Cnidaria, Octocorallia, Primnoidae)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
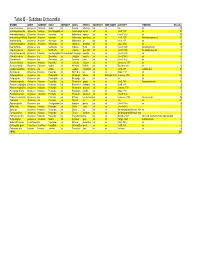
Table B – Subclass Octocorallia
Table B – Subclass Octocorallia BINOMEN ORDER SUBORDER FAMILY SUBFAMILY GENUS SPECIES SUBSPECIES COMN_NAMES AUTHORITY SYNONYMS #Records Acanella arbuscula Alcyonacea Calcaxonia Isididae n/a Acanella arbuscula n/a n/a n/a n/a 59 Acanthogorgia armata Alcyonacea Holaxonia Acanthogorgiidae n/a Acanthogorgia armata n/a n/a Verrill, 1878 n/a 95 Anthomastus agassizii Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus agassizii n/a n/a (Verrill, 1922) n/a 35 Anthomastus grandiflorus Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus grandiflorus n/a n/a Verrill, 1878 Anthomastus purpureus 37 Anthomastus sp. Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus sp. n/a n/a Verrill, 1878 n/a 1 Anthothela grandiflora Alcyonacea Scleraxonia Anthothelidae n/a Anthothela grandiflora n/a n/a (Sars, 1856) n/a 24 Capnella florida Alcyonacea n/a Nephtheidae n/a Capnella florida n/a n/a (Verrill, 1869) Eunephthya florida 44 Capnella glomerata Alcyonacea n/a Nephtheidae n/a Capnella glomerata n/a n/a (Verrill, 1869) Eunephthya glomerata 4 Chrysogorgia agassizii Alcyonacea Holaxonia Acanthogorgiidae Chrysogorgiidae Chrysogorgia agassizii n/a n/a (Verrill, 1883) n/a 2 Clavularia modesta Alcyonacea n/a Clavulariidae n/a Clavularia modesta n/a n/a (Verrill, 1987) n/a 6 Clavularia rudis Alcyonacea n/a Clavulariidae n/a Clavularia rudis n/a n/a (Verrill, 1922) n/a 1 Gersemia fruticosa Alcyonacea Alcyoniina Alcyoniidae n/a Gersemia fruticosa n/a n/a Marenzeller, 1877 n/a 3 Keratoisis flexibilis Alcyonacea Calcaxonia Isididae n/a Keratoisis flexibilis n/a n/a Pourtales, 1868 n/a 1 Lepidisis caryophyllia Alcyonacea n/a Isididae n/a Lepidisis caryophyllia n/a n/a Verrill, 1883 Lepidisis vitrea 13 Muriceides sp. -

Tree Coral, Primnoa Pacifica
Sexual Reproduction and Seasonality of the Alaskan Red Tree Coral, Primnoa pacifica Rhian G. Waller1*, Robert P. Stone2, Julia Johnstone3, Jennifer Mondragon4 1 University of Maine, School of Marine Sciences, Darling Marine Center, Walpole, Maine, United States of America, 2 Alaska Fisheries Science Center, National Marine Fisheries Service, National Oceanic and Atmospheric Administration, Juneau, Alaska, United States of America, 3 University of Maine, Darling Marine Center, Walpole, Maine, United States of America, 4 Alaska Regional Office, National Marine Fisheries Service, National Oceanic and Atmospheric Administration, Juneau, Alaska, United States of America Abstract The red tree coral Primnoa pacifica is an important habitat forming octocoral in North Pacific waters. Given the prominence of this species in shelf and upper slope areas of the Gulf of Alaska where fishing disturbance can be high, it may be able to sustain healthy populations through adaptive reproductive processes. This study was designed to test this hypothesis, examining reproductive mode, seasonality and fecundity in both undamaged and simulated damaged colonies over the course of 16 months using a deepwater-emerged population in Tracy Arm Fjord. Females within the population developed asynchronously, though males showed trends of synchronicity, with production of immature spermatocysts heightened in December/January and maturation of gametes in the fall months. Periodicity of individuals varied from a single year reproductive event to some individuals taking more than the 16 months sampled to produce viable gametes. Multiple stages of gametes occurred in polyps of the same colony during most sampling periods. Mean oocyte size ranged from 50 to 200 mm in any season, and maximum oocyte size (802 mm) suggests a lecithotrophic larva. -

Deep-Sea Origin and In-Situ Diversification of Chrysogorgiid Octocorals
Deep-Sea Origin and In-Situ Diversification of Chrysogorgiid Octocorals Eric Pante1*¤, Scott C. France1, Arnaud Couloux2, Corinne Cruaud2, Catherine S. McFadden3, Sarah Samadi4, Les Watling5,6 1 Department of Biology, University of Louisiana at Lafayette, Lafayette, Louisiana, United States of America, 2 GENOSCOPE, Centre National de Se´quenc¸age, Evry, France, 3 Department of Biology, Harvey Mudd College, Claremont, California, United States of America, 4 De´partement Syste´matique et Evolution, UMR 7138 UPMC-IRD-MNHN- CNRS (UR IRD 148), Muse´um national d’Histoire naturelle, Paris, France, 5 Department of Biology, University of Hawaii at Manoa, Honolulu, Hawaii, United States of America, 6 Darling Marine Center, University of Maine, Walpole, Maine, United States of America Abstract The diversity, ubiquity and prevalence in deep waters of the octocoral family Chrysogorgiidae Verrill, 1883 make it noteworthy as a model system to study radiation and diversification in the deep sea. Here we provide the first comprehensive phylogenetic analysis of the Chrysogorgiidae, and compare phylogeny and depth distribution. Phylogenetic relationships among 10 of 14 currently-described Chrysogorgiidae genera were inferred based on mitochondrial (mtMutS, cox1) and nuclear (18S) markers. Bathymetric distribution was estimated from multiple sources, including museum records, a literature review, and our own sampling records (985 stations, 2345 specimens). Genetic analyses suggest that the Chrysogorgiidae as currently described is a polyphyletic family. Shallow-water genera, and two of eight deep-water genera, appear more closely related to other octocoral families than to the remainder of the monophyletic, deep-water chrysogorgiid genera. Monophyletic chrysogorgiids are composed of strictly (Iridogorgia Verrill, 1883, Metallogorgia Versluys, 1902, Radicipes Stearns, 1883, Pseudochrysogorgia Pante & France, 2010) and predominantly (Chrysogorgia Duchassaing & Michelotti, 1864) deep-sea genera that diversified in situ. -

Guide to the Identification of Precious and Semi-Precious Corals in Commercial Trade
'l'llA FFIC YvALE ,.._,..---...- guide to the identification of precious and semi-precious corals in commercial trade Ernest W.T. Cooper, Susan J. Torntore, Angela S.M. Leung, Tanya Shadbolt and Carolyn Dawe September 2011 © 2011 World Wildlife Fund and TRAFFIC. All rights reserved. ISBN 978-0-9693730-3-2 Reproduction and distribution for resale by any means photographic or mechanical, including photocopying, recording, taping or information storage and retrieval systems of any parts of this book, illustrations or texts is prohibited without prior written consent from World Wildlife Fund (WWF). Reproduction for CITES enforcement or educational and other non-commercial purposes by CITES Authorities and the CITES Secretariat is authorized without prior written permission, provided the source is fully acknowledged. Any reproduction, in full or in part, of this publication must credit WWF and TRAFFIC North America. The views of the authors expressed in this publication do not necessarily reflect those of the TRAFFIC network, WWF, or the International Union for Conservation of Nature (IUCN). The designation of geographical entities in this publication and the presentation of the material do not imply the expression of any opinion whatsoever on the part of WWF, TRAFFIC, or IUCN concerning the legal status of any country, territory, or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. The TRAFFIC symbol copyright and Registered Trademark ownership are held by WWF. TRAFFIC is a joint program of WWF and IUCN. Suggested citation: Cooper, E.W.T., Torntore, S.J., Leung, A.S.M, Shadbolt, T. and Dawe, C. -

Anthozoa: Octocorallia) from the Clarion-Clipperton Fracture Zone, Equatorial Northeastern Pacific
Mar Biodiv DOI 10.1007/s12526-015-0340-x ORIGINAL PAPER New abyssal Primnoidae (Anthozoa: Octocorallia) from the Clarion-Clipperton Fracture Zone, equatorial northeastern Pacific Stephen D. Cairns1 Received: 28 January 2015 /Revised: 20 March 2015 /Accepted: 23 March 2015 # Senckenberg Gesellschaft für Naturforschung and Springer-Verlag Berlin Heidelberg 2015 Abstract Three new species, including a new genus, nodules that contain iron, manganese, copper, nickel, cobalt, Abyssoprimnoa, are described from abyssal depths from the zinc, silver, and other metals (Bluhm 1994), and thus have easternmost Clarion-Clipperton Fracture Zone in the equato- been of interest to deep-sea mining companies for several rial northeastern Pacific. This prompted the listing of all 39 decades since the region was discovered in the 1950s. octocorallian taxa collected deeper than 3000 m, which con- Currently, at least 12 claim sites have been awarded to various stitutes only about 1.2 % of the octocoral species. To place this mining companies, each associated with one or more coun- in perspective, the depth records for other benthic cnidarian tries. The hard substrate afforded by the nodules allows the orders are compared. settlement of various benthic invertebrates that require a hard surface for attachment and subsequent anchoring, one of these groups being the octocorals. Because of the extreme depth and Keywords Primnoidae . Clarion-clipperton fracture zone . logistic difficulty of collecting organisms from such depths, New species . New genus . Octocorallia -

Phylogenetic Relationships Within Chrysogorgia (Alcyonacea
Phylogenetic Relationships Within Chrysogorgia (Alcyonacea: Octocorallia), a Morphologically Diverse Genus of Octocoral, Revealed Using a Target Enrichment Approach Candice Untiedt, Andrea Quattrini, Catherine Mcfadden, Phil Alderslade, Eric Pante, Christopher Burridge To cite this version: Candice Untiedt, Andrea Quattrini, Catherine Mcfadden, Phil Alderslade, Eric Pante, et al.. Phy- logenetic Relationships Within Chrysogorgia (Alcyonacea: Octocorallia), a Morphologically Diverse Genus of Octocoral, Revealed Using a Target Enrichment Approach. Frontiers in Marine Science, Frontiers Media, 2021, 7, 10.3389/fmars.2020.599984. hal-03154113 HAL Id: hal-03154113 https://hal.sorbonne-universite.fr/hal-03154113 Submitted on 27 Feb 2021 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. fmars-07-599984 January 11, 2021 Time: 10:55 # 1 ORIGINAL RESEARCH published: 12 January 2021 doi: 10.3389/fmars.2020.599984 Phylogenetic Relationships Within Chrysogorgia (Alcyonacea: Octocorallia), a Morphologically Diverse Genus of Octocoral, Revealed Using a Target Enrichment -

Primnoidae (Octocorallia: Calcaxonia) from the Emperor Seamounts, with Notes on Callogorgia Elegans (Gray, 1870)
Primnoidae (Octocorallia: Calcaxonia) from the Emperor Seamounts, with Notes on Callogorgia elegans (Gray, 1870) Stephen D. Cairns, Robert P. Stone, Hye-Won Moon, Jong Hee Lee Pacific Science, Volume 72, Number 1, January 2018, pp. 125-142 (Article) Published by University of Hawai'i Press For additional information about this article https://muse.jhu.edu/article/683173 [ This content has been declared free to read by the pubisher during the COVID-19 pandemic. ] Primnoidae (Octocorallia: Calcaxonia) from the Emperor Seamounts, with Notes on Callogorgia elegans (Gray, 1870)1 Stephen D. Cairns,2,6 Robert P. Stone,3 Hye-Won Moon,4 and Jong Hee Lee 5 Abstract: Six primnoid species are reported from depths of 280 – 480 m from the southern Emperor Seamounts, including two new species (Callogorgia imperialis and Thouarella taylorae). Only the new species are fully described and illustrated. Also, Callogorgia elegans, which has a confused taxonomic history, is discussed and illustrated. Not unexpectedly, the Emperor Seamount primnoids have a strong affinity with the fauna of the Hawaiian Islands, an affinity that is expected to increase as more collecting is done in the region. The United Nations General Assembly nations around the world are developing pro- approved Resolution 61/105 in December tocol and policy on fishing encounters with 2006 ( United Nations General Assembly 2007) the sensitive biota (Durán Muñoz et al. 2012). that calls on States to directly, or through Here we report on collections made on fish- Regional Fisheries Management Organiza- ing vessels in the Emperor Seamounts, North tions, apply a precautionary ecosystem ap- Pacific Ocean, as part of a joint project be- proach to sustainably manage fish stocks and tween the United States and the Republic of protect vulnerable marine ecosystems ( VMEs) Korea. -

Alcyonacea: Primnoidae) from the Southwestern Atlantic
New primnoid genus and species (Alcyonacea: Primnoidae) from the southwestern Atlantic Stephen D. Cairns Department of Invertebrate Zoology, National Museum of Natural History, Smithsonian Institution, Washington, D.C. 20560, U.S.A., e-mail: [email protected] PROCEEDINGS OF THE BIOLOGICAL SOCIETY OF WASHINGTON 125(2):180–188. 2012. New primnoid genus and species (Alcyonacea: Primnoidae) from the southwestern Atlantic Stephen D. Cairns Department of Invertebrate Zoology, National Museum of Natural History, Smithsonian Institution, Washington, D.C. 20560, U.S.A., e-mail: [email protected] Abstract.—A new genus and species of primnoid octocoral, Heptaprimnoa patagonica, is described from deep water from the cold temperate/ Subantarctic region off Argentina and Burdwood Bank. It is distinguished from other primnoid genera by having only seven rows of body wall scales. Keywords: Alcyonacea, Argentina, new genus, new species, Octocorallia, Primnoidae, Subantarctic As noted by Cairns & Bayer (2009), the a new species; Orejas et al. (2007), repro- Primnoidae is a large and diverse octoco- ductive habits of two species; Zapata- ral family, ranking fourth among the 44 Guardiola & Lo´pez-Gonza´lez (2009), two octocoral families in number of species, new species; Cairns & Bayer (2009), three and third in number of genera. Whereas new genera and two new subgenera; Zapa- the primnoids are cosmopolitan in distri- ta-Guardiola & Lo´pez-Gonza´lez (2010a), bution and occur over a depth range of 8– two new genera and species; Zapata-Guar- 5850 m, they are found primarily in deep diola & Lo´pez-Gonza´lez (2010b), four new water, and there seems to be a dispropor- species; Zapata-Guardiola & Lo´pez-Gon- tionate number of species and genera za´lez (2010c), a new genus; and Taylor et al. -

Hourigan TF, Etnoyer PJ, Cairns SD (2017) Introduction to the State of Deep‐Sea Coral and Sponge Ecosystems of the United States
Introduction to the State of Deep-Sea Coral and Sponge Ecosystems of the United States Chapter 1 in The State of Deep‐Sea Coral and Sponge Ecosystems of the United States Report Recommended citation: Hourigan TF, Etnoyer PJ, Cairns SD (2017) Introduction to the State of Deep‐Sea Coral and Sponge Ecosystems of the United States. In: Hourigan TF, Etnoyer, PJ, Cairns, SD (eds.). The State of Deep‐Sea Coral and Sponge Ecosystems of the United States. NOAA Technical Memorandum NMFS‐OHC‐4, Silver Spring, MD. 38 p. Available online: http://deepseacoraldata.noaa.gov/library. Iridogorgia soft coral with squat lobsters in the northwestern Gulf of Mexico. Courtesy of the NOAA Office of Ocean xii Exploration and Research. INTRODUCTION TO THE STATE OF DEEP‐SEA CORAL AND SPONGE ECOSYSTEMS OF THE UNITED STATES INTRODUCTION TO THE Thomas F. STATE OF DEEP-SEA Hourigan1*, Peter J. CORAL AND SPONGE Etnoyer2, and ECOSYSTEMS OF THE Stephen D. Cairns3 UNITED STATES 1 NOAA Deep Sea Coral Research and Technology Program, Office of Habitat Conservation, Silver I. Introduction Spring, MD * Corresponding Author: Large, long‐lived, sessile organisms contribute structural [email protected] complexity to seafloor habitats and play an important role in marine ecosystems. In deep or cold oceanic waters, corals and 2 NOAA Center for Coastal sponges are the most important organisms forming such biogenic Monitoring and Assessment, National habitats (Roberts et al. 2009, Buhl‐Mortensen et al. 2010, Hogg et al. Centers for Coastal Ocean 2010, Rossi et al. 2017). They increase the physical heterogeneity of Science, Charleston, SC habitat, provide refuge and substrate, increase the number and 3 National Museum of availability of micro‐habitats for other organisms, and thereby Natural History, create hotspots of biological diversity in the deep sea. -

CNIDARIA Corals, Medusae, Hydroids, Myxozoans
FOUR Phylum CNIDARIA corals, medusae, hydroids, myxozoans STEPHEN D. CAIRNS, LISA-ANN GERSHWIN, FRED J. BROOK, PHILIP PUGH, ELLIOT W. Dawson, OscaR OcaÑA V., WILLEM VERvooRT, GARY WILLIAMS, JEANETTE E. Watson, DENNIS M. OPREsko, PETER SCHUCHERT, P. MICHAEL HINE, DENNIS P. GORDON, HAMISH J. CAMPBELL, ANTHONY J. WRIGHT, JUAN A. SÁNCHEZ, DAPHNE G. FAUTIN his ancient phylum of mostly marine organisms is best known for its contribution to geomorphological features, forming thousands of square Tkilometres of coral reefs in warm tropical waters. Their fossil remains contribute to some limestones. Cnidarians are also significant components of the plankton, where large medusae – popularly called jellyfish – and colonial forms like Portuguese man-of-war and stringy siphonophores prey on other organisms including small fish. Some of these species are justly feared by humans for their stings, which in some cases can be fatal. Certainly, most New Zealanders will have encountered cnidarians when rambling along beaches and fossicking in rock pools where sea anemones and diminutive bushy hydroids abound. In New Zealand’s fiords and in deeper water on seamounts, black corals and branching gorgonians can form veritable trees five metres high or more. In contrast, inland inhabitants of continental landmasses who have never, or rarely, seen an ocean or visited a seashore can hardly be impressed with the Cnidaria as a phylum – freshwater cnidarians are relatively few, restricted to tiny hydras, the branching hydroid Cordylophora, and rare medusae. Worldwide, there are about 10,000 described species, with perhaps half as many again undescribed. All cnidarians have nettle cells known as nematocysts (or cnidae – from the Greek, knide, a nettle), extraordinarily complex structures that are effectively invaginated coiled tubes within a cell. -

Articles and Detrital Matter
Biogeosciences, 7, 2851–2899, 2010 www.biogeosciences.net/7/2851/2010/ Biogeosciences doi:10.5194/bg-7-2851-2010 © Author(s) 2010. CC Attribution 3.0 License. Deep, diverse and definitely different: unique attributes of the world’s largest ecosystem E. Ramirez-Llodra1, A. Brandt2, R. Danovaro3, B. De Mol4, E. Escobar5, C. R. German6, L. A. Levin7, P. Martinez Arbizu8, L. Menot9, P. Buhl-Mortensen10, B. E. Narayanaswamy11, C. R. Smith12, D. P. Tittensor13, P. A. Tyler14, A. Vanreusel15, and M. Vecchione16 1Institut de Ciencies` del Mar, CSIC. Passeig Mar´ıtim de la Barceloneta 37-49, 08003 Barcelona, Spain 2Biocentrum Grindel and Zoological Museum, Martin-Luther-King-Platz 3, 20146 Hamburg, Germany 3Department of Marine Sciences, Polytechnic University of Marche, Via Brecce Bianche, 60131 Ancona, Italy 4GRC Geociencies` Marines, Parc Cient´ıfic de Barcelona, Universitat de Barcelona, Adolf Florensa 8, 08028 Barcelona, Spain 5Universidad Nacional Autonoma´ de Mexico,´ Instituto de Ciencias del Mar y Limnolog´ıa, A.P. 70-305 Ciudad Universitaria, 04510 Mexico,` Mexico´ 6Woods Hole Oceanographic Institution, MS #24, Woods Hole, MA 02543, USA 7Integrative Oceanography Division, Scripps Institution of Oceanography, La Jolla, CA 92093-0218, USA 8Deutsches Zentrum fur¨ Marine Biodiversitatsforschung,¨ Sudstrand¨ 44, 26382 Wilhelmshaven, Germany 9Ifremer Brest, DEEP/LEP, BP 70, 29280 Plouzane, France 10Institute of Marine Research, P.O. Box 1870, Nordnes, 5817 Bergen, Norway 11Scottish Association for Marine Science, Scottish Marine Institute, Oban, -

Deep-Sea Coral Taxa in the U.S. Southeast Region: Depth and Geographic Distribution (V
Deep-Sea Coral Taxa in the U.S. Southeast Region: Depth and Geographic Distribution (v. 2020) by Thomas F. Hourigan1, Stephen D. Cairns2, John K. Reed3, and Steve W. Ross4 1. NOAA Deep Sea Coral Research and Technology Program, Office of Habitat Conservation, Silver Spring, MD 2. National Museum of Natural History, Smithsonian Institution, Washington, DC 3. Cooperative Institute of Ocean Exploration, Research, and Technology, Harbor Branch Oceanographic Institute, Florida Atlantic University, Fort Pierce, FL 4. Center for Marine Science, University of North Carolina, Wilmington This annex to the U.S. Southeast chapter in “The State of Deep-Sea Coral and Sponge Ecosystems in the United States” provides a list of deep-sea coral taxa in the Phylum Cnidaria, Classes Anthozoa and Hydrozoa, known to occur in U.S. waters from Cape Hatteras to the Florida Keys (Figure 1). Deep-sea corals are defined as azooxanthellate, heterotrophic coral species occurring in waters 50 meters deep or more. Details are provided on the vertical and geographic extent of each species (Table 1). This list is an update of the peer-reviewed 2017 list (Hourigan et al. 2017) and includes taxa recognized through 2019, including one newly described species. Taxonomic names are generally those currently accepted in the World Register of Marine Species (WoRMS), and are arranged by order, and alphabetically within order by family, genus, and species. Data sources (references) listed are those principally used to establish geographic and depth distribution. Figure 1. U.S. Southeast region delimiting the geographic boundaries considered in this work. The region extends from Cape Hatteras to the Florida Keys and includes the Jacksonville Lithoherms (JL), Blake Plateau (BP), Oculina Coral Mounds (OC), Miami Terrace (MT), Pourtalès Terrace (PT), Florida Straits (FS), and Agassiz/Tortugas Valleys (AT).