Analysis of Inflammasomes and Antiviral Sensing Components
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Golgi Phosphoprotein 3 Promotes the Proliferation of Gallbladder Carcinoma Cells Via Regulation of the NLRP3 Inflammasome
ONCOLOGY REPORTS 45: 113, 2021 Golgi phosphoprotein 3 promotes the proliferation of gallbladder carcinoma cells via regulation of the NLRP3 inflammasome ZHENCHENG ZHU1,2*, QINGZHOU ZHU1,2*, DONGPING CAI3, LIANG CHEN4, WEIXUAN XIE2, YANG BAI2 and KUNLUN LUO1,2 1Anhui Medical University, Hefei, Anhui 230032; Departments of 2Hepatobiliary Surgery, 3Laboratory and 4Cardiology, The 904th Hospital of Joint Logistic Support Force of PLA, Wuxi, Jiangsu 214044, P.R. China Received January 18, 2021; Accepted April 2, 2021 DOI: 10.3892/or.2021.8064 Abstract. Golgi phosphoprotein 3 (GOLPH3) has been Introduction demonstrated to promote tumor progression in various gastro‑ intestinal malignancies. However, its effects in gallbladder Gallbladder carcinoma (GBC) is a highly malignant tumor carcinoma (GBC) remain unknown. In the present study, the of the biliary system with a median survival time of only expression levels of GOLPH3 and nucleotide‑binding domain 6 months (1‑3). The primary pathological type of GBC leucine‑rich repeat and pyrin domain containing receptor 3 observed in patients is adenocarcinoma. The effects of current (NLRP3) in human GBC tissues were detected by immuno‑ chemotherapeutic regimens are not sufficient for GBC due histochemistry, and the clinical data and survival of these to the lack of effective drugs, making it particularly difficult patients were analyzed. Next, whether GOLPH3 could affect to control the mortality rate of GBC (3,4). Due to the close tumor proliferation via regulation of the NLRP3 inflamma‑ relationship between inflammation and GBC, investigation of some was investigated in vitro. The results demonstrated that inflammatory‑related molecular mechanisms may highlight GOLPH3 could promote GBC cell proliferation, and that it novel specific targets for the treatment of GBC (4). -

Inflammasome Activation-Induced Hypercoagulopathy
cells Review Inflammasome Activation-Induced Hypercoagulopathy: Impact on Cardiovascular Dysfunction Triggered in COVID-19 Patients Lealem Gedefaw, Sami Ullah, Polly H. M. Leung , Yin Cai, Shea-Ping Yip * and Chien-Ling Huang * Department of Health Technology and Informatics, The Hong Kong Polytechnic University, Kowloon, Hong Kong, China; [email protected] (L.G.); [email protected] (S.U.); [email protected] (P.H.M.L.); [email protected] (Y.C.) * Correspondence: [email protected] (S.-P.Y.); [email protected] (C.-L.H.) Abstract: Coronavirus disease 2019 (COVID-19) is the most devastating infectious disease in the 21st century with more than 2 million lives lost in less than a year. The activation of inflammasome in the host infected by SARS-CoV-2 is highly related to cytokine storm and hypercoagulopathy, which significantly contribute to the poor prognosis of COVID-19 patients. Even though many studies have shown the host defense mechanism induced by inflammasome against various viral infections, mechanistic interactions leading to downstream cellular responses and pathogenesis in COVID-19 remain unclear. The SARS-CoV-2 infection has been associated with numerous cardiovascular disor- ders including acute myocardial injury, myocarditis, arrhythmias, and venous thromboembolism. The inflammatory response triggered by the activation of NLRP3 inflammasome under certain car- diovascular conditions resulted in hyperinflammation or the modulation of angiotensin-converting enzyme 2 signaling pathways. Perturbations of several target cells and tissues have been described in inflammasome activation, including pneumocytes, macrophages, endothelial cells, and dendritic cells. Citation: Gedefaw, L.; Ullah, S.; Leung, P.H.M.; Cai, Y.; Yip, S.-P.; The interplay between inflammasome activation and hypercoagulopathy in COVID-19 patients is an Huang, C.-L. -

NLRP6 Induces Pyroptosis by Activation of Caspase-1 in Gingival
JDRXXX10.1177/0022034518775036Journal of Dental ResearchNLRP6 Induces Pyroptosis 775036research-article2018 Research Reports: Biological Journal of Dental Research 2018, Vol. 97(12) 1391 –1398 © International & American Associations NLRP6 Induces Pyroptosis by Activation for Dental Research 2018 Article reuse guidelines: of Caspase-1 in Gingival Fibroblasts sagepub.com/journals-permissions DOI:https://doi.org/10.1177/0022034518775036 10.1177/0022034518775036 journals.sagepub.com/home/jdr W. Liu1* , J. Liu1*, W. Wang1, Y. Wang2,3, and X. Ouyang1 Abstract NLRP6, a member of the nucleotide-binding domain, leucine-rich repeat-containing (NLR) innate immune receptor family, has been reported to participate in inflammasome formation. Activation of inflammasome triggers a caspase-1–dependent programming cell death called pyroptosis. However, whether NLRP6 induces pyroptosis has not been investigated. In this study, we showed that NLRP6 overexpression activated caspase-1 and gasdermin-D and then induced pyroptosis of human gingival fibroblasts, resulting in release of proinflammatory mediators interleukin (IL)–1β and IL-18. Moreover, NLRP6 was highly expressed in gingival tissue of periodontitis compared with healthy controls. Porphyromonas gingivalis, which is a commensal bacterium and has periodontopathic potential, induced pyroptosis of gingival fibroblasts by activation of NLRP6. Together, we, for the first time, identified that NLRP6 could induce pyroptosis of gingival fibroblasts by activation of caspase-1 and may play a role in periodontitis. Keywords: periodontitis, pattern recognition receptors, cell death, Porphyromonas gingivalis, inflammasomes, flow cytometry Introduction have been demonstrated to participate in periodontitis (Huang et al. 2015; Chaves de Souza et al. 2016; Marchesan et al. Pyroptosis is a newly identified type of programmed cell death 2016). -

Scholarly Commons NLRX1 Modulates Differentially NLRP3
University of the Pacific Scholarly Commons Dugoni School of Dentistry Faculty Articles Arthur A. Dugoni School of Dentistry 10-1-2018 NLRX1 modulates differentially NLRP3 inflammasome activation and NF-κB signaling during Fusobacterium nucleatum infection Shu Chen Hung University of the Pacific Arthur A. Dugoni School of Dentistry Pei Rong Huang Chang Gung University Cassio Luiz Coutinho Almeida-Da-Silva University of the Pacific Arthur A. Dugoni School of Dentistry, [email protected] Kalina R. Atanasova University of Florida Ozlem Yilmaz Medical University of South Carolina See next page for additional authors Follow this and additional works at: https://scholarlycommons.pacific.edu/dugoni-facarticles Part of the Dentistry Commons Recommended Citation Hung, S., Huang, P., Almeida-Da-Silva, C. L., Atanasova, K. R., Yilmaz, O., & Ojcius, D. M. (2018). NLRX1 modulates differentially NLRP3 inflammasome activation and NF-κB signaling during Fusobacterium nucleatum infection. Microbes and Infection, 20(9-10), 615–625. DOI: 10.1016/j.micinf.2017.09.014 https://scholarlycommons.pacific.edu/dugoni-facarticles/705 This Article is brought to you for free and open access by the Arthur A. Dugoni School of Dentistry at Scholarly Commons. It has been accepted for inclusion in Dugoni School of Dentistry Faculty Articles by an authorized administrator of Scholarly Commons. For more information, please contact [email protected]. Authors Shu Chen Hung, Pei Rong Huang, Cassio Luiz Coutinho Almeida-Da-Silva, Kalina R. Atanasova, Ozlem Yilmaz, and David M. Ojcius This article is available at Scholarly Commons: https://scholarlycommons.pacific.edu/dugoni-facarticles/705 Version of Record: https://www.sciencedirect.com/science/article/pii/S1286457917301582 Manuscript_dd7f93413c97aff4865d54242a8b21e7 1 NLRX1 modulates differentially NLRP3 inflammasome activation 2 and NF-κB signaling during Fusobacterium nucleatum infection 3 4 5 Shu-Chen Hung 1, *, Pei-Rong Huang 2, Cássio Luiz Coutinho Almeida-da-Silva 1,3 , 6 Kalina R. -

The Intestinal Parasite Cryptosporidium Is Controlled by an Enterocyte Intrinsic Inflammasome That Depends on NLRP6
The intestinal parasite Cryptosporidium is controlled by an enterocyte intrinsic inflammasome that depends on NLRP6 Adam Saterialea,1, Jodi A. Gullicksruda, Julie B. Engilesa, Briana I. McLeoda, Emily M. Kuglera, Jorge Henao-Mejiab,c, Ting Zhoud, Aaron M. Ringd, Igor E. Brodskya, Christopher A. Huntera, and Boris Striepena,2 aDepartment of Pathobiology, School of Veterinary Medicine, University of Pennsylvania, Philadelphia, PA 19104; bDepartment of Pathology and Laboratory Medicine, Institute for Immunology, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA 19104; cDivision of Protective Immunity, Department of Pathology and Laboratory Medicine, Children’s Hospital of Philadelphia, Philadelphia, PA 19104; and dDepartment of Immunology, Yale School of Medicine, Yale University, New Haven, CT 06519 Edited by Stephen M. Beverley, Washington University School of Medicine, St. Louis, MO, and approved December 1, 2020 (received for review April 24, 2020) The apicomplexan parasite Cryptosporidium infects the intestinal Murine infection with C. tyzzeri resembles human cryptospo- epithelium. While infection is widespread around the world, chil- ridiosis in location, pathology, and resolution and provides an dren in resource-poor settings suffer a disproportionate disease important tool to define the host and parasite factors that burden. Cryptosporidiosis is a leading cause of diarrheal disease, determine the outcome of infection and to identify the im- responsible for mortality and stunted growth in children. CD4 mune -

Post-Transcriptional Inhibition of Luciferase Reporter Assays
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 287, NO. 34, pp. 28705–28716, August 17, 2012 © 2012 by The American Society for Biochemistry and Molecular Biology, Inc. Published in the U.S.A. Post-transcriptional Inhibition of Luciferase Reporter Assays by the Nod-like Receptor Proteins NLRX1 and NLRC3* Received for publication, December 12, 2011, and in revised form, June 18, 2012 Published, JBC Papers in Press, June 20, 2012, DOI 10.1074/jbc.M111.333146 Arthur Ling‡1,2, Fraser Soares‡1,2, David O. Croitoru‡1,3, Ivan Tattoli‡§, Leticia A. M. Carneiro‡4, Michele Boniotto¶, Szilvia Benko‡5, Dana J. Philpott§, and Stephen E. Girardin‡6 From the Departments of ‡Laboratory Medicine and Pathobiology and §Immunology, University of Toronto, Toronto M6G 2T6, Canada, and the ¶Modulation of Innate Immune Response, INSERM U1012, Paris South University School of Medicine, 63, rue Gabriel Peri, 94276 Le Kremlin-Bicêtre, France Background: A number of Nod-like receptors (NLRs) have been shown to inhibit signal transduction pathways using luciferase reporter assays (LRAs). Results: Overexpression of NLRX1 and NLRC3 results in nonspecific post-transcriptional inhibition of LRAs. Conclusion: LRAs are not a reliable technique to assess the inhibitory function of NLRs. Downloaded from Significance: The inhibitory role of NLRs on specific signal transduction pathways needs to be reevaluated. Luciferase reporter assays (LRAs) are widely used to assess the Nod-like receptors (NLRs)7 represent an important class of activity of specific signal transduction pathways. Although pow- intracellular pattern recognition molecules (PRMs), which are erful, rapid and convenient, this technique can also generate implicated in the detection and response to microbe- and dan- www.jbc.org artifactual results, as revealed for instance in the case of high ger-associated molecular patterns (MAMPs and DAMPs), throughput screens of inhibitory molecules. -

NOD-Like Receptors in the Eye: Uncovering Its Role in Diabetic Retinopathy
International Journal of Molecular Sciences Review NOD-like Receptors in the Eye: Uncovering Its Role in Diabetic Retinopathy Rayne R. Lim 1,2,3, Margaret E. Wieser 1, Rama R. Ganga 4, Veluchamy A. Barathi 5, Rajamani Lakshminarayanan 5 , Rajiv R. Mohan 1,2,3,6, Dean P. Hainsworth 6 and Shyam S. Chaurasia 1,2,3,* 1 Ocular Immunology and Angiogenesis Lab, University of Missouri, Columbia, MO 652011, USA; [email protected] (R.R.L.); [email protected] (M.E.W.); [email protected] (R.R.M.) 2 Department of Biomedical Sciences, University of Missouri, Columbia, MO 652011, USA 3 Ophthalmology, Harry S. Truman Memorial Veterans’ Hospital, Columbia, MO 652011, USA 4 Surgery, University of Missouri, Columbia, MO 652011, USA; [email protected] 5 Singapore Eye Research Institute, Singapore 169856, Singapore; [email protected] (V.A.B.); [email protected] (R.L.) 6 Mason Eye Institute, School of Medicine, University of Missouri, Columbia, MO 652011, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-573-882-3207 Received: 9 December 2019; Accepted: 27 January 2020; Published: 30 January 2020 Abstract: Diabetic retinopathy (DR) is an ocular complication of diabetes mellitus (DM). International Diabetic Federations (IDF) estimates up to 629 million people with DM by the year 2045 worldwide. Nearly 50% of DM patients will show evidence of diabetic-related eye problems. Therapeutic interventions for DR are limited and mostly involve surgical intervention at the late-stages of the disease. The lack of early-stage diagnostic tools and therapies, especially in DR, demands a better understanding of the biological processes involved in the etiology of disease progression. -

Pattern Recognition Receptors in Health and Diseases
Signal Transduction and Targeted Therapy www.nature.com/sigtrans REVIEW ARTICLE OPEN Pattern recognition receptors in health and diseases Danyang Li1,2 and Minghua Wu1,2 Pattern recognition receptors (PRRs) are a class of receptors that can directly recognize the specific molecular structures on the surface of pathogens, apoptotic host cells, and damaged senescent cells. PRRs bridge nonspecific immunity and specific immunity. Through the recognition and binding of ligands, PRRs can produce nonspecific anti-infection, antitumor, and other immunoprotective effects. Most PRRs in the innate immune system of vertebrates can be classified into the following five types based on protein domain homology: Toll-like receptors (TLRs), nucleotide oligomerization domain (NOD)-like receptors (NLRs), retinoic acid-inducible gene-I (RIG-I)-like receptors (RLRs), C-type lectin receptors (CLRs), and absent in melanoma-2 (AIM2)-like receptors (ALRs). PRRs are basically composed of ligand recognition domains, intermediate domains, and effector domains. PRRs recognize and bind their respective ligands and recruit adaptor molecules with the same structure through their effector domains, initiating downstream signaling pathways to exert effects. In recent years, the increased researches on the recognition and binding of PRRs and their ligands have greatly promoted the understanding of different PRRs signaling pathways and provided ideas for the treatment of immune-related diseases and even tumors. This review describes in detail the history, the structural characteristics, ligand recognition mechanism, the signaling pathway, the related disease, new drugs in clinical trials and clinical therapy of different types of PRRs, and discusses the significance of the research on pattern recognition mechanism for the treatment of PRR-related diseases. -

Examining the Role of Non-Canonical NOD-Like Receptors and Inflammasomes in Inflammation and Disease
University of Calgary PRISM: University of Calgary's Digital Repository Graduate Studies The Vault: Electronic Theses and Dissertations 2018-03-21 Examining the Role of Non-Canonical NOD-like Receptors and Inflammasomes in Inflammation and Disease Platnich, Jaye Matthew Platnich, J. M. (2018). Examining the Role of Non-Canonical NOD-like Receptors and Inflammasomes in Inflammation and Disease (Unpublished doctoral thesis). University of Calgary, Calgary, AB. doi:10.11575/PRISM/31757 http://hdl.handle.net/1880/106465 doctoral thesis University of Calgary graduate students retain copyright ownership and moral rights for their thesis. You may use this material in any way that is permitted by the Copyright Act or through licensing that has been assigned to the document. For uses that are not allowable under copyright legislation or licensing, you are required to seek permission. Downloaded from PRISM: https://prism.ucalgary.ca UNIVERSITY OF CALGARY Examining the Role of Non-Canonical NOD-like Receptors and Inflammasomes in Inflammation and Disease by Jaye Matthew Platnich A THESIS SUBMITTED TO THE FACULTY OF GRADUATE STUDIES IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY GRADUATE PROGRAM IN IMMUNOLOGY CALGARY, ALBERTA MARCH, 2018 © Jaye Matthew Platnich 2018 Abstract The NOD-like Receptors (NLRs) are a family of pattern recognition receptors known to regulate a variety of immune signaling pathways. A substantial portion of NLR research focuses on the pyrin domain-containing NLRP subfamily. The canonical NLRPs are inflammasome-forming proteins responsible for the activation of caspase-1 and the maturation and secretion of the pro-inflammatory cytokines IL-1β and IL-18. -
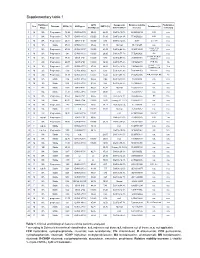
Supplementary Table 2 Supplementary Table 1
Supplementary table 1 Rai/ Binet IGHV Cytogenetic Relative viability Fludarabine- Sex Outcome CD38 (%) IGHV gene ZAP70 (%) Treatment (s) Stage identity (%) abnormalities* increase refractory 1 M 0/A Progressive 14,90 IGHV3-64*05 99,65 28,20 Del17p 18.0% 62,58322819 FCR n.a. 2 F 0/A Progressive 78,77 IGHV3-48*03 100,00 51,90 Del17p 24.8% 77,88052021 FCR n.a. 3 M 0/A Progressive 29,81 IGHV4-b*01 100,00 9,10 Del17p 12.0% 36,48 Len, Chl n.a. 4 M 1/A Stable 97,04 IGHV3-21*01 97,22 18,11 Normal 85,4191657 n.a. n.a. Chl+O, PCR, 5 F 0/A Progressive 87,00 IGHV4-39*07 100,00 43,20 Del13q 68.3% 35,23314039 n.a. HDMP+R 6 M 0/A Progressive 1,81 IGHV3-43*01 100,00 20,90 Del13q 77.7% 57,52490626 Chl n.a. Chl, FR, R-CHOP, 7 M 0/A Progressive 97,80 IGHV1-3*01 100,00 9,80 Del17p 88.5% 48,57389901 n.a. HDMP+R 8 F 2/B Progressive 69,07 IGHV5-a*03 100,00 16,50 Del17p 77.2% 107,9656878 FCR, BA No R-CHOP, FCR, 9 M 1/A Progressive 2,13 IGHV3-23*01 97,22 29,80 Del11q 16.3% 134,5866919 Yes Flavopiridol, BA 10 M 2/A Progressive 0,36 IGHV3-30*02 92,01 0,38 Del13q 81.9% 78,91844953 Unknown n.a. 11 M 2/B Progressive 15,17 IGHV3-20*01 100,00 13,20 Del11q 95.3% 75,52880995 FCR, R-CHOP, BR No 12 M 0/A Stable 0,14 IGHV3-30*02 90,62 7,40 Del13q 13.0% 13,0939004 n.a. -

ATP-Binding and Hydrolysis in Inflammasome Activation
molecules Review ATP-Binding and Hydrolysis in Inflammasome Activation Christina F. Sandall, Bjoern K. Ziehr and Justin A. MacDonald * Department of Biochemistry & Molecular Biology, Cumming School of Medicine, University of Calgary, 3280 Hospital Drive NW, Calgary, AB T2N 4Z6, Canada; [email protected] (C.F.S.); [email protected] (B.K.Z.) * Correspondence: [email protected]; Tel.: +1-403-210-8433 Academic Editor: Massimo Bertinaria Received: 15 September 2020; Accepted: 3 October 2020; Published: 7 October 2020 Abstract: The prototypical model for NOD-like receptor (NLR) inflammasome assembly includes nucleotide-dependent activation of the NLR downstream of pathogen- or danger-associated molecular pattern (PAMP or DAMP) recognition, followed by nucleation of hetero-oligomeric platforms that lie upstream of inflammatory responses associated with innate immunity. As members of the STAND ATPases, the NLRs are generally thought to share a similar model of ATP-dependent activation and effect. However, recent observations have challenged this paradigm to reveal novel and complex biochemical processes to discern NLRs from other STAND proteins. In this review, we highlight past findings that identify the regulatory importance of conserved ATP-binding and hydrolysis motifs within the nucleotide-binding NACHT domain of NLRs and explore recent breakthroughs that generate connections between NLR protein structure and function. Indeed, newly deposited NLR structures for NLRC4 and NLRP3 have provided unique perspectives on the ATP-dependency of inflammasome activation. Novel molecular dynamic simulations of NLRP3 examined the active site of ADP- and ATP-bound models. The findings support distinctions in nucleotide-binding domain topology with occupancy of ATP or ADP that are in turn disseminated on to the global protein structure. -

Inflammasome Activation and Regulation
Zheng et al. Cell Discovery (2020) 6:36 Cell Discovery https://doi.org/10.1038/s41421-020-0167-x www.nature.com/celldisc REVIEW ARTICLE Open Access Inflammasome activation and regulation: toward a better understanding of complex mechanisms Danping Zheng1,2,TimurLiwinski1,3 and Eran Elinav 1,4 Abstract Inflammasomes are cytoplasmic multiprotein complexes comprising a sensor protein, inflammatory caspases, and in some but not all cases an adapter protein connecting the two. They can be activated by a repertoire of endogenous and exogenous stimuli, leading to enzymatic activation of canonical caspase-1, noncanonical caspase-11 (or the equivalent caspase-4 and caspase-5 in humans) or caspase-8, resulting in secretion of IL-1β and IL-18, as well as apoptotic and pyroptotic cell death. Appropriate inflammasome activation is vital for the host to cope with foreign pathogens or tissue damage, while aberrant inflammasome activation can cause uncontrolled tissue responses that may contribute to various diseases, including autoinflammatory disorders, cardiometabolic diseases, cancer and neurodegenerative diseases. Therefore, it is imperative to maintain a fine balance between inflammasome activation and inhibition, which requires a fine-tuned regulation of inflammasome assembly and effector function. Recently, a growing body of studies have been focusing on delineating the structural and molecular mechanisms underlying the regulation of inflammasome signaling. In the present review, we summarize the most recent advances and remaining challenges in understanding the ordered inflammasome assembly and activation upon sensing of diverse stimuli, as well as the tight regulations of these processes. Furthermore, we review recent progress and challenges in translating inflammasome research into therapeutic tools, aimed at modifying inflammasome-regulated human diseases.