Administrative
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

BAFF-Neutralizing Interaction of Belimumab Related to Its Therapeutic Efficacy for Treating Systemic Lupus Erythematosus
ARTICLE DOI: 10.1038/s41467-018-03620-2 OPEN BAFF-neutralizing interaction of belimumab related to its therapeutic efficacy for treating systemic lupus erythematosus Woori Shin1, Hyun Tae Lee1, Heejin Lim1, Sang Hyung Lee1, Ji Young Son1, Jee Un Lee1, Ki-Young Yoo1, Seong Eon Ryu2, Jaejun Rhie1, Ju Yeon Lee1 & Yong-Seok Heo1 1234567890():,; BAFF, a member of the TNF superfamily, has been recognized as a good target for auto- immune diseases. Belimumab, an anti-BAFF monoclonal antibody, was approved by the FDA for use in treating systemic lupus erythematosus. However, the molecular basis of BAFF neutralization by belimumab remains unclear. Here our crystal structure of the BAFF–belimumab Fab complex shows the precise epitope and the BAFF-neutralizing mechanism of belimumab, and demonstrates that the therapeutic activity of belimumab involves not only antagonizing the BAFF–receptor interaction, but also disrupting the for- mation of the more active BAFF 60-mer to favor the induction of the less active BAFF trimer through interaction with the flap region of BAFF. In addition, the belimumab HCDR3 loop mimics the DxL(V/L) motif of BAFF receptors, thereby binding to BAFF in a similar manner as endogenous BAFF receptors. Our data thus provides insights for the design of new drugs targeting BAFF for the treatment of autoimmune diseases. 1 Department of Chemistry, Konkuk University, 120 Neungdong-ro, Gwangjin-gu, Seoul 05029, Republic of Korea. 2 Department of Bio Engineering, Hanyang University, 222 Wangsimni-ro, Seongdong-gu, Seoul 04763, Republic of Korea. These authors contributed equally: Woori Shin, Hyun Tae Lee, Heejin Lim, Sang Hyung Lee. -

Fig. L COMPOSITIONS and METHODS to INHIBIT STEM CELL and PROGENITOR CELL BINDING to LYMPHOID TISSUE and for REGENERATING GERMINAL CENTERS in LYMPHATIC TISSUES
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date Χ 23 February 2012 (23.02.2012) WO 2U12/U24519ft ft A2 (51) International Patent Classification: AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, A61K 31/00 (2006.01) CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (21) International Application Number: HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, PCT/US201 1/048297 KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, (22) International Filing Date: ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, 18 August 201 1 (18.08.201 1) NO, NZ, OM, PE, PG, PH, PL, PT, QA, RO, RS, RU, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, (25) Filing Language: English TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, (26) Publication Language: English ZW. (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 61/374,943 18 August 2010 (18.08.2010) US kind of regional protection available): ARIPO (BW, GH, 61/441,485 10 February 201 1 (10.02.201 1) US GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, 61/449,372 4 March 201 1 (04.03.201 1) US ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, (72) Inventor; and EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, ΓΓ, LT, LU, (71) Applicant : DEISHER, Theresa [US/US]; 1420 Fifth LV, MC, MK, MT, NL, NO, PL, PT, RO, RS, SE, SI, SK, Avenue, Seattle, WA 98101 (US). -

Pharmacologic Considerations in the Disposition of Antibodies and Antibody-Drug Conjugates in Preclinical Models and in Patients
antibodies Review Pharmacologic Considerations in the Disposition of Antibodies and Antibody-Drug Conjugates in Preclinical Models and in Patients Andrew T. Lucas 1,2,3,*, Ryan Robinson 3, Allison N. Schorzman 2, Joseph A. Piscitelli 1, Juan F. Razo 1 and William C. Zamboni 1,2,3 1 University of North Carolina (UNC), Eshelman School of Pharmacy, Chapel Hill, NC 27599, USA; [email protected] (J.A.P.); [email protected] (J.F.R.); [email protected] (W.C.Z.) 2 Division of Pharmacotherapy and Experimental Therapeutics, UNC Eshelman School of Pharmacy, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599, USA; [email protected] 3 Lineberger Comprehensive Cancer Center, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-919-966-5242; Fax: +1-919-966-5863 Received: 30 November 2018; Accepted: 22 December 2018; Published: 1 January 2019 Abstract: The rapid advancement in the development of therapeutic proteins, including monoclonal antibodies (mAbs) and antibody-drug conjugates (ADCs), has created a novel mechanism to selectively deliver highly potent cytotoxic agents in the treatment of cancer. These agents provide numerous benefits compared to traditional small molecule drugs, though their clinical use still requires optimization. The pharmacology of mAbs/ADCs is complex and because ADCs are comprised of multiple components, individual agent characteristics and patient variables can affect their disposition. To further improve the clinical use and rational development of these agents, it is imperative to comprehend the complex mechanisms employed by antibody-based agents in traversing numerous biological barriers and how agent/patient factors affect tumor delivery, toxicities, efficacy, and ultimately, biodistribution. -

Xolair/Omalizumab
Therapeutic/Humanized Antibodies ELISA Kits ‘Humanized antibodies” are ‘Animal Antibodies’ that have been modified by recombinant DNA-technology to reduce the overall content of the animal- portion of immunoglobulin so as to increase acceptance by humans or minimize ‘rejection’. By analogy, if the hands of a mouse is actually responsible for grabbing things then ‘humanized-mouse’ will only contain the ‘mouse hands’ and the rest of the body will be human. Since the size of the IgGs is similar in mouse and human, the size of the native mouse IgG and the ‘humanized IgG’ does not significantly change. Antigen recognition property of an antibody actually resides in small portion of the IgG molecule called the ‘antigen binding site or Fab’. Therefore, humanized antibodies contain the minimal portion from the Fab or the epitopes necessary for antigen binding. The process of "humanization of antibodies" is usually applied to animal (mouse) derived monoclonal antibodies for therapeutic use in humans (for example, antibodies developed as anti-cancer drugs). Xolair (Anti-IgE) is an example of humanized IgG. The portion of the mouse IgG that remains in the ‘humanized IgG’ may be recognized as foreign by humans and may result into the generation of “Human Anti-Drug Antibodies (HADA). The presence of anti-Drug antibody (e.g., Human Anti-Rituximab IgG) that may limit the long-term usage the humanized antibody (Rituximab). Not all monoclonal antibodies designed for human therapeutic use need be humanized since many therapies are short-term. The International Nonproprietary Names of humanized antibodies end in “-zumab”, as in “omalizumab”. Humanized antibodies are distinct from chimeric or fusion antibodies. -

Cutaneous Adverse Effects of Biologic Medications
REVIEW CME MOC Selena R. Pasadyn, BA Daniel Knabel, MD Anthony P. Fernandez, MD, PhD Christine B. Warren, MD, MS Cleveland Clinic Lerner College Department of Pathology Co-Medical Director of Continuing Medical Education; Department of Dermatology, Cleveland Clinic; of Medicine of Case Western and Department of Dermatology, W.D. Steck Chair of Clinical Dermatology; Director of Clinical Assistant Professor, Cleveland Clinic Reserve University, Cleveland, OH Cleveland Clinic Medical and Inpatient Dermatology; Departments of Lerner College of Medicine of Case Western Dermatology and Pathology, Cleveland Clinic; Assistant Reserve University, Cleveland, OH Clinical Professor, Cleveland Clinic Lerner College of Medicine of Case Western Reserve University, Cleveland, OH Cutaneous adverse effects of biologic medications ABSTRACT iologic therapy encompasses an expo- B nentially expanding arena of medicine. Biologic therapies have become widely used but often As the name implies, biologic therapies are de- cause cutaneous adverse effects. The authors discuss the rived from living organisms and consist largely cutaneous adverse effects of tumor necrosis factor (TNF) of proteins, sugars, and nucleic acids. A clas- alpha inhibitors, epidermal growth factor receptor (EGFR) sic example of an early biologic medication is inhibitors, small-molecule tyrosine kinase inhibitors insulin. These therapies have revolutionized (TKIs), and cell surface-targeted monoclonal antibodies, medicine and offer targeted therapy for an including how to manage these reactions -

Where Do Novel Drugs of 2016 Fit In?
FORMULARY JEOPARDY: WHERE DO NOVEL DRUGS OF 2016 FIT IN? Maabo Kludze, PharmD, MBA, CDE, BCPS, Associate Director Elizabeth A. Shlom, PharmD, BCPS, SVP & Director Clinical Pharmacy Program Acurity, Inc. Privileged and Confidential August 15, 2017 Privileged and Confidential Program Objectives By the end of the presentation, the pharmacist or pharmacy technician participant will be able to: ◆ Identify orphan drugs and first-in-class medications approved by the FDA in 2016. ◆ Describe the role of new agents approved for use in oncology patients. ◆ Identify and discuss the role of novel monoclonal antibodies. ◆ Discuss at least two new medications that address public health concerns. Neither Dr. Kludze nor Dr. Shlom have any conflicts of interest in regards to this presentation. Privileged and Confidential 2016 NDA Approvals (NMEs/BLAs) ◆ Nuplazid (primavanserin) P ◆ Adlyxin (lixisenatide) ◆ Ocaliva (obeticholic acid) P, O ◆ Anthim (obitoxaximab) O ◆ Rubraca (rucaparib camsylate) P, O ◆ Axumin (fluciclovive F18) P ◆ Spinraza (nusinersen sodium) P, O ◆ Briviact (brivaracetam) ◆ Taltz (ixekizumab) ◆ Cinqair (reslizumab) ◆ Tecentriq (atezolizumab) P ◆ Defitelio (defibrotide sodium) P, O ◆ Venclexta (venetoclax) P, O ◆ Epclusa (sofosburvir and velpatasvir) P ◆ Xiidra (lifitigrast) P ◆ Eucrisa (crisaborole) ◆ Zepatier (elbasvir and grazoprevir) P ◆ Exondys 51 (eteplirsen) P, O ◆ Zinbyrta (daclizumab) ◆ Lartruvo (olaratumab) P, O ◆ Zinplava (bezlotoxumab) P ◆ NETSTPOT (gallium Ga 68 dotatate) P, O O = Orphan; P = Priority Review; Red = BLA Privileged and Confidential History of FDA Approvals Privileged and Confidential Orphan Drugs ◆FDA Office of Orphan Products Development • Orphan Drug Act (1983) – drugs and biologics . “intended for safe and effective treatment, diagnosis or prevention of rare diseases/disorders that affect fewer than 200,000 people in the U.S. -

Changes in Serum Micrornas After Anti-IL-5 Biological Treatment of Severe Asthma
International Journal of Molecular Sciences Article Changes in Serum MicroRNAs after Anti-IL-5 Biological Treatment of Severe Asthma Manuel J. Rial 1,2, José A. Cañas 2,3 , José M. Rodrigo-Muñoz 2,3 , Marcela Valverde-Monge 1 , Beatriz Sastre 2,3 , Joaquín Sastre 1,3 and Victoria del Pozo 2,3,* 1 Allergy Unit, Hospital Universitario Fundación Jiménez Díaz, 28040 Madrid, Spain; [email protected] (M.J.R.); [email protected] (M.V.-M.); [email protected] (J.S.) 2 Department of Immunology, IIS-Fundación Jiménez Díaz, 28040 Madrid, Spain; [email protected] (J.A.C.); [email protected] (J.M.R.-M.); [email protected] (B.S.) 3 CIBER de Enfermedades Respiratorias (CIBERES), Instituto de Salud Carlos III, 28029 Madrid, Spain * Correspondence: [email protected]; Tel.: +34-9155-048-91 Abstract: There is currently enough evidence to think that miRNAs play a role in several key points in asthma, including diagnosis, severity of the disease, and response to treatment. Cells release different types of lipid double-membrane vesicles into the extracellular microenvironment, including exosomes, which function as very important elements in intercellular communication. They are capable of distributing genetic material, mRNA, mitochondrial DNA, and microRNAs (miRNAs). Serum miRNA screening was performed in order to analyze possible changes in serum miRNAs in 10 patients treated with reslizumab and 6 patients with mepolizumab after 8 weeks of treatment. The expression of miR-338-3p was altered after treatment (p < 0.05), although no significant differences between reslizumab and mepolizumab were found. Bioinformatic analysis showed that miR-338-3p regulates important pathways in asthma, such as the MAPK and TGF-β signaling pathways and the Citation: Rial, M.J.; Cañas, J.A.; biosynthesis/degradation of glucans (p < 0.05). -
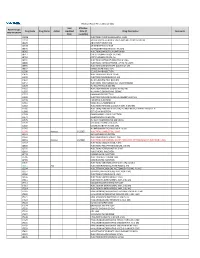
2021 Prior Authorization List Part B Appendix a (PDF)
Medicare Part B PA List Effective 2021 Last Effective Part B Drugs: Drug Code Drug Name Action Updated Date (if Drug Description Comments STEP THERAPY Date available) C9050 INJECTION, EMAPALUMAB-LZSG, 1 MG C9122 MOMETASONE FUROATE SINUS IMPLANT 10 MCG SINUVA J0129 ABATACEPT INJECTION J0178 AFLIBERCEPT INJECTION J0570 BUPRENORPHINE IMPLANT 74.2MG J0585 INJECTION,ONABOTULINUMTOXINA J0717 CERTOLIZUMAB PEGOL INJ 1MG J0718 CERTOLIZUMAB PEGOL INJ J0791 INJECTION CRIZANLIZUMAB-TMCA 5 MG J0800 INJECTION, CORTICOTROPIN, UP TO 40 UNITS J0896 INJECTION LUSPATERCEPT-AAMT 0.25 MG J0897 DENOSUMAB INJECTION J1300 ECULIZUMAB INJECTION J1428 INJECTION ETEPLIRSEN 10 MG J1429 INJECTION GOLODIRSEN 10 MG J1442 INJ FILGRASTIM EXCL BIOSIMIL J1447 INJECTION, TBO-FILGRASTIM, 1 MICROGRAM J1459 INJ IVIG PRIVIGEN 500 MG J1555 INJECTION IMMUNE GLOBULIN 100 MG J1556 INJ, IMM GLOB BIVIGAM, 500MG J1557 GAMMAPLEX INJECTION J1558 INJECTION IMMUNE GLOBULIN XEMBIFY 100 MG J1559 HIZENTRA INJECTION J1561 GAMUNEX-C/GAMMAKED J1562 INJECTION; IMMUNE GLOBULIN 10%, 5 GRAMS J1566 INJECTION, IMMUNE GLOBULIN, INTRAVENOUS, LYOPHILIZED (E.G. P J1568 OCTAGAM INJECTION J1569 GAMMAGARD LIQUID INJECTION J1572 FLEBOGAMMA INJECTION J1575 INJ IG/HYALURONIDASE 100 MG IG J1599 IVIG NON-LYOPHILIZED, NOS J1602 GOLIMUMAB FOR IV USE 1MG J1745 INJ INFLIXIMAB EXCL BIOSIMILR 10 MG J1930 Remove 1/1/2021 INJECTION, LANREOTIDE, 1 MG J2323 NATALIZUMAB INJECTION J2350 INJECTION OCRELIZUMAB 1 MG J2353 Remove 1/1/2021 INJECTION, OCTREOTIDE, DEPOT FORM FOR INTRAMUSCULAR INJECTION, 1 MG J2357 INJECTION, OMALIZUMAB, -

MEP-AST Mepolizumab Versus Placebo for Asthma
MEP-AST Mepolizumab versus placebo for asthma Mepolizumab versus placebo for asthma Review information Review type: Intervention Review number: MEP-AST Authors Colin Powell1, Stephen J Milan2, Kerry Dwan3, Lynne Bax4, Nicola Walters5 1Department of Child Health, Institute of Molecular and Experimental Medicine, Cardiff University School of Medicine, Cardiff, UK 2Lancaster Health Hub, Lancaster University, Lancaster, UK 3Department of Biostatistics, University of Liverpool, Liverpool, UK 4Lancashire Care NHS Foundation Trust, Preston, UK 5Chest Unit, St George's University Hospitals NHS Foundation Trust, London, UK Citation example: Powell C, Milan SJ, Dwan K, Bax L, Walters N. Mepolizumab versus placebo for asthma. Cochrane Database of Systematic Reviews 2015 , Issue 7 . Art. No.: CD010834. DOI: 10.1002/14651858.CD010834.pub2 . Contact person Colin Powell Senior Lecturer Department of Child Health Institute of Molecular and Experimental Medicine, Cardiff University School of Medicine Cardiff UK E-mail: [email protected] Dates Assessed as Up-to-date:19 November 2014 Date of Search: 19 November 2014 Next Stage Expected: 6 June 2017 Protocol First Published:Issue 11 , 2013 Review First Published: Issue 7 , 2015 Last Citation Issue: Issue 7 , 2015 What's new Date Event Description History Date Event Description Abstract Background Mepolizumab is a human monoclonal antibody against interleukin-5 (IL-5), the main cytokine involved in the activation of eosinophils, which in turn causes airway inflammation. Recent studies have suggested these agents may have a role in reducing exacerbations and improving health-related quality of life (HRQoL). There are no recommendations for the use of mepolizumab in adults or children in the recent update of the BTS/SIGN guidelines (BTS/SIGN 2014). -

Asthma Agents
APPROVED PA Criteria Initial Approval Date: July 10, 2019 Revised Date: January 20, 2021 CRITERIA FOR PRIOR AUTHORIZATION Asthma Agents BILLING CODE TYPE For drug coverage and provider type information, see the KMAP Reference Codes webpage. MANUAL GUIDELINES Prior authorization will be required for all current and future dose forms available. All medication-specific criteria, including drug-specific indication, age, and dose for each agent is defined in Table 1 below. Benralizumab (Fasenra®) Dupilumab (Dupixent®) Mepolizumab (Nucala®) Omalizumab (Xolair®) Reslizumab (Cinqair®) GENERAL CRITERIA FOR INITIAL PRIOR AUTHORIZATION: (must meet all of the following) • Must be approved for the indication, age, and not exceed dosing limits listed in Table 1. • Must be prescribed by or in consultation with a pulmonologist, allergist, or immunologist.1,2 • For all agents listed, the preferred PDL drug, which treats the PA indication, is required unless the patient meets the non-preferred PDL PA criteria. • Must have experienced ≥ 2 exacerbations within the last 12 months despite meeting all of the following (exacerbation is defined as requiring the use of oral/systemic corticosteroids, urgent care/hospital admission, or intubation: o Patient adherence to two long-term controller medications, including a high-dose inhaled corticosteroid 1,2 (ICS) and a long-acting beta2-agonist (LABA) listed in Table 2. ▪ Combination ICS/LABA and ICS/LABA/LAMA products meet the requirement of two controller medications. o Patient must have had an adequate trial (at least 90 consecutive days) of a leukotriene modifier or a long-acting muscarinic antagonist (LAMA) as a third long-term controller medication listed in Table 2. -

Cimzia (Certolizumab Pegol) AHM
Cimzia (Certolizumab Pegol) AHM Clinical Indications • Cimzia (Certolizumab Pegol) is considered medically necessary for adult members 18 years of age or older with moderately-to-severely active disease when ALL of the following conditions are met o Moderately-to-severely active Crohn's disease as manifested by 1 or more of the following . Diarrhea . Abdominal pain . Bleeding . Weight loss . Perianal disease . Internal fistulae . Intestinal obstruction . Megacolon . Extra-intestinal manifestations: arthritis or spondylitis o Crohn's disease has remained active despite treatment with 1 or more of the following . Corticosteroids . 6-mercaptopurine/azathioprine • Certollizumab pegol (see note) is considered medically necessary for persons with active psoriatic arthritis who meet criteria in Psoriasis and Psoriatic Arthritis: Biological Therapies. • Cimzia, (Certolizumab Pegol), alone or in combination with methotrexate (MTX), is considered medically necessary for the treatment of adult members 18 years of age or older with moderately-to-severely active rheumatoid arthritis (RA). • Cimzia (Certolizumab pegol is considered medically necessary for reducing signs and symptoms of members with active ankylosing spondylitis who have an inadequate response to 2 or more NSAIDs. • Cimzia (Certolizumab Pegol) is considered investigational for all other indications (e.g.,ocular inflammation/uveitis; not an all-inclusive list) because its effectiveness for indications other than the ones listed above has not been established. Notes • There are several brands of targeted immune modulators on the market. There is a lack of reliable evidence that any one brand of targeted immune modulator is superior to other brands for medically necessary indications. Enbrel (etanercept), Humira (adalimumab), Remicade (infliximab), Simponi (golimumab), Simponi Aria (golimumab intravneous), and Stelara (ustekinumab) brands of targeted immune modulators ("least cost brands of targeted immune modulators") are less costly to the plan. -

Anti-TNF-Α (Certolizumab), Humanized Antibody
FOR RESEARCH USE ONLY! rev 06/21 Anti-TNF-α (Certolizumab), Humanized Antibody CATALOG NO.: A2142-100 (100 µg) BACKGROUND DESCRIPTION: The research-grade biosimilar is a humanized TNF-α monoclonal antibody. The original monoclonal antibody (Certolizumab pegol (CZP)) consists of polyethylene glycol conjugated to the antigen-binding fragment of the humanized monoclonal antibody. The antibody targets and neutralizes soluble and membrane forms of tumor necrosis factor-alpha (TNF-α) and inhibits TNF-α mediated signaling in vitro. Unlike other TNF inhibitors, this antibody does not contain the Fc region. As a result, decreased Fc-mediated effects such as antibody-dependent cell-mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC) are observed. The original monoclonal antibody received FDA approval to treat moderate to severe rheumatoid arthritis, Crohn’s disease, ankylosing spondylitis, and psoriatic arthritis. TNFSF2, TNF-alpha, TNFA, TNF-α, TNFα ALTERNATE NAMES: ANTIBODY TYPE: Monoclonal HOST/ISOTYPE: Recombinant / Fab-IgG1-kappa SOURCE: CHO cells IMMUNOGEN: Human TNF-α FORM: Liquid FORMULATION: In PBS, pH 7.5 SPECIES REACTIVITY: Human STORAGE CONDITIONS: Store at -80 ºC. Avoid repeated freeze-thaw cycles This information is only intended as a guide. The optimal dilutions must be determined by the user RELATED PRODUCTS: Anti-TNF-α (Adalimumab), humanized Antibody (Cat. No. A1048) Anti-TNF alpha (Infliximab), Human IgG1 Antibody (Cat. No. A1097) Anti-TNF alpha (Humicade), Human IgG4 Antibody (Cat. No. A1093) Anti-EGFR (Cetuximab), Chimeric Antibody (Cat. No. A1047) Anti-EGFR (Matuzumab), Human IgG1 Antibody (Cat. No. A1090) FOR RESEARCH USE ONLY! Not to be used on humans. 155 S. Milpitas Blvd., Milpitas, CA 95035 USA | T: (408)493-1800 F: (408)493-1801 | www.biovision.com | [email protected] .