Probing the Solubility of Selected Nanoscale Building Blocks
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Calorimetric Studies of Solvates of C60 and C70 with Aromatic Solvents
J. Phys. Chem. B 1999, 103, 1339-1346 1339 Calorimetric Studies of Solvates of C60 and C70 with Aromatic Solvents Mikhail V. Korobov, Andrej L. Mirakyan, and Natalia V. Avramenko Department of Chemistry, Moscow State UniVersity, 119899 Moscow, Russia Gerd Olofsson* Thermochemistry DiVision, Center for Chemistry and Chemical Engineering, Lund UniVersity, S-22100 Lund, Sweden Allan L. Smith* Chemistry Department, Drexel UniVersity, Philadelphia, PennsylVania 19104 Rodney S. Ruoff Physics Department, Washington UniVersity, St. Louis, Missouri 63130 ReceiVed: July 24, 1998; In Final Form: December 18, 1998 To improve the understanding of the solution properties of C60 and C70 in aromatic solvents, binary systems of C60 and C70 with benzene, toluene, 1,2- and 1,3-dimethylbenzene, 1,2,4- and 1,3,5-trimethylbenzene, bromobenzene, and 1,2- and 1,3-dichlorobenzene were studied using differential scanning calorimetry, solution calorimetry, and thermogravimetry. Solid solvates with different compositions were identified in many of the systems. The solvates were characterized by composition and by the temperature and the enthalpy of the incongruent melting transition. Enthalpies of solution of C60 in toluene, 1,2-dimethylbenzene, 1,2- and 1,3- dichlorobenzene, and 1,2,4-trimethylbenzene and of C70 in 1,2-dimethylbenzene and in 1,2- and 1,3- dichlorobenzene were determined. The formation-incongruent melting of solid solvates causes maxima in the temperature-solubility curves of fullerenes in aromatic solvents. Trends in solubility behavior of fullerenes were discussed in terms of thermodynamics of solution and solvate formation. Introduction solvate (II), or more probably, pure C60. Phase equilibrium conditions relate the mole fraction of C60, x(C60), to T (tem- The unusual temperature dependence of solubility of fullerenes perature) for both low- and high-temperature parts of the (ln x)- 1 in a number of solvents has attracted attention and provoked T curve. -

Carbon Nanostructures Doped with Transition Metals for Pollutant Gas Adsorption Systems
molecules Review Carbon Nanostructures Doped with Transition Metals for Pollutant Gas Adsorption Systems J. M. Ramirez-de-Arellano 1 , M. Canales 2 and L. F. Magaña 3,* 1 Escuela de Ingeniería y Ciencias, Tecnologico de Monterrey, Av. Eugenio Garza Sada 2501, Monterrey 64849, Mexico; [email protected] 2 Universidad Autónoma Metropolitana Unidad Azcapotzalco, Av. San Pablo Xalpa No. 180, Colonia Reynosa Tamaulipas, Delegación Azcapotzalco, Ciudad de México 02200, Mexico; [email protected] 3 Instituto de Física, Universidad Nacional Autónoma de Mexico, Apartado Postal 20-364, Ciudad de México 01000, Mexico * Correspondence: fernando@fisica.unam.mx Abstract: The adsorption of molecules usually increases capacity and/or strength with the doping of surfaces with transition metals; furthermore, carbon nanostructures, i.e., graphene, carbon nanotubes, fullerenes, graphdiyne, etc., have a large specific area for gas adsorption. This review focuses on the reports (experimental or theoretical) of systems using these structures decorated with transition metals for mainly pollutant molecules’ adsorption. Furthermore, we aim to present the expanding application of nanomaterials on environmental problems, mainly over the last 10 years. We found a wide range of pollutant molecules investigated for adsorption in carbon nanostructures, including greenhouse gases, anticancer drugs, and chemical warfare agents, among many more. Keywords: carbon nanostructures; doping; adsorption; transition metals; pollutant gas; nanotubes; Citation: Ramirez-de-Arellano, -

Na‐Doped C70 Fullerene/N‐Doped Graphene/Fe‐Based Quantum Dot
Author Manuscript Title: Na-doped C70 fullerenes/N-doped graphene/Fe-based quantum dots nano- composites for sodium-ion batteries with ultra-high coulombic efficiency Authors: Chunlian Wang; Yang Zhang; Wen He; Xudong Zhang; Zhaoyang Wang; Guihua Yang; Manman Ren; Lianzhou Wang This is the author manuscript accepted for publication and has undergone full peer review but has not been through the copyediting, typesetting, pagination and proofrea- ding process, which may lead to differences between this version and the Version of Record. To be cited as: ChemElectroChem 10.1002/celc.201700899 Link to VoR: https://doi.org/10.1002/celc.201700899 ARTICLE 1 Na-doped C70 fullerenes/N-doped graphene/Fe-based quantum dots nanocomposites 2 for sodium-ion batteries with ultra-high coulombic efficiency 3 Chunlian Wang,[a] Yang Zhang,[b] Wen He,*[ad] Xudong Zhang,*[a] Guihua Yang,[c] Zhaoyang Wang,[a] Manman Ren,[ad] 4 Lianzhou Wang[d] 5 [5-8] 6 Abstract: We fabricate Na-doped C70 fullerenes (Na-C70)/N-doped39 better performance. Carbon materials showing potential using 7 graphene (N-GN)/Fe-based nanocomposites (Na-C70/N-GN/FBNCs)40 in other area, such as carbon silicon composites for 8 with the multiple morphologies via an in situ one step method used41 electromagnetic materials[9], poly/multi-walled carbon nanotubes 9 the multifunction sodium lignosulfonate (SLS) as the structural42 with conductive segregated structure[10], graphene quantum dots 10 template and the main raw material. Fe-based nanoparticles can43 decorated oxide as solar cells[11]. Nanocarbon materials are 11 embed in ordered mesoporous hybrid carbon structure of Na-C7044 recognized as the leading electrode material for commercial 12 and N-GN via spontancous chelation reaction of SLS with iron ions45 LIBs anodes because of their high electrical conductivity, low 13 and carbonization under relatively mild hydrothermal treatment. -
![Arxiv:2108.12221V1 [Physics.Chem-Ph] 27 Aug 2021](https://docslib.b-cdn.net/cover/6044/arxiv-2108-12221v1-physics-chem-ph-27-aug-2021-416044.webp)
Arxiv:2108.12221V1 [Physics.Chem-Ph] 27 Aug 2021
Experimental Determination of the Interaction Potential between a Helium Atom and the Interior Surface of a C60 Fullerene Molecule George Razvan Bacanu,1 Tanzeeha Jafari,2 Mohamed Aouane,3 Jyrki Rantaharju,1 Mark Walkey,1 Gabriela Hoffman,1 Anna Shugai,2 Urmas Nagel,2 Monica Jiménez-Ruiz,3 Anthony J. Horsewill,4 Stéphane Rols,3 Toomas Rõõm,2 Richard J. Whitby,1 and Malcolm H. Levitt1 1)School of Chemistry, University of Southampton, Southampton, SO17 1BJ, UK 2)National Institute of Chemical Physics and Biophysics, Tallinn, 12618, Estonia 3)Institut Laue-Langevin, BP 156, 38042 Grenoble, France 4)School of Physics and Astronomy, University of Nottingham, Nottingham, NG7 2RD (*Electronic mail: [email protected]) (Dated: 30 August 2021) The interactions between atoms and molecules may be described by a potential energy function of the nuclear coordi- nates. Non-bonded interactions are dominated by repulsive forces at short range and attractive dispersion forces at long range. Experimental data on the detailed interaction potentials for non-bonded interatomic and intermolecular forces is scarce. Here we use terahertz spectroscopy and inelastic neutron scattering to determine the potential energy function for the non-bonded interaction between single He atoms and encapsulating C60 fullerene cages, in the helium endo- 3 4 fullerenes He@C60 and He@C60, synthesised by molecular surgery techniques. The experimentally derived potential is compared to estimates from quantum chemistry calculations, and from sums of empirical two-body potentials. 18 21 19 22 I. INTRODUCTION including H2@C60 ,H2@C70 ,H2O@C60 , HF@C60 , 23 CH4@C60 , and their isotopologues. Endofullerenes con- Non-bonded intermolecular interactions determine the taining noble gas atoms, and containing two encapsulated 21,24–30 structure and properties of most forms of matter. -

Fullerene C70 Derivatives Dampen Anaphylaxis and Allergic Asthma Pathogenesis in Mice
Virginia Commonwealth University VCU Scholars Compass Theses and Dissertations Graduate School 2012 Fullerene C70 derivatives dampen anaphylaxis and allergic asthma pathogenesis in mice Sarah Norton Virginia Commonwealth University Follow this and additional works at: https://scholarscompass.vcu.edu/etd Part of the Medicine and Health Sciences Commons © The Author Downloaded from https://scholarscompass.vcu.edu/etd/2759 This Dissertation is brought to you for free and open access by the Graduate School at VCU Scholars Compass. It has been accepted for inclusion in Theses and Dissertations by an authorized administrator of VCU Scholars Compass. For more information, please contact [email protected]. Fullerene C70 derivatives dampen anaphylaxis and allergic asthma pathogenesis in mice A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy at Virginia Commonwealth University By Sarah Brooke Norton B.S., University of Virginia, 2003 M.S., Virginia Commonwealth University, 2007 Director: Daniel H. Conrad, Professor, Microbiology and Immunology Virginia Commonwealth University Richmond, Virginia April, 2012 DEDICATION This dissertation is dedicated to my family for all their encouragement and support throughout his process. My parents, Ronald Kennedy and Penny Kennedy have supported me at every stage of my education and strongly encouraged me to attend graduate school. All my life they have made sacrifices to ensure I was given every opportunity to succeed, and have done everything possible to make this journey easier and celebrated my accomplishments with me. To my aunt, Patricia Petro who sparked my interest in science at a young age and encouraged my studies throughout this journey. To my mother-in-law, Lorraine Norton who has a kind heart and a listening ear and helps me keep everything in perspective. -

A First Experimental Electron Density Study on a C Fullerene
A First Experimental Electron Density Study on a C70 Fullerene: (C70C2F5)10 Roman Kalinowskia, Manuela Webera, Sergey I. Troyanovb, Carsten Paulmannc, and Peter Lugera a Institut f¨ur Chemie und Biochemie /Anorganische Chemie, Freie Universit¨at Berlin, Fabeckstraße 36a, 14195 Berlin, Germany b Department of Chemistry, Moscow State University, 119991 Moscow, Leninskie Gory, Russia c Mineralogisch-Petrologisches Institut, Universit¨at Hamburg, Grindelallee 48, 20146 Hamburg, Germany Reprint requests to Prof. Dr. Peter Luger. Fax: +49-30-838 53464. E-mail: [email protected] Z. Naturforsch. 2010, 65b, 1 – 7; received October 1, 2009 The electron density of the C70 fullerene C70(C2F5)10 was determined from a high-resolution X-ray data set measured with synchrotron radiation (beamline F1 of Hasylab/DESY, Germany) at a temperature of 100 K. With 140 atoms in the asymmetric unit this fullerene belongs to the largest problems examined until now by electron density methods. Using the QTAIM formalism quantitative bond topological and atomic properties have been derived and compared with the results of theoretical calculations on the title compound and on free C70. Key words: Fullerenes, Electron Density, Topological Analysis, Synchrotron Radiation Introduction Although recent technical and methodical advances made experimental electron density (ED) determina- tions possible also on larger molecules, fullerenes with 60 or more atoms are still a major challenge. Their in- vestigation is complicated by the generally poor crystal quality and by the high mobility of these molecules in the crystal structure. Since their discovery in the mid- eighties a considerable number of more than 800 X-ray structures of fullerenes and fullerene derivatives are listed in the Cambridge Data File [1]. -

Polymer Solar Cells Based on Poly(3-Hexylthiophene) and Fullerene: Pyrene Acceptor Systems
Polymer solar cells based on poly(3-hexylthiophene) and fullerene: Pyrene acceptor systems a a a a, * Alessandra Cominetti , Andrea Pellegrino , Luca Longo , Riccardo Po , Alessandra a a a b, 1 Tacca , Chiara Carbonera , Mario Salvalaggio , Michele Baldrighi , Stefano Valdo b Meille a Research Center for Renewable Energies and Environment, Istituto Donegani, Eni S.p.A, Via Fauser 4, IT-28100 Novara, Italy b Dipartimento di Chimica, Materiali e Ingegneria Chimica “G. Natta”, Politecnico di Milano, via Mancinelli 7, IT-20131 Milano, Italy Received 12 May 2014 Received in revised form 30 January 2015 Accepted 27 March 2015 Available online 4 April 2015 1. Introduction The literature on novel materials for polymer solar cells [1e3] deals mainly with the development of the donor components of the active layer [4e9], while much fewer papers are devoted to novel acceptors [10,11]. [6,6]-phenyl-C61-butyric acid methyl ester * Corresponding author. E-mail address: [email protected] (R. Po). 1 Present address: Istituto Italiano di Tecnologia (IIT), via Morego 30, IT-16163 Genova, Italy. (PCBM) is the oldest [12], the most used [13] and, to date, still one filtered, and the solvent was distilled under reduced pressure. 1.03 g of the most effective acceptors for bulk heterojunction polymer (yield 84%) of a pale yellow oil were obtained, which became a solar cells. The popularity of PCBM, aside from its unique electronic white solid after 24 h of storage at þ4 C in a refrigerator. ¼ 1 0 properties, is mainly due to its good solubility in many organic Tm 37.4 C (DSC). -

Molecular Vibrational Modes of C60 and C70 Via Finite Element Method
European Journal of Mechanics A/Solids 28 (2009) 948–954 Contents lists available at ScienceDirect European Journal of Mechanics A/Solids journal homepage: www.elsevier.com/locate/ejmsol Molecular vibrational modes of C60 and C70 via finite element method Du Jing a,b, Zeng Pan a,b,* a Department of Mechanical Engineering, Tsinghua University, Beijing 100084, China b Key Laboratory for Advanced Materials Processing Technology, Ministry of Education of China, China article info abstract Article history: Molecular vibration spectra are of great significance in the study of molecular structures and characters. Received 26 September 2006 A widely-used analytical method of structural mechanics, finite element method, is imported into the Accepted 17 February 2009 domain of micro-scaled molecular spectra. The vibrational modes of fullerenes C60 and C70 are calculated Available online 28 February 2009 depending on uniform carbon–carbon bonding elements, each of which has three force constants that are determined by fitting the C60 Raman spectra experimental data and one ratio factor which is the Keywords: bond lengths ratio between the single and double bonds. The computational result shows reasonable Finite element method agreement with both calculated and experimental results published before. Fullerene Ó 2009 Published by Elsevier Masson SAS. C70 Molecular spectra 1. Introduction overlap (MNDO) (Stanton and Newton, 1988), Quantum-mechan- ical Consistent Force Field Method for Pi-Electron Systems (QCFF/ Since Kroto et al. discovered C60 by laser vaporizing graphite PI) (Negri et al., 1988), Austin Model 1 (AM1) (Slanina et al., 1989), into a helium stream (Kroto et al., 1985); an impressive experi- density functional theory (Adams et al., 1991; Choi et al., 2000; mental and theoretical effort has been undertaken towards this Schettino et al., 2001; Sun and Kertesz, 2002; Schettino et al., 2002). -

Decontamination of Surfaces Exposed to Carbon-Based Nanotubes and Nanomaterials
Hindawi Publishing Corporation Journal of Nanomaterials Volume 2014, Article ID 249603, 9 pages http://dx.doi.org/10.1155/2014/249603 Research Article Decontamination of Surfaces Exposed to Carbon-Based Nanotubes and Nanomaterials Paul Su,1 Babak Haghpanah,2 William W. Doerr,1 Zahra Karimi,2 Syed Hassan,2 Louis Gritzo,1 Ahmed A. Busnaina,2 and Ashkan Vaziri2 1 FMGlobal,Norwood,MA02062,USA 2 DepartmentofMechanicalandIndustrialEngineering,NortheasternUniversity,Boston,MA02115,USA Correspondence should be addressed to Paul Su; [email protected] Received 10 October 2013; Accepted 12 December 2013; Published 16 January 2014 Academic Editor: Aiying Wang Copyright © 2014 Paul Su et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Contamination of surfaces by nanomaterials can happen due to accidental spillage and release or gradual accumulation during processing or handling. Considering the increasingly wide use of nanomaterials in industry and research labs and also taking into account the diversity of physical and chemical properties of different nanomaterials (such as solubility, aggregation/agglomeration, and surface reactivity), there is a pressing need to define reliable nanomaterial-specific decontamination guidelines. In this paper, we propose and investigate a potential method for surface decontamination of carbon-based nanomaterials using solvent cleaning and wipes. The results show that the removal efficiency for single- and multiwalled carbon nanotubes from silicon wafers sprayed with water-surfactant solutions prior to mechanical wiping is greater than 90% and 95%, respectively. The need for further studies to understand the mechanisms of nanomaterial removal from surfaces and development of standard techniques for surface decontamination of nanomaterials is highlighted. -

Molecules in Benzene Solutions Prepared by Various Methods
Bulletin of National University of Uzbekistan: Mathematics and Natural Sciences Volume 1 Issue 2 Article 4 9-25-2018 Analysis of clusterization of C70 molecules in benzene solutions prepared by various methods U. Makhmanov Tashkent State Technical University O. Ismailova Tashkent State Technical University A. Kokhkharov Tashkent State Technical University S. Bakhramov Tashkent State Technical University Follow this and additional works at: https://uzjournals.edu.uz/mns_nuu Part of the Physics Commons Recommended Citation Makhmanov, U.; Ismailova, O.; Kokhkharov, A.; and Bakhramov, S. (2018) "Analysis of clusterization of C70 molecules in benzene solutions prepared by various methods," Bulletin of National University of Uzbekistan: Mathematics and Natural Sciences: Vol. 1 : Iss. 2 , Article 4. Available at: https://uzjournals.edu.uz/mns_nuu/vol1/iss2/4 This Article is brought to you for free and open access by 2030 Uzbekistan Research Online. It has been accepted for inclusion in Bulletin of National University of Uzbekistan: Mathematics and Natural Sciences by an authorized editor of 2030 Uzbekistan Research Online. For more information, please contact [email protected]. Volume 1, Issue 2 (2018) ANALYSIS OF CLUSTERIZATION OF C70 MOLECULES IN BENZENE SOLUTIONS PREPARED BY VARIOUS METHODS Makhmanov U. K., Ismailova O. B., Kokhkharov A. M., Bakhramov S. A. Tashkent State Technical University, Tashkent, Uzbekistan Abstract Self-assembling features of molecules of C70 in benzene solution prepared in two various ways – equilibrium and non-equilibrium – has been investigated ex- perimentally by method of high-resolution transmission electron microscopy. It was demonstrated, that the formation of densely packed monomolecular fullerene aggregates with a diameter of not more than 30 nm in solutions pre- pared by the equilibrium method (without the use of external mechanical in- fluences on solution). -
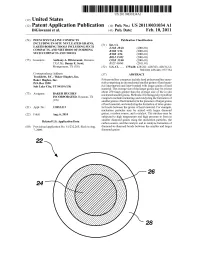
Us 2011/0031034 A1
Illll Illlllll II Illlll Illll Illll IllllUS Illll20110031034A1 Illll Illll Illll Illll Illll Illll Illlll Illl Illl Illl (19) nited States (12) Patent Application Publication (10) Pub. No.: S 2011/0031034 A1 DiGiovanni et al. (43) Pub. Date: Feb. 10, 2011 (54) POLYCRYSTALLINE COMPACTS Publication Classification INCLUDING IN-SITU NUCLEATED GRAINS, (51) Int. (71. EARTH-BORING TOOLS INCLUDING SUCH E21B 10/46 (2006.01) COMPACTS, AND METHODS OF FORMING B32B 5/16 (2006.01) SUCH COMPACTS AND TOOLS B32B 3/26 (2006.01) BO1J 35/02 (2006.01) (75) Inventors: Anthony A. DiGiovanni, Houston, CO1B 31/06 (2006.01) TX (US); Danny E. Scott, B82 Y 40/O0 (2011.01) Montgomery, TX (US) (52) U.S. CI ....... 175/428; 428/221; 428/323; 428/312.2; 502/100; 423/446; 977/734 Correspondence Address: (57) ABSTRACT Traskbritt, P.C. / Baker Hughes, Inc. Baker Hughes, Inc. Polycrystalline compacts include hard polycrystalline mate- P.O. Box 2550 rials comprising in situ nucleated smaller grains of hard mate- Salt Lake City, UT 84110 (US) rial interspersed and inter-bonded with larger grains of hard material. The average size of the larger grains may be at least about 250 times greater than the average size of the in situ Assignee: BAKER HUGHES (73) nucleated smaller grains. Methods of forming polycrystalline INCORPORATED, Houston, TX compacts include nucleating and catalyzing the formation of (US) smaller grains of hard material in the presence of larger grains of hard material, and catalyzing the formation of inter-granu- (21) Appl. No.: 12/852,313 lar bonds between the grains of hard material. -

Ep 2 098 549 A1
(19) & (11) EP 2 098 549 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 153(4) EPC (43) Date of publication: (51) Int Cl.: 09.09.2009 Bulletin 2009/37 C08G 61/00 (2006.01) C01B 31/02 (2006.01) C09D 201/02 (2006.01) H01L 51/30 (2006.01) (2006.01) (21) Application number: 07860265.3 H01L 51/42 (22) Date of filing: 26.12.2007 (86) International application number: PCT/JP2007/075036 (87) International publication number: WO 2008/081845 (10.07.2008 Gazette 2008/28) (84) Designated Contracting States: (72) Inventors: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR • TANAKA, Katsutomo HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE Kitakyusyu-shi SI SK TR Fukuoka 806-0004 (JP) • TAKAGI, Masatoshi (30) Priority: 27.12.2006 JP 2006351751 Yokohama-shi Kanagawa 227-8502 (JP) (71) Applicants: • Frontier Carbon Corporation (74) Representative: Vossius & Partner Fukuoka 806-0004 (JP) Siebertstraße 4 • Mitsubishi Chemical Corporation 81675 München (DE) Tokyo 108-0014 (JP) (54) FULLERENE FILM USING FULLERENE DERIVATIVE AS RAW MATERIAL, FULLERENE POLYMER AND THEIR PRODUCTION METHODS (57) A fullerene film and a fullerene polymer both produced from a fullerene derivative are provided which can be easily formed by a wet process and can retain the intact properties inherent in the fullerene. Also provided are processes for producing the film and polymer. A solution of a fullerene derivative decomposing at a temperature lower than the pyrolysis temperature of the fullerene, e.g., the fullerene derivative represented by the following formula, is applied to a substrate.