüdes a, Folke Ebert a, Christian Bär a,b, Kathrin L. Thüring c, Aileen Harrer a, Roland Klassen a, ⇑ Mark Helm c, Michael J.R. Stark d, Raffael Schaffrath a, a Universität Kassel, Institut für Biologie, FG Mikrobiologie, Heinrich-Plett-Str. 40, 34132 Kassel, Germany b Spanish National Cancer Centre, Melchor Fernandez Almagro 3, Madrid, Spain c Johannes Gutenberg Universität Mainz, Institut für Pharmazie und Biochemie, Staudinger Weg 5, 55128 Mainz, Germany d Centre for Gene Regulation & Expression, College of Life Sciences, MSI/WTB Complex, University of Dundee, Dundee DD1 5EH, Scotland, UK article info abstract Article history: The ubiquitin-like protein Urm1 from budding yeast and its E1-like activator Uba4 have dual roles in Received 12 January 2015 protein urmylation and tRNA thiolation pathways. To study whether these are conserved among Revised 11 February 2015 eukaryotes, we used gene shuffles to replace the yeast proteins by their human counterparts, Accepted 24 February 2015 hURM1 and hUBA4/MOCS3. As judged from biochemical and genetical assays, hURM1 and hUBA4 Available online 3 March 2015 are functional in yeast, albeit at reduced efficiencies. They mediate urmylation of the peroxiredoxin Edited by Michael Ibba Ahp1, a known urmylation target in yeast, and support tRNA thiolation. Similar to hUBA4, yeast Uba4 itself is modified by Urm1 and hURM1 suggesting target overlap between eukaryal urmylation pathways. In sum, our study shows that dual-function ubiquitin-like Urm1 Uba4 systems are con- Keywords: Á Urm1 (hURM1) served and exchangeable between human and yeast cells. -

SUMO Playing Tag with Ubiquitin
TIBS-872; No. of Pages 9 Review SUMO playing tag with ubiquitin 1 2 3 Gerrit J.K. Praefcke , Kay Hofmann and R. Ju¨ rgen Dohmen 1 Center for Molecular Medicine Cologne (CMMC), Institute for Genetics, University of Cologne, Cologne Biocenter, Zu¨ lpicher Straße 47a, D-50674 Cologne, Germany 2 Bioinformatics Group, Miltenyi Biotec GmbH, Friedrich-Ebert-Straße 68, D-51429 Bergisch-Gladbach, Germany 3 Institute for Genetics, University of Cologne, Cologne Biocenter, Zu¨ lpicher Straße 47a, D-50674 Cologne, Germany In addition to being structurally related, the protein modi- Similarly, SUMO modification has been implicated in fiers ubiquitin and SUMO (small ubiquitin-related modifi- many important cellular processes including the control of er), share a multitude of functional interrelations. These genome stability, signal transduction, targeting to and include the targeting of the same attachment sites in formation of nuclear compartments, cell cycle and meiosis certain substrates, and SUMO-dependent ubiquitylation [3–5]. Whereas budding yeast (Saccharomyces cerevisiae) in others. Notably, several cellular processes, including and fission yeast (Schizosaccharomyces pombe) have only the targeting of repair machinery to DNA damage sites, one type of SUMO, multiple SUMO isoforms are present in require the sequential sumoylation and ubiquitylation of many metazoans. For example, four SUMO isoforms are distinct substrates. Some proteins promote both modifi- present in humans (Table 1). Only three of these (SUMO-1, cations. By contrast, the activity of some enzymes that SUMO-2 and SUMO-3) can be processed in vivo to bear the control either sumoylation or ubiquitylation is regulated C-terminal diglycine motif required for post-translational by the respective other modification.