Alkanes and Cycloalkanes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
![C9-14 Aliphatic [2-25% Aromatic] Hydrocarbon Solvents Category SIAP](https://docslib.b-cdn.net/cover/2852/c9-14-aliphatic-2-25-aromatic-hydrocarbon-solvents-category-siap-12852.webp)
C9-14 Aliphatic [2-25% Aromatic] Hydrocarbon Solvents Category SIAP
CoCAM 2, 17-19 April 2012 BIAC/ICCA SIDS INITIAL ASSESSMENT PROFILE Chemical C -C Aliphatic [2-25% aromatic] Hydrocarbon Solvents Category Category 9 14 Substance Name CAS Number Stoddard solvent 8052-41-3 Chemical Names Kerosine, petroleum, hydrodesulfurized 64742-81-0 and CAS Naphtha, petroleum, hydrodesulfurized heavy 64742-82-1 Registry Solvent naphtha, petroleum, medium aliphatic 64742-88-7 Numbers Note: Substances in this category are also commonly known as mineral spirits, white spirits, or Stoddard solvent. CAS Number Chemical Description † 8052-41-3 Includes C8 to C14 branched, linear, and cyclic paraffins and aromatics (6 to 18%), <50ppmV benzene † 64742-81-0 Includes C9 to C14 branched, linear, and cyclic paraffins and aromatics (10 to Structural 25%), <100 ppmV benzene Formula † and CAS 64742-82-1 Includes C8 to C13 branched, linear, and cyclic paraffins and aromatics (15 to 25%), <100 ppmV benzene Registry † Numbers 64742-88-7 Includes C8 to C13 branched, linear, and cyclic paraffins and aromatics (14 to 20%), <50 ppmV benzene Individual category member substances are comprised of aliphatic hydrocarbon molecules whose carbon numbers range between C9 and C14; approximately 80% of the aliphatic constituents for a given substance fall within the C9-C14 carbon range and <100 ppmV benzene. In some instances, the carbon range of a test substance is more precisely defined in the test protocol. In these instances, the specific carbon range (e.g. C8-C10, C9-C10, etc.) will be specified in the SIAP. * It should be noted that other substances defined by the same CAS RNs may have boiling ranges outside the range of 143-254° C and that these substances are not covered by the category. -

Chapter 3 • Ratio: Cnh2n+2
Organic Chemistry, 5th Edition Alkane Formulas L. G. Wade, Jr. • All C-C single bonds • Saturated with hydrogens Chapter 3 • Ratio: CnH2n+2 Structure and Stereochemistry • Alkane homologs: CH3(CH2)nCH3 of Alkanes • Same ratio for branched alkanes H H H H H H CH H C C C C H H H H C CCH Jo Blackburn H H H H H H Richland College, Dallas, TX H => Butane, C4H10 Dallas County Community College District Chapter 3Isobutane, C H 2 © 2003, Prentice Hall 4 10 Common Names Pentanes H H H H H H H CH • Isobutane, “isomer of butane” H C C C C C H H H • Isopentane, isohexane, etc., methyl H H H H H H H C CCC H branch on next-to-last carbon in chain. H H H H • Neopentane, most highly branched n-pentane, C5H12 isopentane, C5H12 • Five possible isomers of hexane, CH3 18 isomers of octane and 75 for H3C C CH3 decane! CH3 => => neopentane, C5H12 Chapter 3 3 Chapter 3 4 Longest Chain IUPAC Names • The number of carbons in the longest • Find the longest continuous carbon chain determines the base name: chain. ethane, hexane. (Listed in Table 3.2, • Number the carbons, starting closest to page 81.) the first branch. • If there are two possible chains with the • Name the groups attached to the chain, same number of carbons, use the chain using the carbon number as the locator. with the most substituents. • Alphabetize substituents. H3C CH CH CH • Use di-, tri-, etc., for multiples of same 2 3 CH3 substituent. -

1 Refinery and Petrochemical Processes
3 1 Refinery and Petrochemical Processes 1.1 Introduction The combination of high demand for electric cars and higher automobile engine effi- ciency in the future will mean less conversion of petroleum into fuels. However, the demand for petrochemicals is forecast to rise due to the increase in world popula- tion. With this, it is expected that modern and more innovative technologies will be developed to serve the growth of the petrochemical market. In a refinery process, petroleum is converted into petroleum intermediate prod- ucts, including gases, light/heavy naphtha, kerosene, diesel, light gas oil, heavy gas oil, and residue. From these intermediate refinery product streams, several fuels such as fuel gas, liquefied petroleum gas, gasoline, jet fuel, kerosene, auto diesel, and other heavy products such as lubricants, bunker oil, asphalt, and coke are obtained. In addition, these petroleum intermediates can be further processed and separated into products for petrochemical applications. In this chapter, petroleum will be introduced first. Petrochemicals will be intro- duced in the second part of the chapter. Petrochemicals – the main subject of this book – will address three major areas, (i) the production of the seven cornerstone petrochemicals: methane and synthesis gas, ethylene, propylene, butene, benzene, toluene, and xylenes; (ii) the uses of the seven cornerstone petrochemicals, and (iii) the technology to separate petrochemicals into individual components. 1.2 Petroleum Petroleum is derived from the Latin words “petra” and “oleum,” which means “rock” and “oil,” respectively. Petroleum also is known as crude oil or fossil fuel. It is a thick, flammable, yellow-to-black mixture of gaseous, liquid, and solid hydrocarbons formed from the remains of plants and animals. -

Heterocyclic Chemistry Updated
1 | Page Heterocyclic Chemistry By Dr Vipul Kataria Definition: A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements (N, O, S, P) as members of its ring. Introduction: Heterocyclic compounds may be defined as cyclic compounds having as ring members atoms of at least two different elements. It means the cyclic compound has one another atom different than carbon. Heterocyclic compounds have much importance in organic chemistry because of abundant presence of heterocyclic compounds in present pharmaceutical drugs. Like other organic compounds, there are several defined rules for naming heterocyclic compounds. IUPAC rules for nomenclature of heterocyclic systems (SPU 2012, 2 Marks) The system for nomenclature for heterocyclic systems was given by Hantzsch and Widman. The Hantzsch-Widman nomenclature is based on the type (Z) of the heteroatom; the ring size (n) and nature of the ring, whether it is saturated or unsaturated. This system of nomenclature applies to monocyclic 3-to-10-membered ring heterocycles. Type of the heteroatom: The type of heteroatom is indicated by a prefix as shown below for common hetreroatoms. Dr Vipul Kataria, Chemistry Department, V. P. & R. P. T. P. Science College, VV Nagar 2 | Pag e When two or more of the same heteroatoms are present, the prrefix di, tri, tetra… are used. e.g. dioxa, triaza, dithia. If the heteroatoms are different their atomic number in that grroup is considered. Thus order of numbering will be O, S, N, P, Si. Ring size: The ring size is indicated by a suffix according to Table I below. -

Dehydrogenation by Heterogeneous Catalysts
Dehydrogenation by Heterogeneous Catalysts Daniel E. Resasco School of Chemical Engineering and Materials Science University of Oklahoma Encyclopedia of Catalysis January, 2000 1. INTRODUCTION Catalytic dehydrogenation of alkanes is an endothermic reaction, which occurs with an increase in the number of moles and can be represented by the expression Alkane ! Olefin + Hydrogen This reaction cannot be carried out thermally because it is highly unfavorable compared to the cracking of the hydrocarbon, since the C-C bond strength (about 246 kJ/mol) is much lower than that of the C-H bond (about 363 kJ/mol). However, in the presence of a suitable catalyst, dehydrogenation can be carried out with minimal C-C bond rupture. The strong C-H bond is a closed-shell σ orbital that can be activated by oxide or metal catalysts. Oxides can activate the C-H bond via hydrogen abstraction because they can form O-H bonds, which can have strengths comparable to that of the C- H bond. By contrast, metals cannot accomplish the hydrogen abstraction because the M- H bonds are much weaker than the C-H bond. However, the sum of the M-H and M-C bond strengths can exceed the C-H bond strength, making the process thermodynamically possible. In this case, the reaction is thought to proceed via a three centered transition state, which can be described as a metal atom inserting into the C-H bond. The C-H bond bridges across the metal atom until it breaks, followed by the formation of the corresponding M-H and M-C bonds.1 Therefore, dehydrogenation of alkanes can be carried out on oxides as well as on metal catalysts. -

Cyclobutane Derivatives in Drug Discovery
Cyclobutane Derivatives in Drug Discovery Overview Key Points Unlike larger and conformationally flexible cycloalkanes, Cyclobutane adopts a rigid cyclobutane and cyclopropane have rigid conformations. Due to the ring strain, cyclobutane adopts a rigid puckered puckered conformation Offer ing advantages on (~30°) conformation. This unique architecture bestowed potency, selectivity and certain cyclobutane-containing drugs with unique pharmacokinetic (PK) properties. When applied appropriately, cyclobutyl profile. scaffolds may offer advantages on potency, selectivity and pharmacokinetic (PK) profile. Bridging Molecules for Innovative Medicines 1 PharmaBlock designs and Cyclobutane-containing Drugs synthesizes over 1846 At least four cyclobutane-containing drugs are currently on the market. cyclobutanes, and 497 Chemotherapy carboplatin (Paraplatin, 1) for treating ovarian cancer was cyclobutane products are prepared to lower the strong nephrotoxicity associated with cisplatin. By in stock. CLICK HERE to replacing cisplatin’s two chlorine atoms with cyclobutane-1,1-dicarboxylic find detailed product acid, carboplatin (1) has a much lower nephrotoxicity than cisplatin. On information on webpage. the other hand, Schering-Plough/Merck’s hepatitis C virus (HCV) NS3/4A protease inhibitor boceprevir (Victrelis, 2) also contains a cyclobutane group in its P1 region. It is 3- and 19-fold more potent than the 1 corresponding cyclopropyl and cyclopentyl analogues, respectively. Androgen receptor (AR) antagonist apalutamide (Erleada, 4) for treating castration-resistant prostate cancer (CRPC) has a spirocyclic cyclobutane scaffold. It is in the same series as enzalutamide (Xtandi, 3) discovered by Jung’s group at UCLA in the 2000s. The cyclobutyl- (4) and cyclopentyl- derivative have activities comparable to the dimethyl analogue although the corresponding six-, seven-, and eight-membered rings are slightly less 2 active. -

Condensed Cyclic Compound, Composition Including The
(19) TZZ¥_ _T (11) EP 3 184 522 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 28.06.2017 Bulletin 2017/26 C07D 403/14 (2006.01) H01L 51/00 (2006.01) (21) Application number: 16205894.5 (22) Date of filing: 21.12.2016 (84) Designated Contracting States: • INAYAMA, Satoshi AL AT BE BG CH CY CZ DE DK EE ES FI FR GB Yokohama-city, Kanagawa 230-0027 (JP) GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO • ISHII, Norihito PL PT RO RS SE SI SK SM TR Yokohama-city, Kanagawa 230-0027 (JP) Designated Extension States: • SHIBATA, Katsunori BA ME Yokohama-city, Kanagawa 230-0027 (JP) Designated Validation States: • ITO, Mitsunori MA MD Yokohama-city, Kanagawa 230-0027 (JP) (30) Priority: 22.12.2015 JP 2015250531 (74) Representative: Scheuermann, Erik Elkington and Fife LLP (71) Applicant: Samsung Electronics Co., Ltd. Prospect House Gyeonggi-do 16677 (KR) 8 Pembroke Road Sevenoaks, Kent TN13 1XR (GB) (72) Inventors: • KATO, Fumiaki Yokohama-city, Kanagawa 230-0027 (JP) (54) CONDENSED CYCLIC COMPOUND, COMPOSITION INCLUDING THE CONDENSED CYCLIC COMPOUND, ORGANIC LIGHT-EMITTING DEVICE INCLUDING THE CONDENSED CYCLIC COMPOUND, AND METHOD OF MANUFACTURING THE ORGANIC LIGHT-EMITTING DEVICE (57) A condensed cyclic compound, a composition including the condensed cyclic compound, an organic light-emit- ting device including the condensed cyclic compound, and a method of manufacturing the organic light-emitting device are provided. EP 3 184 522 A1 Printed by Jouve, 75001 PARIS (FR) EP 3 184 522 A1 Description FIELD OF THE INVENTION 5 [0001] One or more embodiments relate to a condensed cyclic compound, a composition including the condensed cyclic compound, an organic light-emitting device including the condensed cyclic compound, and a method of manufac- turing the organic light-emitting device. -

Recent Advances in the Total Synthesis of Cyclobutane-Containing Natural Products Cite This: Org
Volume 7 | Number 1 | 7 January 2020 ORGANIC CHEMISTRY FRONTIERS rsc.li/frontiers-organic ORGANIC CHEMISTRY FRONTIERS View Article Online REVIEW View Journal | View Issue Recent advances in the total synthesis of cyclobutane-containing natural products Cite this: Org. Chem. Front., 2020, 7, 136 Jinshan Li,†a Kai Gao, †a Ming Bianb and Hanfeng Ding *a,c Complex natural products bearing strained cyclobutane subunits, including terpenoids, alkaloids and steroids, not only display fascinating architectures, but also show potent biological activities. Due to their unique structures as critical core skeletons in these molecules, a variety of new strategies for the con- Received 24th September 2019, struction of cyclobutane rings have greatly emerged during the last decade. In this review, we wish to Accepted 11th November 2019 summarize the recent progress in the cyclobutane-containing natural product synthesis with an emphasis DOI: 10.1039/c9qo01178a on disconnection tactics employed to forge the four-membered rings, aiming to provide a complement rsc.li/frontiers-organic to existing reviews. 1. Introduction stereoselectively, poses significant challenges in synthetic chemistry. On the other hand, cyclobutanes readily undergo a In the class of strained carbocycles, cyclobutanes have been number of ring-opening reactions by virtue of their tendency known as intriguing structural motifs for more than one to release inherent strain energies. In some cases, however, century but remained relatively less explored in parallel with striking ring strains can be dramatically reduced by the instal- their homologues.1 Due to the highly strained ring systems (ca. lation of a gem-dialkyl substituent (through the Thorpe–Ingold − 26.7 kcal mol 1), construction of cyclobutane rings, especially effect),2 a carbonyl group, a heteroatom, or other functional- ities (Fig. -

Chemistry 0310 - Organic Chemistry 1 Chapter 3
Dr. Peter Wipf Chemistry 0310 - Organic Chemistry 1 Chapter 3. Reactions of Alkanes The heterolysis of covalent bonds yields anions and cations, whereas the homolysis creates radicals. Radicals are species with unpaired electrons that react mostly as electrophiles, seeking a single electron to complete their octet. Free radicals are important reaction intermediates and are formed in initiation reactions under conditions that cause the homolytic cleavage of bonds. In propagation steps, radicals abstract hydrogen or halogen atoms to create new radicals. Combinations of radicals are rare due to the low concentration of these reactive intermediates and result in termination of the radical chain. !CHAIN REACTION SUMMARY reactant product initiation PhCH3 HCl Cl 2 h DH = -16 kcal/mol chain-carrying intermediates n o r D (low concentrations) PhCH2 . Cl . propagation PhCH2 . or Cl . PhCH . 2 DH = -15 kcal/mol or Cl . PhCH2Cl or PhCH CH Ph PhCH2Cl Cl2 2 2 PhCH2Cl or Cl2 termination product reactant termination Alkanes are converted to alkyl halides by free radical halogenation reactions. The relative stability of radicals is increased by conjugation and hyperconjugation: R H H H . CH2 > R C . > R C . > H C . > H C . R R R H Oxygen is a diradical. In the presence of free-radical initiators such as metal salts, organic compounds and oxygen react to give hydroperoxides. These autoxidation reactions are responsible for the degradation reactions of oils, fatty acids, and other biological substances when exposed to air. Antioxidants such as hindered phenols are important food additives. Vitamins E and C are biological antioxidants. Radical chain reactions of chlorinated fluorocarbons in the stratosphere are responsible for the "ozone hole". -

SAFETY DATA SHEET Cyclopentane
SAFETY DATA SHEET Cyclopentane Section 1. Identification GHS product identifier : Cyclopentane Chemical name : cyclopentane Other means of : cyclopentane (dot); pentamethylene identification Product use : Synthetic/Analytical chemistry. Synonym : cyclopentane (dot); pentamethylene SDS # : 001124 Supplier's details : Airgas USA, LLC and its affiliates 259 North Radnor-Chester Road Suite 100 Radnor, PA 19087-5283 1-610-687-5253 24-hour telephone : 1-866-734-3438 Section 2. Hazards identification OSHA/HCS status : This material is considered hazardous by the OSHA Hazard Communication Standard (29 CFR 1910.1200). Classification of the : FLAMMABLE LIQUIDS - Category 2 substance or mixture AQUATIC HAZARD (ACUTE) - Category 3 AQUATIC HAZARD (LONG-TERM) - Category 3 GHS label elements Hazard pictograms : Signal word : Danger Hazard statements : Highly flammable liquid and vapor. May form explosive mixtures in Air. Harmful to aquatic life with long lasting effects. Precautionary statements General : Read label before use. Keep out of reach of children. If medical advice is needed, have product container or label at hand. Prevention : Wear protective gloves. Wear eye or face protection. Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. No smoking. Use explosion- proof electrical, ventilating, lighting and all material-handling equipment. Use only non- sparking tools. Take precautionary measures against static discharge. Keep container tightly closed. Avoid release to the environment. Response : IF ON SKIN (or hair): Take off immediately all contaminated clothing. Rinse skin with water or shower. Storage : Store in a well-ventilated place. Keep cool. Disposal : Dispose of contents and container in accordance with all local, regional, national and international regulations. Hazards not otherwise : None known. -
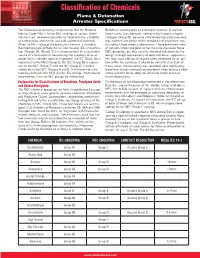
Classification of Chemicals
Classification of Chemicals Flame & Detonation Arrester Specifications PROTECTOSEAL ® The Protectoseal Company recommends that the National Butadiene would qualify as a Group D material. In each of Electric Code (NEC) Article 500, rankings of various chemi - these cases, the chemicals were primarly listed in a higher cals be used, whenever possible, to determine the suitability category (Group B), because of relatively high pressure read - of a detonation arrester for use with a particular chemical. ings noted in one phase of the standard test procedure con - When no NEC rating of the particular chemical is available, ducted by Underwriters Laboratories. These pressures were the International Electrotechnical Commission (IEC) classifica - of concern when categorizing the chemicals because these tion (Groups IIA, IIB and IIC) is recommended as a secondary NEC groupings are also used as standard indicators for the source of information for determining the suitability of an ar - design strength requirements of electrical boxes, apparatus, rester for its intended service. In general, the IEC Group IIA is etc. that must withstand the pressures generated by an igni - equivalent to the NEC Group D; the IEC Group IIB is equiva - tion within the container. It should be noted that, in each of lent to the NEC Group C; and the IEC Group IIC includes these cases, the test pressures recorded were significantly chemicals in the NEC Groups A and B. In the event of a dis - lower than those commonly encountered when testing a deto - crepancy between the NEC and the IEC ratings, Protectoseal nation arrester for its ability to withstand stable and over - recommends that the NEC groups be referenced. -

Organic Chemistry PEIMS Code: N1120027 Abbreviation: ORGCHEM Number of Credits That May Be Earned: 1/2-1
Course: Organic Chemistry PEIMS Code: N1120027 Abbreviation: ORGCHEM Number of credits that may be earned: 1/2-1 Brief description of the course (150 words or less): Organic chemistry is an introductory course that is designed for the student who intends to continue future study in the sciences. The student will learn the concepts and applications of organic chemistry. Topics covered include aliphatic and aromatic compounds, alcohols, aldehydes, ketones, acids, ethers, amines, spectra, and stereochemistry. A brief introduction into biochemistry is also provided. The laboratory experiments will familiarize the student with the important laboratory techniques, specifically spectroscopy. Traditional high school chemistry courses focus on the inorganic aspects of chemistry whereas organic chemistry introduces the student to organic compounds and their properties, mechanisms of formations, and introduces the student to laboratory techniques beyond the traditional high school chemistry curriculum. Essential Knowledge and Skills of the course: (a) General requirements. This course is recommended for students in Grades 11-12. The recommended prerequisite for this course is AP Chemistry. (b) Introduction. Organic chemistry is designed to introduce students to the fundamental concepts of organic chemistry and key experimental evidence and data, which support these concepts. Students will learn to apply these data and concepts to chemical problem solving. Additionally, students will learn that organic chemistry is still evolving by reading about current breakthroughs in the field. Finally, students will gain appreciation for role that organic chemistry plays in modern technological developments in diverse fields, ranging from biology to materials science. (c) Knowledge and skills. (1) The student will be able to write both common an IUPAC names for the hydrocarbon.