In Vitro Efficacy of Flomoxef Against Extended-Spectrum Beta
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

Severe Sepsis Tipsheet
Severe Sepsis Tipsheet SEVERE SEPSIS A, B, and C of the following must be met within 6 hours of each other. A. Documentation of a suspected infection. There may be reference to “possible infection,” “suspect infection,” “rule out infection,” or similar documentation. B. TWO or more manifestations of systemic infection according to the Systemic Inflammatory Response Syndrome (SIRS) criteria, which are: • Temperature > 38.3 C or < 36.0 C (> 100.9 F or < 96.8 F) • Heart rate (pulse) > 90 • Respiration > 20 per minute • White blood cell count > 12,000 or < 4,000 or > 10% bands C. Organ dysfunction, evidenced by any one of the following: • Systolic blood pressure (SBP) < 90 mmHg, or mean arterial pressure < 65 mmHg, or a systolic blood pressure decrease of more than 40 mmHg. Physician/APN/PA documentation must be present in the medical record indicating a > 40 mmHg decrease in SBP has occurred and is related to infection, severe sepsis or septic shock and not other causes. • Acute respiratory failure as evidenced by a new need for invasive or non-invasive mechanical ventilation. To use acute respiratory failure as a sign of organ dysfunction there must be: • Documentation of acute respiratory failure AND • Documentation the patient is on mechanical ventilation • Invasive mechanical ventilation requires an endotracheal or tracheostomy tube. Non-invasive mechanical ventilation may be referred to as BiPAP or CPAP. • New need for mechanical ventilation indicates the patient was not using the same type of mechanical ventilation prior to the current acute respiratory failure. • Use the time at which there is documentation the patient has both acute respiratory failure and is on mechanical ventilation. -

Antimicrobial Resistance Benchmark 2020 Antimicrobial Resistance Benchmark 2020
First independent framework for assessing pharmaceutical company action Antimicrobial Resistance Benchmark 2020 Antimicrobial Resistance Benchmark 2020 ACKNOWLEDGEMENTS The Access to Medicine Foundation would like to thank the following people and organisations for their contributions to this report.1 FUNDERS The Antimicrobial Resistance Benchmark research programme is made possible with financial support from UK AID and the Dutch Ministry of Health, Welfare and Sport. Expert Review Committee Research Team Reviewers Hans Hogerzeil - Chair Gabrielle Breugelmans Christine Årdal Gregory Frank Fatema Rafiqi Karen Gallant Nina Grundmann Adrián Alonso Ruiz Hans Hogerzeil Magdalena Kettis Ruth Baron Hitesh Hurkchand Joakim Larsson Dulce Calçada Joakim Larsson Marc Mendelson Moska Hellamand Marc Mendelson Margareth Ndomondo-Sigonda Kevin Outterson Katarina Nedog Sarah Paulin (Observer) Editorial Team Andrew Singer Anna Massey Deirdre Cogan ACCESS TO MEDICINE FOUNDATION Rachel Jones The Access to Medicine Foundation is an independent Emma Ross non-profit organisation based in the Netherlands. It aims to advance access to medicine in low- and middle-income Additional contributors countries by stimulating and guiding the pharmaceutical Thomas Collin-Lefebvre industry to play a greater role in improving access to Alex Kong medicine. Nestor Papanikolaou Address Contact Naritaweg 227-A For more information about this publication, please contact 1043 CB, Amsterdam Jayasree K. Iyer, Executive Director The Netherlands [email protected] +31 (0) 20 215 35 35 www.amrbenchmark.org 1 This acknowledgement is not intended to imply that the individuals and institutions referred to above endorse About the cover: Young woman from the Antimicrobial Resistance Benchmark methodology, Brazil, where 40%-60% of infections are analyses or results. -

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

The Efficacy of Cefmetazole Against Pyelonephritis Caused by Extended
International Journal of Infectious Diseases 17 (2013) e159–e163 Contents lists available at SciVerse ScienceDirect International Journal of Infectious Diseases jou rnal homepage: www.elsevier.com/locate/ijid The efficacy of cefmetazole against pyelonephritis caused by extended-spectrum beta-lactamase-producing Enterobacteriaceae a, b c d a,e Asako Doi *, Toshihiko Shimada , Sohei Harada , Kentaro Iwata , Toru Kamiya a Department of Infectious Diseases, Rakuwakai Otowa Hospital, Otowachinji-cho 2, Yamashina-ku, Kyoto, Japan b Department of General Internal Medicine, Nara City Hospital, Nara, Japan c Department of Microbiology and Infectious Diseases, Toho University School of Medicine, Ota-ku, Tokyo, Japan d Division of Infectious Diseases, Kobe University Hospital, Chuo-ku, Kobe, Hyogo, Japan e Department of General Internal Medicine, Rakuwakai Otowa Hospital, Otowachinji-cho 2, Yamashina-ku, Kyoto, Japan A R T I C L E I N F O S U M M A R Y Article history: Objectives: Urinary tract infections (UTIs) caused by extended-spectrum beta-lactamase (ESBL)- Received 3 April 2012 producing Enterobacteriaceae are on the increase. Although cefmetazole is stable in vitro against the Accepted 26 September 2012 hydrolyzing activity of ESBLs, no clinical study has ever evaluated its role in infections caused by these Corresponding Editor: William Cameron, organisms. We therefore evaluated the efficacy of cefmetazole compared to carbapenems against Ottawa, Canada pyelonephritis caused by ESBL-producing Enterobacteriaceae. Methods: A retrospective chart review was conducted at a tertiary care hospital from August 2008 to July Keywords: 2010. Chart reviews were done for patients with ESBL-producing organisms in urine identified in the Extended-spectrum beta-lactamase (ESBL) microbiology database. -
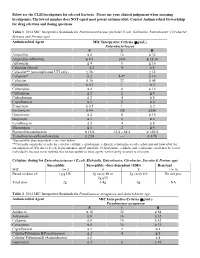
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

Yungjin-Pharm-Co-Ltd-Party-Content
Name Yungjin Pharm. Co., Ltd. Foundation •1952 (Public opened in 1973) • 2004 (Merged with KT&G, Korean Tomorrow and Global) Representatives Byung-Hwan Ryoo / President, CEO Location 451-20, Cheonho-3dong, Gangdong-gu, Seoul, Korea Core Business •Manufacturing Pharmaceutical goods (API and Finished Products) •Sales in Domestic and Overseas Markets Employees 538 (Headquarter : 48, Factory : 198, R&D Lab. : 45, MR : 247) Key Facilities Head office (Seoul), 3 Factories (API, Finished Dosage, Drinks) 13 Branches, 1 Central Research Lab. (Synthesis, Formulation) Annual Sales KRW 101 Billion (USD 101 Million), as of Dec. 2011 2 Corporate Profile Since its establishment in 1952, Yungjin Pharm. Co., Ltd has been enjoying the privilege of playing a major role as a forerunner in the Korean pharmaceutical industry for over half a century. In accordance with our inspiring mission statement, “Relieve the suffering mankind from disease with effective and safe pharmaceutical products on our own” we have been contributing to improve lives of human beings for the better. In order to establish a solid foundation, Yungjin Pharm. Co., Ltd. joined the KT&G as a member group on March 29, 2004. This merge enables us to reform ourselves as a company of enduring strength with sufficient resources for R&D sector and further development. Based on our long-term goal, we strive for further growth and expansion. Strong alliance with the worldwide pharmaceutical companies allows us to expand our market into the worldwide and cope with the challenging environment of global pharmaceutical industry. Through active involvement in R&D activities, quality management and overseas business, we will fulfill the expectations of our customers and shareholders. -

Title Antimicrobial Therapy for Acute Cholecystitis: Tokyo Guidelines
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by HKU Scholars Hub Title Antimicrobial therapy for acute cholecystitis: Tokyo Guidelines Yoshida, M; Takada, T; Kawarada, Y; Tanaka, A; Nimura, Y; Gomi, H; Hirota, M; Miura, F; Wada, K; Mayumi, T; Solomkin, JS; Author(s) Strasberg, S; Pitt, HA; Belghiti, J; de Santibanes, E; Fan, ST; Chen, MF; Belli, G; Hilvano, SC; Kim, SW; Ker, CG Journal Of Hepato-Biliary-Pancreatic Surgery, 2007, v. 14 n. 1, p. Citation 83-90 Issued Date 2007 URL http://hdl.handle.net/10722/84203 Rights J Hepatobiliary Pancreat Surg (2007) 14:83–90 DOI 10.1007/s00534-006-1160-y Antimicrobial therapy for acute cholecystitis: Tokyo Guidelines Masahiro Yoshida1, Tadahiro Takada1, Yoshifumi Kawarada2, Atsushi Tanaka3, Yuji Nimura4, Harumi Gomi5, Masahiko Hirota6, Fumihiko Miura1, Keita Wada1, Toshihiko Mayumi7, Joseph S. Solomkin8, Steven Strasberg9, Henry A. Pitt10, Jacques Belghiti11, Eduardo de Santibanes12, Sheung-Tat Fan13, Miin-Fu Chen14, Giulio Belli15, Serafin C. Hilvano16, Sun-Whe Kim17, and Chen-Guo Ker18 1 Department of Surgery, Teikyo University School of Medicine, 2-11-1 Kaga, Itabashi-ku, Tokyo 173-8605, Japan 2 Mie University School of Medicine, Mie, Japan 3 Department of Medicine, Teikyo University School of Medicine, Tokyo, Japan 4 Division of Surgical Oncology, Department of Surgery, Nagoya University Graduate School of Medicine, Nagoya, Japan 5 Division of Infection Control and Prevention, Jichi Medical University Hospital, Tochigi, Japan 6 Department of Gastroenterological -

Consideration of Antibacterial Medicines As Part Of
Consideration of antibacterial medicines as part of the revisions to 2019 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) Section 6.2 Antibacterials including Access, Watch and Reserve Lists of antibiotics This summary has been prepared by the Health Technologies and Pharmaceuticals (HTP) programme at the WHO Regional Office for Europe. It is intended to communicate changes to the 2019 WHO Model List of Essential Medicines for adults (EML) and Model List of Essential Medicines for children (EMLc) to national counterparts involved in the evidence-based selection of medicines for inclusion in national essential medicines lists (NEMLs), lists of medicines for inclusion in reimbursement programs, and medicine formularies for use in primary, secondary and tertiary care. This document does not replace the full report of the WHO Expert Committee on Selection and Use of Essential Medicines (see The selection and use of essential medicines: report of the WHO Expert Committee on Selection and Use of Essential Medicines, 2019 (including the 21st WHO Model List of Essential Medicines and the 7th WHO Model List of Essential Medicines for Children). Geneva: World Health Organization; 2019 (WHO Technical Report Series, No. 1021). Licence: CC BY-NC-SA 3.0 IGO: https://apps.who.int/iris/bitstream/handle/10665/330668/9789241210300-eng.pdf?ua=1) and Corrigenda (March 2020) – TRS1021 (https://www.who.int/medicines/publications/essentialmedicines/TRS1021_corrigenda_March2020. pdf?ua=1). Executive summary of the report: https://apps.who.int/iris/bitstream/handle/10665/325773/WHO- MVP-EMP-IAU-2019.05-eng.pdf?ua=1. -

WO 2010/025328 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 4 March 2010 (04.03.2010) WO 2010/025328 Al (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every A61K 31/00 (2006.01) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, (21) International Application Number: CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, PCT/US2009/055306 DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (22) International Filing Date: HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, 28 August 2009 (28.08.2009) KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, (25) Filing Language: English NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (26) Publication Language: English SE, SG, SK, SL, SM, ST, SV, SY, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: 61/092,497 28 August 2008 (28.08.2008) US (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (71) Applicant (for all designated States except US): FOR¬ GM, KE, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, ZM, EST LABORATORIES HOLDINGS LIMITED [IE/ ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, —]; 18 Parliament Street, Milner House, Hamilton, TM), European (AT, BE, BG, CH, CY, CZ, DE, DK, EE, Bermuda HM12 (BM). -

Management of Penicillin and Beta-Lactam Allergy
Management of Penicillin and Beta-Lactam Allergy (NB Provincial Health Authorities Anti-Infective Stewardship Committee, September 2017) Key Points • Beta-lactams are generally safe; allergic and adverse drug reactions are over diagnosed and over reported • Nonpruritic, nonurticarial rashes occur in up to 10% of patients receiving penicillins. These rashes are usually not allergic and are not a contraindication to the use of a different beta-lactam • The frequently cited risk of 8 to 10% cross-reactivity between penicillins and cephalosporins is an overestimate based on studies from the 1970’s that are now considered flawed • Expect new intolerances (i.e. any allergy or adverse reaction reported in a drug allergy field) to be reported after 0.5 to 4% of all antimicrobial courses depending on the gender and specific antimicrobial. Expect a higher incidence of new intolerances in patients with three or more prior medication intolerances1 • For type-1 immediate hypersensitivity reactions (IgE-mediated), cross-reactivity among penicillins (table 1) is expected due to similar core structure and/or major/minor antigenic determinants, use not recommended without desensitization • For type-1 immediate hypersensitivity reactions, cross-reactivity between penicillins (table 1) and cephalosporins is due to similarities in the side chains; risk of cross-reactivity will only be significant between penicillins and cephalosporins with similar side chains • Only type-1 immediate hypersensitivity to a penicillin manifesting as anaphylaxis, bronchospasm, -

Antibiotic Use for Sepsis in Neonates and Children: 2016 Evidence Update
Antibiotic Use for Sepsis in Neonates and Children: 2016 Evidence Update Aline Fuchsa, Julia Bielickia,b, Shrey Mathurb, Mike Sharlandb, Johannes N. Van Den Ankera,c a Paediatric Pharmacology and Pharmacometrics, University Children's Hospital Basel, Basel, Switzerland b Paediatric Infectious Disease Research Group, Institute for Infection and Immunity, St George's University of London, London, United Kingdom c Division of Clinical Pharmacology, Children’s National Health System, Washington, DC, USA WHO-Reviews 1 TABLE OF CONTENTS 1. INTRODUCTION ............................................................................................................................... 3 1.1. Aims ......................................................................................................................................... 3 1.2. Background ............................................................................................................................. 3 1.2.1. Definition and diagnosis ................................................................................................. 3 Neonatal Sepsis ............................................................................................................................... 3 Paediatric Sepsis ............................................................................................................................. 4 Community versus hospital acquired sepsis .................................................................................. 5 1.2.2. Microbiology ..................................................................................................................