Antiretroviral Drugs: Critical Issues and Recent Advances
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

WO 2018/005909 Al 04 January 2018 (04.01.2018) W !P O PCT
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2018/005909 Al 04 January 2018 (04.01.2018) W !P O PCT (51) International Patent Classification: A61P 31/12 (2006 .01) A61K 31/505 (2006 .0 1) A61K 31/4985 (2006.01) (21) International Application Number: PCT/US20 17/040 175 (22) International Filing Date: 30 June 2017 (30.06.2017) (25) Filing Language: English (26) Publication Language: English (30) Priority Data: 62/357,458 0 1 July 2016 (01 .07.2016) US (71) Applicant: VIIV HEALTHCARE COMPANY [US/US]; 25 1 Little Falls Drive, Wilmington, DE 19808 (US). (72) Inventor: SPREEN, William, R.; 5 Moore Drive, Re search Triangle Park, NC 27709-3398 (US). (74) Agent: HAN, William, T. et al; Glaxosmithkline, Glob al Patents, UW2220, 709 Swedeland Road, P.O. Box 1539, King of Prussia, PA 19406-0939 (US). (81) Designated States (unless otherwise indicated, for every kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DJ, DK, DM, DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, HN, HR, HU, ID, IL, IN, IR, IS, JO, JP, KE, KG, KH, KN, KP, KR, KW, KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. -

Download Article PDF/Slides
Kan Lu, PharmD New Antiretrovirals for Based on a presentation at prn by Roy M. Gulick, md, mph the Treatment of HIV: Kan Lu, PharmD | Drug Development Fellow University of North Carolina School of Pharmacy Chapel Hill, North Carolina The View in 2006 Roy M. Gulick, md, mph Reprinted from The prn Notebook® | october 2006 | Dr. James F. Braun, Editor-in-Chief Director, Cornell Clinical Trials Unit | Associate Professor of Medicine, Meri D. Pozo, PhD, Managing Editor. Published in New York City by the Physicians’ Research Network, Inc.® Weill Medical College of Cornell University | New York, New York John Graham Brown, Executive Director. For further information and other articles available online, visit http://www.prn.org | All rights reserved. ©october 2006 substantial progress continues to be made in the arena of cokinetics and a long extracellular half-life of approximately 10 hours antiretroviral drug development. prn is again proud to present its annual (Zhu, 2003). During apricitabine’s development, a serious drug interac- review of the experimental agents to watch for in the coming months and tion with lamivudine (Epivir) was noted. Although the plasma years. This year’s review is based on a lecture by Dr. Roy M. Gulick, a long- concentrations of apricitabine were unaffected by coadministration of time friend of prn, and no stranger to the antiretroviral development lamivudine, the intracellular concentrations of apricitabine were reduced pipeline. by approximately sixfold. Additionally, the 50% inhibitory concentration To date, twenty-two antiretrovirals have been approved by the Food (ic50) of apricitabine against hiv with the M184V mutation was increased and Drug Administration (fda) for the treatment of hiv infection. -

Ep 2531027 B1
(19) TZZ ¥_Z _T (11) EP 2 531 027 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 31/4985 (2006.01) A61K 31/52 (2006.01) 06.05.2015 Bulletin 2015/19 A61K 31/536 (2006.01) A61K 31/513 (2006.01) A61K 38/55 (2006.01) A61P 31/18 (2006.01) (21) Application number: 11737484.3 (86) International application number: (22) Date of filing: 24.01.2011 PCT/US2011/022219 (87) International publication number: WO 2011/094150 (04.08.2011 Gazette 2011/31) (54) Therapeutic combination comprising dolutegravir, abacavir and lamivudine Therapeutische Zusammensetzung enthaltend Dolutegravir, Abacavir und Lamivudine Combinaison thérapeutique comprenant du dolutégravir, de l’abacavir et de la lamivudine (84) Designated Contracting States: (74) Representative: Gladwin, Amanda Rachel AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GlaxoSmithKline GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO Global Patents (CN925.1) PL PT RO RS SE SI SK SM TR 980 Great West Road Designated Extension States: Brentford, Middlesex TW8 9GS (GB) BA ME (56) References cited: (30) Priority: 27.01.2010 US 298589 P WO-A1-2010/011812 WO-A2-2009/148600 US-A1- 2006 084 627 US-A1- 2006 084 627 (43) Date of publication of application: US-A1- 2008 076 738 US-A1- 2009 318 421 12.12.2012 Bulletin 2012/50 US-A1- 2009 318 421 US-B1- 6 544 961 (73) Proprietor: VIIV Healthcare Company • SONG1 et al: "The Effect of Ritonavir-Boosted Research Triangle Park, NC 27709 (US) ProteaseInhibitors on the HIV Integrase Inhibitor, S/GSK1349572,in Healthy Subjects", INTERNET , (72) Inventor: UNDERWOOD, Mark, Richard 15 September 2009 (2009-09-15), XP002697436, Research Triangle Park Retrieved from the Internet: URL:http: North Carolina 27709 (US) //www.natap.org/2009/ICCAC/ICCAC_ 52.htm [retrieved on 2013-05-21] Note: Within nine months of the publication of the mention of the grant of the European patent in the European Patent Bulletin, any person may give notice to the European Patent Office of opposition to that patent, in accordance with the Implementing Regulations. -

Trends in Antiretroviral Treatment in Australia
AUSTRALIAN HIV OBSERVATIONAL DATABASE (AHOD) ANNUAL REPORT (Volume 20, Number 1: December 2020) 2020 Clinical characteristics of overseas-born men who have sex with men (MSM) in the AHOD cohort and implications for clinical practice In Australia HIV notifications are increasing among overseas-born men who have sex with men (MSM), particularly among Asian-born MSM. Australian evidence suggests that culturally and/or linguistically diverse populations are less likely to start treatment early irrespective of CD4 cell count at diagnosis, but little is known about response once in care. Using data from AHOD, Jolie L Hutchinson and colleagues (2020) compared treatment response in overseas-born MSM from non-English-speaking countries with Australian-born MSM, further categorised based on participation in the Australian Temporary Residents Access Study (ATRAS) which provide temporary residents ineligible for Medicare, access to HIV treatment. ATRAS patients were chosen as the closest surrogate to identifying newly arrived overseas-born MSM. The authors explored the time to first virological suppression (VS) (viral load (VL) <400 copies/mL) and time to virological failure (VF) (>400 copies/mL after suppression). CD4 cell counts and VL measurements were taken at treatment initiation. Adjusted Hazard Ratios (HR) are reported with 95% CI. Results, as shown in figure 1, indicate that overseas-born MSM did not differ significantly in the rate of VS or in the rate of first VF after suppression. This result is different from findings in other settings, and differences may, in part, be explained by the nature of healthcare provision. In Australia, all residents can access ART for free or with a small co-payment; those ineligible for Medicare can get pharmaceutical company-provided ART which is not necessarily straightforward for non-English speakers. -

PHARMACEUTICAL APPENDIX to the TARIFF SCHEDULE 2 Table 1
Harmonized Tariff Schedule of the United States (2020) Revision 19 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2020) Revision 19 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names INN which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service CAS registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. -

HIV/AIDS Technologies: a Review of Progress to Date and Current Prospects
Working Paper No.6 HIV/AIDS Technologies: A review of progress to date and current prospects COMMISSIONED BY: aids2031 Science and Technology Working Group AUTHORED BY: KEITH ALCORN NAM Publications Disclaimer: The views expressed in this paper are those of the author(s) and do not necessarily reflect the official policy, position, or opinions of the wider aids2031 initiative or partner organizations aids2031 Science and Technology working group A review of progress to date and current prospects October 2008 Acronyms 3TC lamivudine ANRS Agènce Nationale de Récherche sur la Sida ART Antiretroviral therapy ARV Antiretroviral AZT azidothymidine or zidovudine bDNA branched DNA CDC US Centers for Disease Control CHER Children with HIV Early Antiretroviral therapy (study) CTL Cytotoxic T-lymphocyte D4T stavudine DSMB Data and Safety Monitoring Board EFV Efavirenz ELISA Enzyme Linked Immunosorbent Assay FDC Fixed-dose combination FTC Emtricitabine HAART Highly Active Antiretroviral Therapy HBAC Home-based AIDS care HCV Hepatitis C virus HPTN HIV Prevention Trials Network HSV-2 Herpes simplex virus type 2 IAVI International AIDS Vaccine Initiative IL-2 Interleukin-2 LED Light-emitting diode LPV/r Lopinavir/ritonavir MIRA Methods for Improving Reproductive Health in Africa trial MSF Médecins sans Frontières MSM Men who have sex with men MVA Modified vaccinia Ankara NIH US National Institutes of Health NRTI Nucleoside reverse transcriptase inhibitor NNRTI Non-nucleoside reverse transcriptase inhibitor OBT Optimised background therapy PCR Polymerase -

Synthesis and Development of Long-Acting Abacavir Prodrug Nanoformulations
University of Nebraska Medical Center DigitalCommons@UNMC Theses & Dissertations Graduate Studies Summer 8-19-2016 Synthesis and Development of Long-Acting Abacavir Prodrug Nanoformulations Dhirender Singh University of Nebraska Medical Center Follow this and additional works at: https://digitalcommons.unmc.edu/etd Part of the Pharmaceutical Preparations Commons, and the Virus Diseases Commons Recommended Citation Singh, Dhirender, "Synthesis and Development of Long-Acting Abacavir Prodrug Nanoformulations" (2016). Theses & Dissertations. 140. https://digitalcommons.unmc.edu/etd/140 This Dissertation is brought to you for free and open access by the Graduate Studies at DigitalCommons@UNMC. It has been accepted for inclusion in Theses & Dissertations by an authorized administrator of DigitalCommons@UNMC. For more information, please contact [email protected]. SYNTHESIS AND DEVELOPMENT OF LONG-ACTING ABACAVIR PRODRUG NANOFORMULATIONS by Dhirender Singh A DISSERTATION Presented to the Faculty of the Graduate School in the University of Nebraska Medical Center in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy Department of Pharmaceutical Science Under the Supervision of Dr. Howard E. Gendelman University of Nebraska Medical Center, Omaha, Nebraska August 2016 Supervisory Committee: Howard E. Gendelman, M.D. Ram Mahato, Ph.D JoEllyn M. McMillan, Ph.D. David Oupicky, Ph.D SYNTHESIS AND DEVELOPMENT OF LONG-ACTING ABACAVIR PRODRUG NANOFORMULATIONS Dhirender Singh, Ph.D. University of Nebraska Medical Center, 2016 Supervisor: Howard E Gendelman, M.D. Over the past decade, work from our laboratory has demonstrated the potential of targeted nanoformulated antiretroviral therapy (nanoART) to produce sustained high plasma and tissue drug concentrations for weeks following a single intramuscular (IM) administration that can suppress ongoing viral replication and mitigate dose associated viral resistance. -

GSK and Vertex Pharmaceuticals Announce Presentation of Data Supporting Development of Investigational HIV Protease Inhibitor Brecanavir
December 16, 2005 GSK and Vertex Pharmaceuticals Announce Presentation of Data Supporting Development of Investigational HIV Protease Inhibitor Brecanavir - Positive Data Presented at 45th Annual ICAAC - Cambridge, MA, December 16, 2005 - GlaxoSmithKline (GSK) and Vertex Pharmaceuticals Incorporated (Nasdaq: VRTX) presented positive results today from a study evaluating the safety, tolerability and antiviral activity of the investigational HIV-1 protease inhibitor (PI), brecanavir* (formerly known as GW640385 or VX-385)1. These data were presented at the 45th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC) held in Washington DC. Interim findings following 24 weeks of dosing demonstrated potent antiviral activity for brecanavir in both PI-sensitive and PI-resistant HIV-infected adults. "These results support the ongoing development program for brecanavir, which is anticipated to enter Phase III development in 2006," said Lynn Marks, MD, Senior Vice President of the Infectious Disease Medicines Development Centre at GSK. "If approved, brecanavir may be useful in treating patients infected with strains of HIV that have become resistant to multiple protease inhibitors, and its clinical advancement underscores GSK's commitment to developing new anti-HIV drugs." Clinical Data on Brecanavir Brecanavir is an HIV protease inhibitor in Phase IIb clinical development. Brecanavir has received fast track designation from the U.S. Food and Drug Administration and is being developed by GSK as part of a collaboration with Vertex Pharmaceuticals. Data presented were from a planned, 24-week analysis of an open-label study (HPR10006) of 48 weeks' duration evaluating the safety, tolerability, antiviral activity and pharmacokinetics of ritonavir-boosted brecanavir. Thirty-one HIV-1 infected adults received 300mg of brecanavir twice-daily boosted with 100mg of ritonavir in combination with two nucleoside reverse transcriptase inhibitors based on patient medical history and viral genotype. -
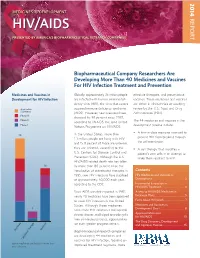
14-258 Phrma HIV/AIDS2014 0819.Indd
2014 MEDICINES IN DEVELOPMENT REPORT HIV/AIDS PRESENTED BY AMERICA’S BIOPHARMACEUTICAL RESEARCH COMPANIES Biopharmaceutical Company Researchers Are Developing More Than 40 Medicines and Vaccines For HIV Infection Treatment and Prevention Medicines and Vaccines in Globally, approximately 35 million people effective therapies, and preventative Development for HIV Infection are infected with human immunodefi - vaccines. These medicines and vaccines ciency virus (HIV), the virus that causes are either in clinical trials or awaiting Application acquired immune defi ciency syndrome review by the U.S. Food and Drug Submitted (AIDS). However, new infections have Administration (FDA). Phase III dropped by 38 percent since 2001, Phase II The 44 medicines and vaccines in the according to UNAIDS, the Joint United Phase I development pipeline include: Nations Programme on HIV/AIDS. • A fi rst-in-class medicine intended to In the United States, more than 25 prevent HIV from breaking through 1.1 million people are living with HIV the cell membrane. and 15.8 percent of those are unaware they are infected, according to the • A cell therapy that modifi es a U.S. Centers for Disease Control and patient’s own cells in an attempt to Prevention (CDC). Although the U.S. make them resistant to HIV. HIV/AIDS-related death rate has fallen 16 by more than 80 percent since the introduction of antiretroviral therapies in Contents 1995, new HIV infections have stabilized HIV Medicines and Vaccines in at approximately 50,000 each year, Development ......................................2 according to the CDC. Incremental Innovation in HIV/AIDS Treatment .......................... 4 Since AIDS was fi rst reported in 1981, Access to HIV/AIDS Medicines in nearly 40 medicines have been approved Exchange Plans ...................................5 to treat HIV infection in the United Facts About HIV/AIDS ........................7 States. -

Innovation in Antiretrovirals to Meet Developing Country Needs
Meeting Summary: Centre on Global Health Security Innovation in Antiretrovirals to Meet Developing Country Needs July 2011 The views expressed in this document are the sole responsibility of the author(s) and do not necessarily reflect the view of Chatham House, its staff, associates or Council. Chatham House is independent and owes no allegiance to any government or to any political body. It does not take institutional positions on policy issues. This document is issued on the understanding that if any extract is used, the author(s)/ speaker(s) and Chatham House should be credited, preferably with the date of the publication or details of the event. Where this document refers to or reports statements made by speakers at an event every effort has been made to provide a fair representation of their views and opinions, but the ultimate responsibility for accuracy lies with this document’s author(s). The published text of speeches and presentations may differ from delivery. Innovation in Antiretrovirals to Meet Developing Country Needs ACRONYMS 3TC Lamivudine ABC Abacavir ART Antiretroviral treatment ARV Antiretroviral ATV Atazanavir AZT Zidovudine CHAI Clinton Health Access Initiative DNDi Drugs for Neglected Diseases initiative EDCTP European and Developing Countries Clinical Trials Partnership EFV Efavirenz FDC Fixed-dose combination IP Intellectual property HBV Hepatitis B virus HCV Hepatitis C virus HIV Human immunodeficiency virus LPV Lopinavir RTV, r Ritonavir TB Tuberculosis TDF Tenofovir Disoproxil Fumarate UN United Nations WHO World Health Organization www.chathamhouse.org.uk 2 Innovation in Antiretrovirals to Meet Developing Country Needs Background On July 11, 2011, Chatham House, the Medicines Patent Pool, UNITAID and the World Health Organization (WHO), convened an expert meeting to identify the key innovation needs in the field of antiretroviral treatment for resource-limited settings, and to discuss the role of different actors in ensuring that the necessary innovation is undertaken. -

Wibke E. Diederich Holger Steuber Editors Therapy of Viral Infections 15 Topics in Medicinal Chemistry
Topics in Medicinal Chemistry 15 Wibke E. Diederich Holger Steuber Editors Therapy of Viral Infections 15 Topics in Medicinal Chemistry Editorial Board: P. R. Bernstein, Rose Valley, USA A. Buschauer, Regensburg, Germany G. I. Georg, Minneapolis, USA J. A. Lowe, Stonington, USA U. Stilz, Malov, Denmark Prof. Dr. C. T. Supuran, Sesto Fiorentino (Firenze), Italy A. K. Saxena, Lucknow, India Aims and Scope Drug research requires interdisciplinary team-work at the interface between chemistry, biology and medicine. Therefore, the new topic-related series Topics in Medicinal Chemistry will cover all relevant aspects of drug research, e.g. pathobiochemistry of diseases, identification and validation of (emerging) drug targets, structural biology, drugability of targets, drug design approaches, chemogenomics, synthetic chemistry including combinatorial methods, bioorganic chemistry, natural compounds, high-throughput screening, pharmacological in vitro and in vivo investigations, drug-receptor interactions on the molecular level, structure-activity relationships, drug absorption, distribution, metabolism, elimina- tion, toxicology and pharmacogenomics. In general, special volumes are edited by well known guest editors. In references Topics in Medicinal Chemistry is abbreviated Top Med Chem and is cited as a journal. More information about this series at http://www.springer.com/series/7355 Wibke E. Diederich • Holger Steuber Editors Therapy of Viral Infections With contributions by E. Bo¨ttcher-Friebertsha¨user Á U. Chiacchio Á F. Christ Á E. Crespan Á J. Demeulemeester Á Z. Debyser Á U. Dietrich Á W.E. Diederich Á R. Du¨rr Á F.G.R. Ehlert Á C. Frezza Á A. Garbelli Á W. Garten Á S.V. Giofre` Á K. Hardes Á M. -

Screening of Clinically Approved and Investigation Drugs As Potential
Screening of Clinically Approved and Investigation Drugs as Potential Inhibitors of COVID-19 Main Protease: A Virtual Drug Repurposing Study Serdar Durdagi1,*, Busecan Aksoydan1,2, Berna Dogan1, Kader Sahin1, Aida Shahraki1,3 1Computational Biology and Molecular Simulations Laboratory, Department of Biophysics, School of Medicine, Bahcesehir University, Istanbul, Turkey 2Neuroscience Program, Graduate School of Health Sciences, Bahçeşehir University, Istanbul, Turkey 3Department of Molecular Biology and Genetics, Bogazici University, Istanbul, Turkey Abstract There is an urgent need for a new drug against COVID-19. Since designing a new drug and testing its pharmacokinetics and pharmacodynamics properties may take years, here we used a physics-driven high throughput virtual screening drug re-purposing approach to identify new compounds against COVID-19. As the molecules considered in repurposing studies passed through several stages and have well-defined profiles, they would not require prolonged pre- clinical studies and hence, they would be excellent candidates in the cases of disease emergencies or outbreaks. While the spike protein is the key for the virus to enter the cell though the interaction with ACE2, enzymes such as main protease are crucial for the life cycle of the virus. This protein is one of the most attractive targets for the development of new drugs against COVID-19 due to its pivotal role in the replication and transcription of the virus. We used 7922 FDA approved small molecule drugs as well as compounds in clinical investigation from NIH Chemical Genomics Center (NCGC) Pharmaceutical Collection (NPC) database in our drug repurposing study. Both apo and holo forms of target protein COVID-19 main proteases were used in virtual screening.