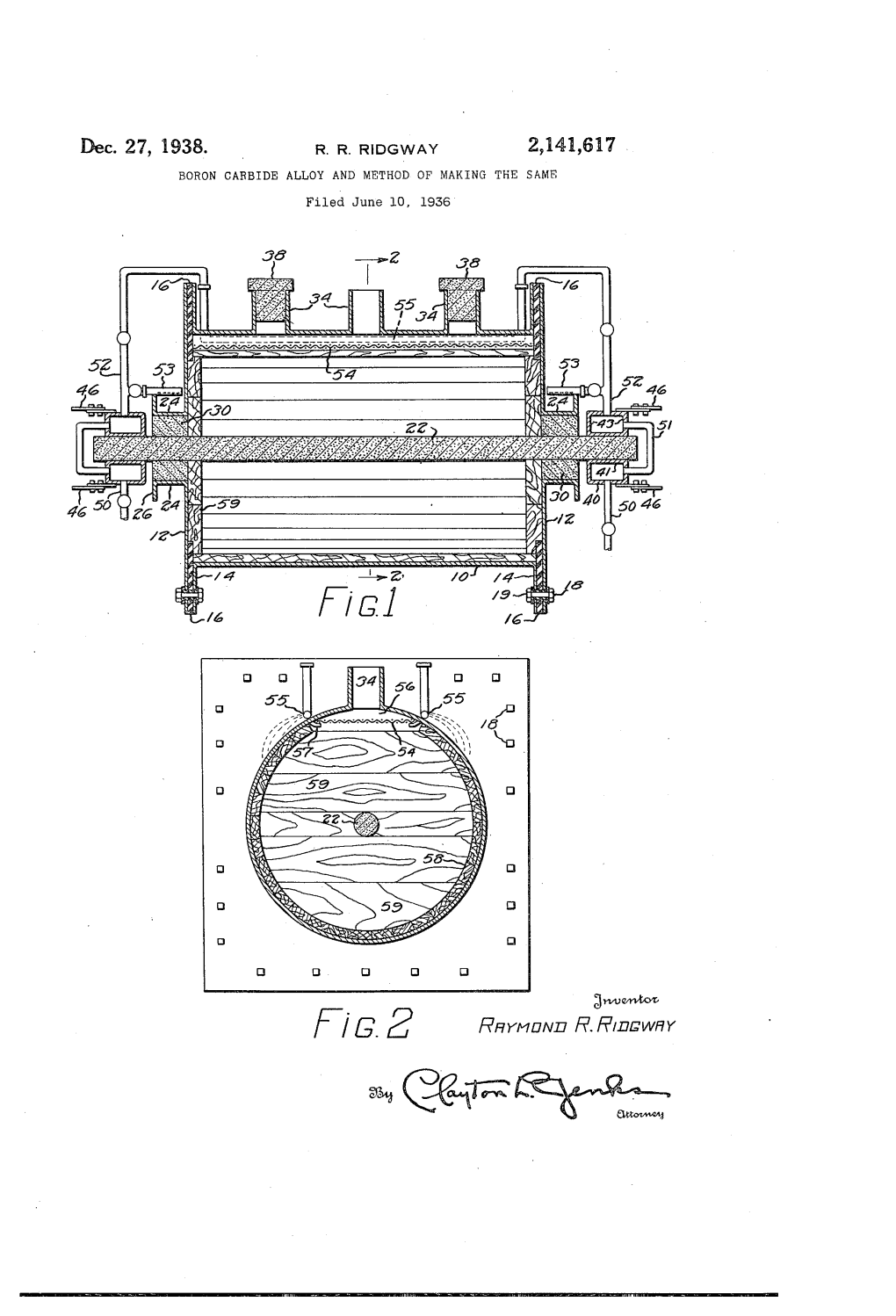
R G ,2 Rhymu/Vu RR/DEWHY Patented Dec
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

United States Patent (19) 11 3,929,495 Broemer Et Al
United States Patent (19) 11 3,929,495 Broemer et al. (45) Dec. 30, 1975 54) OPTICAL BORATE GLASS OF HIGH 3,149,984 9/1964 Faulstich........................... 106/47 R CHEMICAL RESISTANCE AND PROCESS 3,307,929 3/1967 Trap.................................. 106/47 R 3,480,453 it 1/1969 Reid et al.......................... 106/47 R OF MAKING SAME 3,486,915 12/1969 Broemer et al................... 106/47 R (75) Inventors: Heinz Broemer, Hermannstein; 3,510,325 5/1970 Broemer et al................... 106/47 R Norbert Meinert, Wetzlar, both of FOREIGN PATENTS OR APPLICATIONS Germany 863,352 3/1961 United Kingdom............... 106/47 Q 73 Assignee: Ernst Leitz G.m.b.H., Wetzlar, 4,424,420 10/1969 Japan................................ 106/47 Q Germany Filed: May 15, 1973 Primary Examiner-Winston A. Douglas 22) Assistant Examiner-Mark Bell 21 Appl. No.: 360,418 Attorney, Agent, or Firm-Erich M. H. Radde 30 Foreign Application Priority Data 57 ABSTRACT May 15, 1972 Germany............................ 2223564 An optical borate glass of high chemical resistance, with negative anomalous partial dispersion, refraction 52) U.S. Cl. ............................. 106/47 Q; 106/47 R index n between 1,65 and 1,79, and Abbe number ve 51 Int. CI..... C03C 3/14: CO3C 3/00; C03C 3/30 between 40 and 30 is composed of boron trioxide, 58) Field of Search......................... 106/47 O, 47 R lead oxide, and aluminum oxide. It may additionally contain lithium, sodium, and/or potassium oxides, zinc (56) References Cited oxide, zirconium dioxide, tantalum pentoxide, and, if UNITED STATES PATENTS desired, antimony trioxide and/or bismuthum trioxide. -

United States Patent (19) (11) 4,154,806 Szabó Et Al
United States Patent (19) (11) 4,154,806 Szabó et al. 45) May 15, 1979 54 PROCESS FOR THE PRODUCTION OF (56) References Cited NITROUS OXDE U.S. PATENT DOCUMENTS 75) Inventors: Zoltán Szabó; Jenö Trompler; 1,098,305 5/1914 Torley et al. ........................ 423/400 Erzsébet Hollós, née Rakosinyi, all of 2,111,277 3/1938 Castner et al. ....................... 423/400 Budapest, Hungary 2,425,582 8/1947 Vingee ................................. 423/400 3,411,883 11/1968 Smit ................................. 423/400X 73) Assignee: Eötvös Lóránd Tudományegyetem, FOREIGN PATENT DOCUMENTS Budapest, Hungary 276069 5/1913 Fed. Rep. of Germany ........... 423/400 (21) Appl. No.: 865,119 Primary Examiner-G. O. Peters 57 ABSTRACT (22 Filed: Dec. 28, 1977 Ammonium nitrate is mixed, in a weight ratio of higher than 1:5, with a melt containing ammonium hydrogen 30 Foreign Application Priority Data sulfate and ammonium sulfate respectively present in Dec. 30, 1976 HU) Hungary ............................. TO 1047 the melt in a ratio of at least 4:1. The resultant mixture is subjected to thermal decomposition at 200-240 de 51) Int. C.’.............................................. C01B 21/22 grees C to produce nitrous oxide. 52) U.S. C. .................................................... 423/400 58) Field of Search ......................................... 423/400 10 Claims, No Drawings 4,154,806 2 temperature, i.e. over 250 C., the nitrous oxide formed PROCESS FOR THE PRODUCTION OF NITROUS decomposes to higher nitrogen oxides and nitrogen. A OXDE further disadvantage of the increased temperature con sists in that ammonium nitrate sublimates and thus The invention concerns a process for the production causes material loss and operational problems. -

Effects of B2O3 (Boron Trioxide) on Colon Cancer Cells: Our First-Step Experience and in Vitro Results
Turkish Journal of Biology Turk J Biol (2019) 43: 209-223 http://journals.tubitak.gov.tr/biology/ © TÜBİTAK Research Article doi:10.3906/biy-1901-34 Effects of B2O3 (boron trioxide) on colon cancer cells: our first-step experience and in vitro results 1, 2 3 4 5 3 Özgür ALBUZ *, Dilek DÜLGER , Beste Çağdaş TUNALI , Feray AYDIN , Selim YALÇIN , Mustafa TÜRK 1 Department of General Surgery, Keçiören Training and Research Hospital, Ankara, Turkey 2 Department of Medical Microbiology of Basic Medical Sciences, Faculty of Medicine, Karabük University, Karabük, Turkey 3 Department of Bioengineering, Faculty of Engineering, Kırıkkale University, Kırıkkale, Turkey 4 Department of General Surgery, 29 Mayıs State Hospital, Ankara, Turkey 5 Department of Oncology, Faculty of Medicine, Kırıkkale University, Kırıkkale, Turkey Received: 15.01.2019 Accepted/Published Online: 11.06.2019 Final Version: 13.06.2019 Abstract: Boron oxide (B2O3) is derived from dehydration of boric acid and is a colorless, semitransparent, crystalline compound that is moderately soluble in water. On the other hand, boron oxide is chemically hygroscopic. This gives the molecule the ability to soak up water and adhere to tissues. Boron oxide can be used locally after tumor debulking in inoperable tumors and especially when the tumor-free margin distance cannot be provided. For all these reasons we aimed to evaluate the in vitro test results of B2O3 in terms of cytotoxicity, genotoxicity, apoptosis, and necrotic effects on L929 fibroblast cells and DLD-1 colorectal adenocarcinoma cells. Our studies demonstrated that boron oxide compounds appear to be highly cytotoxic for both cell lines according to WST cell viability assay (44.22% and 18.36% on DLD-1 and L929, respectively). -

Improving Boron for Combustion Applications
New Jersey Institute of Technology Digital Commons @ NJIT Dissertations Electronic Theses and Dissertations Summer 8-31-2019 Improving boron for combustion applications Kerri-lee Annique Chintersingh New Jersey Institute of Technology Follow this and additional works at: https://digitalcommons.njit.edu/dissertations Part of the Aerospace Engineering Commons, Chemical Engineering Commons, and the Materials Science and Engineering Commons Recommended Citation Chintersingh, Kerri-lee Annique, "Improving boron for combustion applications" (2019). Dissertations. 1420. https://digitalcommons.njit.edu/dissertations/1420 This Dissertation is brought to you for free and open access by the Electronic Theses and Dissertations at Digital Commons @ NJIT. It has been accepted for inclusion in Dissertations by an authorized administrator of Digital Commons @ NJIT. For more information, please contact [email protected]. Copyright Warning & Restrictions The copyright law of the United States (Title 17, United States Code) governs the making of photocopies or other reproductions of copyrighted material. Under certain conditions specified in the law, libraries and archives are authorized to furnish a photocopy or other reproduction. One of these specified conditions is that the photocopy or reproduction is not to be “used for any purpose other than private study, scholarship, or research.” If a, user makes a request for, or later uses, a photocopy or reproduction for purposes in excess of “fair use” that user may be liable for copyright infringement, -

Position Paper of the European Glass Industry on the Proposed Inclusion of Boron Trioxide on the Candidate List for Authorization
Position Paper of the European Glass Industry on the Proposed Inclusion of Boron Trioxide on the candidate list for authorization April 2012 Glass manufacturers support the REACH Regulation and believe that a reduction of the risk associated with worker exposure and the environmental impact of dangerous substances is welcome in the EU Market. Glass Alliance Europe welcomes the opportunity to provide its contribution to the public consultation on the inclusion of boron trioxide in the candidate list for authorization of the REACH Regulation. CONTRIBUTION TO THE CONSULTATION PROCESS Germany submitted to ECHA an Annex XV dossiers for the identification of boron trioxide as a Substance of Very High Concern to be included in the candidate list for authorization. The rationale for this identification is the classification of boron trioxide as toxic for reproduction category 1B in Regulation (EC) No 1272/2008, as amended and adapted to technical and scientific progress by Regulation (EC) No 790/2009.. Notwithstanding the rationale being perfectly in line with the REACH Regulation, the glass industries believe that the introduction of boron trioxide on the candidate list for authorization could lead to future misunderstanding. The glass industries do not want to engage in any discussion on the right classification of boron trioxide, which is outside of our competence. However, it is very well known that there are still a lot of discussions going on at EU and International level on the correct classification of boron trioxide. For example Poland has indicated its intention to submit borate reclassification dossiers under CLP. This is based on their review of the data arising from new studies on worker exposure (in China and Turkey) and concluded that a reclassification of these substances is warranted. -

United States Patent (19) - (11) 4,178,317 A
United States Patent (19) - (11) 4,178,317 A. Horn et al. 45 Dec. 11, 1979 54 MANUFACTURE OF OLEFINICALLY (56) References Cited UNSATURATED ALPHATIC OR CYCLOALIPHATIC HYDROCARBONS U.S. PATENT DDOCUME MENT S 4,024,171 5/1977 McArthur ....................... 260/666A 75) Inventors: Peter Horn, Hirschberg; Otto-Alfred FOREIGN PATENT DOCUMENTS Grosskinsky; Hugo Fuchs, both of Ludwigshafen, all of Fed. Rep. of 943745 12/1963 United Kingdom. Germany Primary Examiner-Veronica O'Keefe w Attorney, Agent, or Firm-Keil & Witherspoon 73) Assignee: BSAngelschall, Fed. Rep. 57 ABSTRACT Olefinically unsaturated aliphatic or cycloaliphatic hy 21) Appl. No.: 907,678 drocarbons are manufactured by bringing alkanecar boxylic acids of 3 to 20 carbon atoms, alkanedicarboxy ilar. lic acids of 4 to 20 carbon atoms or 5-membered or (22) Filed: May 19, 1978 6-membered cycloalkanecarboxylic acids or their alkyl, O 9 cycloalkyl, aralkyl or phenyl esters, in the gas phase, at 30. Foreign Application Priority Data from 250 to 800 C., into contact with catalysts in Jun. 10, 1977 (DE) Fed. Rep. of Germany. 2726106 which the active composition consists of boron trioxide, boric acid and/or boron nitride, together with one or 51) Int. C.’................................................ C07C3/00 more of the oxides of aluminum, silicon, tin, lead, tita 52 U.S. C. .................................... 585/357; 585/638; nium and zirconium. 585/640 (58) Field of Search .................................... 260/666 A 9 Claims, No Drawings 4,178,317 1. 2 ahydrobenzoate, cyclohexyl -

United States Patent 15 3,650,778 Dumesnil Et Al
United States Patent 15 3,650,778 Dumesnil et al. (45) Mar. 21, 1972 (54) LOW-EXPANSION, LOW-MELTING 3,520,831 7/1970 Trap et al............................. 106/47 X ZINCPHOSPHOVANADATE GLASS 3,393,060 7/1968 Blair et al................................ 106/47 COMPOSITIONS 3,534,209 10, 1970 Anderson et al......................... 106/47 (72) Inventors: Maurice E. Dumesnil, Palo Alto; Robert R. OTHER PUBLICATIONS Hewitt, San Jose; Joseph L. Bozarth, Grossman et al., J. AM Cer. Soc. Vol. 47, (Sept., 1964) “Zinc Mountain View, all of Calif. Borophosphate Glass' page 471 (73) Assignee: Fairchild Camera and Instrument Cor Primary Examiner-Daniel E. Wyman poration, Syosset, Long Island, N.Y. Assistant Examiner-W. R. Satterfield 22) Fied: Oct. 29, 1969 Attorney-Roger S. Borovoy and Alan H. Macpherson (21) Appl. No.: 870,620 57) ABSTRACT Low-melting, low-expansion, lead-free glass compositions are 52) U.S.C. ............................... 106/47 R, 106/46, 117/125 provided for sealing ceramics parts and encapsulating ceramic 51 Int. Cl............................................................ C03c.3/00 substrates. In addition to having a thermal expansion matching 58 Field of Search................ 106/47, 48, 46; 1611196, 192; that of alumina, the glass compositions provide for the forma 252/519; 65/43; 117/125 tion of a glass-to-alumina seal in the 380 to 450° C. tempera ture range. 56) References Cited 1 Claims, 3 Drawing Figures UNITED STATES PATENTS 3,446,695 5/1969 Janakirama-Rao......................106/47 7 8 4 22 >- "l-D 5 6 2 IO Patented March 21, 1972 3,650,778 4 O F. G.2 NVENTORS O MAURCE EDUMESN ROBERT R HEWITT 0 00 200300 400 500 JOSEPH L BOZARTH TEMPERATURE OC BY %a 4 ATTORNEY 3,650,778 1. -

Rohs/REACH Compliance
Page 1 of 2 RoHS/REACH Compliance International Manufacturing Services, Inc. (IMS) recognizes and supports the worldwide effort for environmental protection and conservation. REACH Regulation REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) is a European Union Commission (EUC) Regulation on chemicals and their safe use (EC 1907/2006) entered into force on 01 June, 2007 and will be phased in until 2018. • REACH applies to all imports to the EU • There are three levels of interest defined by REACH: o Substances: Chemical elements and their compounds in the natural state, identifiable by CAS or EINIC number (Ex: metal) o Preparations: A combination or mixture of two or more substances (Ex.: ink, adhesive) o Article: An object whose special shape, surface or design determines its function to a greater degree than does its chemical composition; assembled products (Ex: cars, electronic components) • The REACH regulation focuses on the control of Substances of Very High Concern (SVHCs) published in accordance with Article 59 of the REACH regulation • SVHCs are relevant to Substances and Preparations, not Articles • Registration of SVHCs is required for Substances and Preparations, not Articles • Notification and communication is required for Articles under the following conditions: o The Article has an “intended release” of SVHC o SVHC content > 0.1% of Total Article Weight Products manufactured and supplied by IMS are “articles” as defined by the REACH regulation and do not release substances under their normal use. The diboron trioxide and lead oxide present in certain products supplied by IMS are an element of the glass material. The glass is a UVCB substance (substance of unknown or variable composition, Complex reaction products or biological materials) under the REACH regulation. -

DENTSPLY International PROSTHETICS
DENTSPLY International PROSTHETICS Safety Data Sheet Safety Data Sheet (conforms to with Regulation (EC) Date Issued: 31 August 2016 1907/2006, Regulation (EC) 1272/2008 and Regulation Document Number: 605 (EC) 2015/830), US 29CFR1910.1200, Canada Hazardous Date Revised: 29 August 2018 Products Regulation Revision Number: 3 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING 1.1 Product Identifier: Trade Name (as labeled): Celtra® Ceram Powder Porcelains: Dentin, Opaceous Dentin, Natural Enamel, Opal Enamel, Power Dentin, Dentin Gingiva, Dentin Effect, Enamel Effect, Add-on Correction, Add-on Gingiva. Part/Item Number: 615130-615149, 615150 – 615156, 650130-650149, 615700-615725, 650700-650725; 615201-615206, 650201-650206, 615211-615216, 650211-650216; 615181-615186, 650181-650186; 615171-615175, 650171-650175; 615161-615169, 650161-650168; 615221-615226; 650221-650226; 615401-615404, 650401-650404; 615411-615415, 650411-650415. 1.2 Relevant Identified Uses of the Substance or Mixture and Uses Advised Against: Recommended Use: Used in the fabrication of dental crowns and bridges. Restrictions on Use: For Professional Use Only 1.3 Details of the Supplier of the Safety Data Sheet: Manufacturer/Supplier Name: Dentsply Sirona Prosthetics Manufacturer/Supplier Address: 570 West College Ave. York, PA 17401 Manufacturer/Supplier Telephone Number: 717-845-7511 (Product Information) Email address: [email protected] 1.4 Emergency Telephone Number: Emergency Contact Telephone Number: 800-243-1942 2. HAZARDS IDENTIFICATION 2.1 Classification of the Substance or Mixture: GHS Classification: Health Environmental Physical Not Hazardous Not Hazardous Not Hazardous 2.2 Label Elements: Not Required Celtra® Ceram Powder Porcelains Page 1 of 10 Signal Word: None Hazard Phrases Precautionary Phrases None Required None Required 2.3 Other Hazards: None known. -

Diffusion 2.Key
More on Diffusion • Diffusion systems • Multiple diffusion steps • Diffusion as nuisance • Lateral diffusion • dopant segregation • masks for dopants • The diffusion coefficient EE 432/532 diffusion II - 1 Diffusion systems In general, diffusion systems are similar to oxidation furnaces. The dopant can be provided in the form of gas, liquid, or solid. The basic approach is to deposit a glassy layer containing the dopant on the surface of the silicon. A reaction between the glass and the silicon transfers dopant into the silicon. boron: + + → phosphorus: + + → A potential problem with boron is that excess boron trioxide can cause “boron skin” on the silicon surface. This is difficult to remove by etching. A low-temperature oxidation step can help to break up the boron skin for easier removal. (See the lab information from the manufacturers of the boron source wafers to get more detail.) EE 432/532 diffusion II - 2 Gas sources silicon wafers in quartz boat quartz tube dopant gas + other carrier end cap and reactant gases (N2, O2, H2) boron doping: React diborane + + → with oxygen or carbon dioxide. + + → phosphorus doping: React + + phosphine with oxygen → In either case, the glassy oxide layer is formed on the silicon surface. The gases used are extremely toxic and special provisions must be used for handling and to clean the gas streams of any unreacted material. EE 432/532 diffusion II - 3 Liquid sources N2,O2 liquid dopant source temperature controlled vessel or bubbler boron doping: react boron + + tribromide with oxygen → phosphorus doping: react + + phosphorus oxychloride → with oxygen EE 432/532 diffusion II - 4 Solid sources (what we use) source wafers N2 or reactant gases (O2, H2) boron: Source wafers are made of nitride (BN). -

( 12 ) United States Patent
US010529585B2 (12 ) United States Patent ( 10 ) Patent No.: US 10,529,585 B2 Manna et al. (45 ) Date of Patent : Jan. 7 , 2020 (54 ) DRY STRIPPING OF BORON CARBIDE (58 ) Field of Classification Search HARDMASK None See application file for complete search history . ( 71) Applicant: Applied Materials , Inc. , Santa Clara , CA (US ) (56 ) References Cited ( 72 ) Inventors : Pramit Manna, Sunnyvale , CA (US ) ; U.S. PATENT DOCUMENTS Shishi Jiang , Santa Clara , CA (US ) ; 4,524,587 A 6/1985 Kantor Abhijit Basu Mallick , Fremont, CA 5,050,540 A 9/1991 Lindberg (US ) ; Kurtis Leschkies, San Jose , CA (US ) ( Continued ) FOREIGN PATENT DOCUMENTS ( 73 ) Assignee : APPLIED MATERIALS , INC . , Santa Clara , CA (US ) CN 101871043 A 10/2010 CN 104047676 A 9/2014 ( * ) Notice : Subject to any disclaimer, the term of this ( Continued ) patent is extended or adjusted under 35 U.S.C. 154 ( b ) by 0 days. OTHER PUBLICATIONS (21 ) Appl . No.: 15 /995,698 International Search Report and Written Opinion for PCT/ US2018 / 021715 dated Jun . 22 , 2018 . (22 ) Filed : Jun . 1 , 2018 (Continued ) (65 ) Prior Publication Data Primary Examiner — Jiong - Ping Lu US 2018/0350621 A1 (74 ) Attorney, Agent, or Firm Patterson + Sheridan Dec. 6 , 2018 LLP Related U.S. Application Data (57 ) ABSTRACT (60 ) Provisional application No. 62 /648,073 , filed on Mar. Embodiments of the disclosure generally relate to a method 26 , 2018 , provisional application No. 62/ 514,554 , for dry stripping a boron carbide layer deposited on a filed on Jun . 2 , 2017 . semiconductor substrate. In one embodiment, the method includes loading the substrate with the boron carbide layer (51 ) Int. -

TAXXI.-Y'he Compound H,R,O, and Its Salts
View Article Online / Journal Homepage / Table of Contents for this issue BAY: THE COMPOUND E~R,O~AND ITS SALTS. 803 Published on 01 January 1918. Downloaded by Brown University 26/10/2014 14:32:25. TAXXI.-Y'he Compound H,R,O, and its Salts. By RAMESCHANDRA RAY. TN a previous coinmunication (T., 1914, 105, 2162), it was shown that the so-called amorphous berm, prepared by the reduction of boron trioxicle by means of riiagiiesium, probably consisted of a sollid solution cof a lower oxide of boron, sometimes in combination with a little magnesium oxide; in elenient,ary bo'ron. If thin were View Article Online 804 RAY : THE COMPOUND H,B,O, AND ITS SALTS. really the case, it appelared probable thab the soluble part! of the fusion would contain solme o€ the lower oxide, together with excess of boron trioxide, and the, present investigation was undertaken with a view to detect and isolate the lower oxide. The fusion obtained by heating a mixture oF boron trioxide with magnesium powder generally contains magnesium boride, amorphous boron (so-called), magnesium o'xide, partly in combina- tion with boron trioxide, and, as will be shown later, the lower oxide or oxides of boron. If the operation is carried out under conditions which will now be described, the quantity of magnesium boride in the mixture is very small, although it cannot' be altogether eliminated. A mixture of 1 part, of magnesium powder and 3.5 parts of anhydrous boron trioxide, finely powdered and passed through a 120-mesh sieve, was heated in a covered magnesia-lined crubible, through which a current of hydrogen was passed, to a bright red heat for about three hours.