State of Knowledge of Drug-Impaired Driving
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Withdrawing Benzodiazepines in Primary Care
PC\/ICU/ ADTiriC • CNS Drugs 2009,-23(1): 19-34 KtVltW MKIIWLC 1172-7047/I»/O(X)1«119/S4W5/C1 © 2009 Adis Dato Intocmation BV. All rights reserved. Withdrawing Benzodiazepines in Primary Care Malcolm Luder} Andre Tylee^ and ]ohn Donoghue^ 1 Institute of Psychiatry, King's College London, London, England 2 John Moores University, Liverpool, Scotland Contents Abstract ' 19 1. Benzodiazepine Usage 22 2. Interventions 23 2.1 Simple interventions 23 2.2 Piiarmacoiogicai interventions 25 2.3 Psychoiogical Interventions 26 2.4 Meta-Anaiysis ot Various interventions 27 3. Outcomes 28 4. Practicai Issues 29 5. Otiier Medications 30 5.1 Antidepressants 30 5.2 Symptomatic Treatments 30 6. Conciusions 31 Abstract The use of benzodiazepine anxiolytics and hypnotics continues to excite controversy. Views differ from expert to expert and from country to country as to the extent of the problem, or even whether long-term benzodiazepine use actually constitutes a problem. The adverse effects of these drugs have been extensively documented and their effectiveness is being increasingly questioned. Discontinua- tion is usually beneficial as it is followed by improved psychomotor and cognitive functioning, particularly in the elderly. The potential for dependence and addic- tion have also become more apparent. The licensing of SSRIs for anxiety disorders has widened the prescdbers' therapeutic choices (although this group of medications also have their own adverse effects). Melatonin agonists show promise in some forms of insomnia. Accordingly, it is now even more imperative that long-term benzodiazepine users be reviewed with respect to possible discon- tinuation. Strategies for discontinuation start with primary-care practitioners, who are still the main prescdbers. -

Drug Class Review Newer Drugs for Insomnia
Drug Class Review Newer Drugs for Insomnia Final Update 2 Report October 2008 The Agency for Healthcare Research and Quality has not yet seen or approved this report. The purpose of Drug Effectiveness Review Project reports is to make available information regarding the comparative clinical effectiveness and harms of different drugs. Reports are not usage guidelines, nor should they be read as an endorsement of or recommendation for any particular drug, use, or approach. Oregon Health & Science University does not recommend or endorse any guideline or recommendation developed by users of these reports. Update 1: July 2006 Original: December 2005 Susan Carson, MPH Marian S. McDonagh, PharmD Sujata Thakurta, MPA:HA Po-Yin Yen, MS Drug Effectiveness Review Project Marian McDonagh, PharmD, Principal Investigator Oregon Evidence-based Practice Center Mark Helfand, MD, MPH, Director Copyright © 2008 by Oregon Health & Science University Portland, Oregon 97239. All rights reserved. Final Report Update 2 Drug Effectiveness Review Project The medical literature relating to the topic is scanned periodically (see http://www.ohsu.edu/xd/research/centers-institutes/evidence-based-policy- center/derp/documents/methods.cfm for scanning process description). Upon review of the last scan, the Drug Effectiveness Review Project governance group elected not to proceed with another full update of this report based on the information contained in the scan. Some portions of the report may not be up to date. Prior versions of this report can be accessed at the DERP website. Insomnia Page 2 of 87 Final Report Update 2 Drug Effectiveness Review Project TABLE OF CONTENTS INTRODUCTION ............................................................................................................. 6 Scope and Key Questions ...................................................................................................................... -

GABA Receptors
D Reviews • BIOTREND Reviews • BIOTREND Reviews • BIOTREND Reviews • BIOTREND Reviews Review No.7 / 1-2011 GABA receptors Wolfgang Froestl , CNS & Chemistry Expert, AC Immune SA, PSE Building B - EPFL, CH-1015 Lausanne, Phone: +41 21 693 91 43, FAX: +41 21 693 91 20, E-mail: [email protected] GABA Activation of the GABA A receptor leads to an influx of chloride GABA ( -aminobutyric acid; Figure 1) is the most important and ions and to a hyperpolarization of the membrane. 16 subunits with γ most abundant inhibitory neurotransmitter in the mammalian molecular weights between 50 and 65 kD have been identified brain 1,2 , where it was first discovered in 1950 3-5 . It is a small achiral so far, 6 subunits, 3 subunits, 3 subunits, and the , , α β γ δ ε θ molecule with molecular weight of 103 g/mol and high water solu - and subunits 8,9 . π bility. At 25°C one gram of water can dissolve 1.3 grams of GABA. 2 Such a hydrophilic molecule (log P = -2.13, PSA = 63.3 Å ) cannot In the meantime all GABA A receptor binding sites have been eluci - cross the blood brain barrier. It is produced in the brain by decarb- dated in great detail. The GABA site is located at the interface oxylation of L-glutamic acid by the enzyme glutamic acid decarb- between and subunits. Benzodiazepines interact with subunit α β oxylase (GAD, EC 4.1.1.15). It is a neutral amino acid with pK = combinations ( ) ( ) , which is the most abundant combi - 1 α1 2 β2 2 γ2 4.23 and pK = 10.43. -

52Nd Annual Meeting
ACNP 52nd Annual Meeting Final Program December 8-12, 2013 The Westin Diplomat Resort & Spa Hollywood, Florida President: David A. Lewis, M.D. Program Committee Chair: Randy D. Blakely, Ph.D. Program Committee Co-Chair: Pat R. Levitt, Ph.D. This meeting is jointly sponsored by the Vanderbilt University School of Medicine Department of Psychiatry and the American College of Neuropsychopharmacology. Dear ACNP Members and Guests, It is a distinct pleasure to welcome you to the 2014 meeting of the American College of Neuropsychopharmacology! This 52nd annual meeting will again provide opportunities for the exercise of the College’s core values: the spirit of Collegiality, promoting in each other the best in science, training and service; participation in Community, pursuing together the goals of understanding the neurobiology of brain diseases and eliminating their burden on individuals and our society; and engaging in Celebration, taking the time to recognize and enjoy the contributions and accomplishments of our members and guests. Under the excellent leadership of Randy Blakely and Pat Levitt, the Program Committee has done a superb job in assembling an outstanding slate of scientific presentations. Based on membership feedback, the meeting schedule has been designed with the goals of achieving an optimal mix of topics and types of sessions, increasing the diversity of participating scientists and creating more time for informal interactions. The presentations will highlight both the breadth of the investigative interests of ACNP membership -
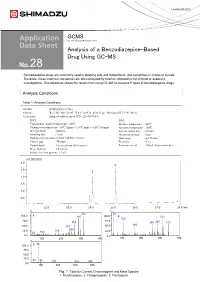
Analysis of a Benzodiazepine-Based Drug Using GC-MS 28
LAAN-E-MS-E028 GCMS Gas Chromatograph Mass Spectrometer Analysis of a Benzodiazepine-Based Drug Using GC-MS 28 Benzodiazepine drugs are commonly used in sleeping aids and tranquilizers, and sometimes in crimes or suicide. Therefore, these chemical substances are often analyzed by forensic laboratories for criminal or academic investigations. This datasheet shows the results from using GC-MS to measure 9 types of benzodiazepine drugs. Analysis Conditions Table 1: Analysis Conditions GC-MS :GCMS-QP2010 Ultra Column : Rxi®-5Sil MS (30 mL. X 0.25 mmI.D., df=0.25 µm, Shimadzu GLC P/N:13623) Glass insert :Silanized splitless insert (P/N: 221-48876-03) [GC] [MS] Vaporization chamber temperature : 260℃ Interface temperature : 280℃ Column oven temperature : 60℃ (2min) -> (10℃/min) -> 320℃ (10min) Ion source temperature : 200℃ Injection mode : Splitless Solvent elution time : 2.0 min Sampling time : 1 min Measurement mode : Scan High pressure injection method: 250 kPa (1.5 min) Mass range : m/z 35-600 Carrier gas : Helium Event time : 0.3 sec Control mode :Linear velocity (45.6 cm/sec) Emission current : 150 µA (high sensitivity) Purge flow rate :3.0 ml/min Sample injection quantity :1.0 µL (x1,000,000) 3.0 3 2.5 2 2.0 1.5 1.0 1 0.5 22.0 23.0 24.0 25.0 26.0 27.0 28.0 min % % 312 55 100.0 1 285 100.0 2 313 109 75.0 75.0 266 259287 342 166 50.0 238 50.0 248 25.0 183 25.0 63 109 0.0 0.0 100 200 300 400 100 200 300 400 % 86 100.0 3 75.0 50.0 25.0 58 99 183 315 387 0.0 100 200 300 400 Fig. -

Treatment of Social Phobia with Antidepressants
Antidepressants for Social Phobia Treatment of Social Phobia With Antidepressants Franklin R. Schneier, M.D. This article reviews evidence for the utility of antidepressant medications in the treatment of social phobia. Monoamine oxidase inhibitors (MAOIs) were the first antidepressants shown to be effective © Copyrightfor social phobia, but 2001dietary restrictions Physicians and a relatively Postgraduate high rate of adverse effects Press, often relegate Inc. MAOIs to use after other treatments have been found ineffective. Reversible inhibitors of monoamine oxidase (RIMAs) hold promise as safer alternatives to MAOIs, but RIMAs may be less effective and are currently unavailable in the United States. Selective serotonin reuptake inhibitors (SSRIs), of which paroxetine has been the best studied in social phobia to date, have recently emerged as a first- line treatment for the generalized subtype of social phobia. The SSRIs are well tolerated and consis- tently have been shown to be efficacious in controlled trials. (J Clin Psychiatry 2001;62[suppl 1]:43–48) arly evidence that antidepressantsOne might personal have utility copy maypressant be printed treatment of other anxiety disorders, such as panic E in the treatment of social phobia emerged in the disorder and obsessive-compulsive disorder. In particular, 1970s when studies found efficacy for monoamine oxidase the high rate of comorbidity of anxiety disorders with de- inhibitors (MAOIs) in patient samples that included both pression3 makes treatment with antidepressants an efficient -

Pharmacological Properties of GABAA- Receptors Containing Gamma1
Molecular Pharmacology Fast Forward. Published on November 4, 2005 as DOI: 10.1124/mol.105.017236 Molecular PharmacologyThis article hasFast not Forward.been copyedited Published and formatted. on The November final version 7, may 2005 differ as from doi:10.1124/mol.105.017236 this version. MOLPHARM/2005/017236 Pharmacological properties of GABAA- receptors containing gamma1- subunits Khom S.1, Baburin I.1, Timin EN, Hohaus A., Sieghart W., Hering S. Downloaded from Department of Pharmacology and Toxicology, University of Vienna Center of Brain Research , Medical University of Vienna, Division of Biochemistry and molpharm.aspetjournals.org Molecular Biology at ASPET Journals on September 27, 2021 1 Copyright 2005 by the American Society for Pharmacology and Experimental Therapeutics. Molecular Pharmacology Fast Forward. Published on November 4, 2005 as DOI: 10.1124/mol.105.017236 This article has not been copyedited and formatted. The final version may differ from this version. MOLPHARM/2005/017236 Running Title: GABAA- receptors containing gamma1- subunits Corresponding author: Steffen Hering Department of Pharmacology and Toxicology University of Vienna Althanstrasse 14 Downloaded from A-1090 Vienna Telephone number: +43-1-4277-55301 Fax number: +43-1-4277-9553 molpharm.aspetjournals.org [email protected] Text pages: 29 at ASPET Journals on September 27, 2021 Tables: 2 Figures: 7 References: 26 Abstract: 236 words Introduction:575 words Discussion:1383 words 2 Molecular Pharmacology Fast Forward. Published on November 4, 2005 as DOI: 10.1124/mol.105.017236 This article has not been copyedited and formatted. The final version may differ from this version. MOLPHARM/2005/017236 Abstract GABAA receptors composed of α1, β2, γ1-subunits are expressed in only a few areas of the brain and thus represent interesting drug targets. -

CNS Drug Reviews Vol
CNS Drug Reviews Vol. 10, No. 1, pp. 45–76 © 2004 Neva Press, Branford, Connecticut Clinical Pharmacology, Clinical Efficacy, and Behavioral Toxicity of Alprazolam: A Review of the Literature Joris C. Verster and Edmund R. Volkerts Utrecht Institute for Pharmaceutical Sciences, Department of Psychopharmacology, University of Utrecht, Utrecht, The Netherlands Keywords: Alprazolam — Benzodiazepines — Anxiety — Depression — Panic — Sedation. ABSTRACT Alprazolam is a benzodiazepine derivative that is currently used in the treatment of generalized anxiety, panic attacks with or without agoraphobia, and depression. Alprazo- lam has a fast onset of symptom relief (within the first week); it is unlikely to produce de- pendency or abuse. No tolerance to its therapeutic effect has been reported. At discontinu- ation of alprazolam treatment, withdrawal and rebound symptoms are common. Hence, alprazolam discontinuation must be tapered. An exhaustive review of the literature showed that alprazolam is significantly superior to placebo, and is at least equally effective in the relief of symptoms as tricyclic antide- pressants (TCAs), such as imipramine. However, although alprazolam and imipramine are significantly more effective than placebo in the treatment of panic attacks, Selective Sero- tonin Reuptake Inhibitors (SSRIs) appear to be superior to either of the two drugs. Therefore, alprazolam is recommended as a second line treatment option, when SSRIs are not effective or well tolerated. In addition to its therapeutic effects, alprazolam produces adverse effects, such as drowsiness and sedation. Since alprazolam is widely used, many clinical studies investi- gated its cognitive and psychomotor effects. It is evident from these studies that alprazo- lam may impair performance in a variety of skills in healthy volunteers as well as in pa- tients. -

Insights Into the Mechanisms of Action Ofthe MAO Inhibitors Phenelzine and Tranylcypromine
Insights into the Mechanisms of Action of the MAO Inhibitors Phenelzine and Tranylcypromine: A Review Glen B. Baker, Ph.D., Ronald T. Coutts, Ph.D., D.Sc., Kevin F. McKenna, M.D., and Rhonda L. Sherry-McKenna, B.Sc. Neurochemical Research Unit, Department of Psychiatry and Faculty of Pharmacy and Pharmaceutical Sciences, University of Alberta, Edmonton, Alberta Submitted: July 10, 1992 Accepted: October 7, 1992 Although the non-selective monoamine oxidase inhibitors phenelzine and tranylcypromine have been used for many years, much still remains to be understood about their mechanisms of action. Other factors, in addition to the inhibition of monoamine oxidase and the subsequent elevation of brain levels of the catecholamines and 5-hydroxytryptamine, may contribute to the overall pharma- cological profiles ofthese drugs. This review also considers the effects on brain levels of amino acids and trace amines, uptake and release of neurotransmitter amines at nerve terminals, receptors for amino acids and amines, and enzymes other than monoamine oxidase, including enzymes involved in metabolism of other drugs. The possible contributions of metabolism and stereochemistry to the actions of these monoamine oxidase inhibitors are discussed. Key Words: amino acids, monoamine oxidase, neurotransmitter amines, phenelzine, tranylcypromine, uptake Despite the fact that the non-selective monoamine oxidase nerve endings (Baker et al 1977; Raiteri et al 1977) and/or (MAO) inhibitors phenelzine (PLZ) and tranylcypromine may act as neuromodulators through direct actions on recep- (TCP) (see Fig. 1) have been used clinically for many years, tors for the catecholamines and/or 5-HT (Jones 1983; much remains to be learned about theirmechanisms ofaction. -

In the Treatment of Generalised Anxiety Disorder Does Pharmacology
Characteristics Table for The Clinical Question: In the treatment of generalised anxiety disorder does 6/18/2010 12:21:10 pharmacology improve outcome? Comparisons Included in this Clinical Question Anticonvulsants versus Placebo Anticonvulsants vs Venlafaxine (SNRI) Antihistamine vs Placebo Benzodiazepines versus vs Placebo Anticonvulsants FELTNER2003 DARCIS1995 KASPER2009 KASPER2009 LADER1998 FELTNER2003 MONTGOMERY2006 MONTGOMERY2006 LLORCA2002 PANDE2003 MONTGOMERY2008 PFIZER2005 PANDE2003 RICKELS2005 PFIZER2005 POHL2005 RICKELS2005 Benzodiazepines versus Azapirones Benzodiazepines versus Placebo Buspirone vs Placebo Duloxetine (SNRI) vs. placebo BOURIN1992 ANDREATINI2002 DAVIDSON1999 HARTFORD2007 ANSSEAU2001 LADER1998 KOPONEN2007 CUTLER1993A MAJERCSIK2003 NICOLINI2009 FELTNER2003 POLLACK1997 RYNN2008 FRESQUET2000 SRAMEK1996 HACKETT2003 LYDIARD1997 MCLEOD1992 MOLLER2001 PANDE2003 PFIZER2008 RICKELS2000B RICKELS2005 Duloxetine (SNRI) vs. Venlafaxine Quetiapine versus Placebo SSRI vs Venlafaxine SSRIs versus Placebo (SNRI) ASTRAZENECA2007A BOSE2008 ALLGULANDER2004 HARTFORD2007 ASTRAZENECA2007B ASTRAZENECA2007A NICOLINI2009 ASTRAZENECA2007C ASTRAZENECA2007B ASTRAZENECA2008 BALDWIN2006 BOSE2008 BRAWMAN-MINTZER2006 DAVIDSON2004 GOODMAN2005 GSK2002 GSK2005 HEWETT2001 LENZE2005 LENZE2009 PFIZER2008 POLLACK2001 RICKELS2003 SSRIs versus SSRIs TCA vs Placebo Venlafaxine (SNRI) versus Azapirones Venlafaxine (SNRI) versus placebo BALDWIN2006 MCLEOD1992 DAVIDSON1999 ALLGULANDER2001 BALL2005 BOSE2008 BIELSKI2005 DAVIDSON1999 GELENBERG2000 HACKETT2003 -

Drug Class Review on Newer Sedative Hypnotics
Drug Class Review on Newer Sedative Hypnotics Final Report December 2005 The Agency for Healthcare Research and Quality has not yet seen or approved this report The purpose of this report is to make available information regarding the comparative effectiveness and safety profiles of different drugs within pharmaceutical classes. Reports are not usage guidelines, nor should they be read as an endorsement of, or recommendation for, any particular drug, use or approach. Oregon Health & Science University does not recommend or endorse any guideline or recommendation developed by users of these reports. Susan Carson, MPH Po-Yin Yen, MS Marian S. McDonagh, PharmD Oregon Evidence-based Practice Center Oregon Health & Science University Mark Helfand, MD, MPH, Director Copyright © 2005 by Oregon Health & Science University Portland, Oregon 97201. All rights reserved. Final Report Drug Effectiveness Review Project TABLE OF CONTENTS Introduction....................................................................................................................... 4 Scope and Key Questions ............................................................................................. 4 Methods.............................................................................................................................. 6 Literature Search........................................................................................................... 6 Study Selection ............................................................................................................ -

Yasakli Maddeler
YASAKLI MADDELER Ülkemizde yarış atlarında kullanılan/kullanılma ihtimali olan ilaçların/maddelerin belirlenmesinde; ilacın farmakolojisi (öncelikle etkisi, etki grubu), farmasötik şekli, kullanılma yolu, kullanılma alanı, yarış sonucunu etkileme durumu, ulusal mevzuata göre veteriner hekimlikte ve/veya yarış atlarında kullanmak için ruhsatlı/izinli olup-olmama durumu, etiket-dışı kullanılma gibi hususlar dikkate alınmıştır. Eşik değeri (Threshold level) olan maddelerin (vücutta şekillenirler, çevre ve yem kaynaklı olabilirler) miktarı, belirlenen miktarı aştığında yasaklı-madde kullanılması/doping ihlali olarak değerlendirilir. Tedavide kullanılan/tarama limiti (Screening level) olan maddelerle ilgili değerlendirmede; bu maddelerle ilgili olarak yapılan doping analizlerinde ölçülen miktar, idrarda/plazmada belirlenen tarama değerini aştığında yasaklı madde kullanılması/doping-ihlali olarakdeğerlendirilir. Ana madde yanında, izomeri, metaboliti, metabolit izomeri, tuzları, esterleri, eterleri, diğer türevleri, ön-ilaç da yasaklı-madde listesinde yer alır; doping analizinde ortaya konulmaları doping ihlali olarak değerlendirilir. Maddeler ve yasaklı uygulamalar 6 başlık altında toplanmıştır. Bu liste ilgili uluslararası kuruluşların mevzuat ve düzenlemeleri ile ilgili maddelerin Farmakolojik özellikleri ve etkileri dikkate alınarak hazırlanmıştır. 1. YasaklıMaddeler Yasaklı maddeler; yarış atlarında tıbbi olarak kullanım yeri olmayan, atın yarış performansını açık şekilde etkileyen maddelerdir. İllegal kullanılan maddeler (Türkiye’de veteriner