Quantitative Proteome Profiling of Dystrophic Dog Skeletal Muscle
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genetic Mutations and Mechanisms in Dilated Cardiomyopathy
Genetic mutations and mechanisms in dilated cardiomyopathy Elizabeth M. McNally, … , Jessica R. Golbus, Megan J. Puckelwartz J Clin Invest. 2013;123(1):19-26. https://doi.org/10.1172/JCI62862. Review Series Genetic mutations account for a significant percentage of cardiomyopathies, which are a leading cause of congestive heart failure. In hypertrophic cardiomyopathy (HCM), cardiac output is limited by the thickened myocardium through impaired filling and outflow. Mutations in the genes encoding the thick filament components myosin heavy chain and myosin binding protein C (MYH7 and MYBPC3) together explain 75% of inherited HCMs, leading to the observation that HCM is a disease of the sarcomere. Many mutations are “private” or rare variants, often unique to families. In contrast, dilated cardiomyopathy (DCM) is far more genetically heterogeneous, with mutations in genes encoding cytoskeletal, nucleoskeletal, mitochondrial, and calcium-handling proteins. DCM is characterized by enlarged ventricular dimensions and impaired systolic and diastolic function. Private mutations account for most DCMs, with few hotspots or recurring mutations. More than 50 single genes are linked to inherited DCM, including many genes that also link to HCM. Relatively few clinical clues guide the diagnosis of inherited DCM, but emerging evidence supports the use of genetic testing to identify those patients at risk for faster disease progression, congestive heart failure, and arrhythmia. Find the latest version: https://jci.me/62862/pdf Review series Genetic mutations and mechanisms in dilated cardiomyopathy Elizabeth M. McNally, Jessica R. Golbus, and Megan J. Puckelwartz Department of Human Genetics, University of Chicago, Chicago, Illinois, USA. Genetic mutations account for a significant percentage of cardiomyopathies, which are a leading cause of conges- tive heart failure. -

Defining Functional Interactions During Biogenesis of Epithelial Junctions
ARTICLE Received 11 Dec 2015 | Accepted 13 Oct 2016 | Published 6 Dec 2016 | Updated 5 Jan 2017 DOI: 10.1038/ncomms13542 OPEN Defining functional interactions during biogenesis of epithelial junctions J.C. Erasmus1,*, S. Bruche1,*,w, L. Pizarro1,2,*, N. Maimari1,3,*, T. Poggioli1,w, C. Tomlinson4,J.Lees5, I. Zalivina1,w, A. Wheeler1,w, A. Alberts6, A. Russo2 & V.M.M. Braga1 In spite of extensive recent progress, a comprehensive understanding of how actin cytoskeleton remodelling supports stable junctions remains to be established. Here we design a platform that integrates actin functions with optimized phenotypic clustering and identify new cytoskeletal proteins, their functional hierarchy and pathways that modulate E-cadherin adhesion. Depletion of EEF1A, an actin bundling protein, increases E-cadherin levels at junctions without a corresponding reinforcement of cell–cell contacts. This unexpected result reflects a more dynamic and mobile junctional actin in EEF1A-depleted cells. A partner for EEF1A in cadherin contact maintenance is the formin DIAPH2, which interacts with EEF1A. In contrast, depletion of either the endocytic regulator TRIP10 or the Rho GTPase activator VAV2 reduces E-cadherin levels at junctions. TRIP10 binds to and requires VAV2 function for its junctional localization. Overall, we present new conceptual insights on junction stabilization, which integrate known and novel pathways with impact for epithelial morphogenesis, homeostasis and diseases. 1 National Heart and Lung Institute, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. 2 Computing Department, Imperial College London, London SW7 2AZ, UK. 3 Bioengineering Department, Faculty of Engineering, Imperial College London, London SW7 2AZ, UK. 4 Department of Surgery & Cancer, Faculty of Medicine, Imperial College London, London SW7 2AZ, UK. -

Unequal Allelic Expression of Wild-Type and Mutated B-Myosin in Familial Hypertrophic Cardiomyopathy
Basic Res Cardiol (2011) 106:1041–1055 DOI 10.1007/s00395-011-0205-9 ORIGINAL CONTRIBUTION Unequal allelic expression of wild-type and mutated b-myosin in familial hypertrophic cardiomyopathy Snigdha Tripathi • Imke Schultz • Edgar Becker • Judith Montag • Bianca Borchert • Antonio Francino • Francisco Navarro-Lopez • Andreas Perrot • Cemil O¨ zcelik • Karl-Josef Osterziel • William J. McKenna • Bernhard Brenner • Theresia Kraft Received: 26 May 2011 / Revised: 29 June 2011 / Accepted: 7 July 2011 / Published online: 19 July 2011 Ó The Author(s) 2011. This article is published with open access at Springerlink.com Abstract Familial hypertrophic cardiomyopathy (FHC) genotyped and clinically well-characterized FHC patients is an autosomal dominant disease, which in about 30% of were analyzed. The fraction of mutated MYH7-mRNA in the patients is caused by missense mutations in one allele five patients with mutation R723G averaged to 66 and 68% of the b-myosin heavy chain (b-MHC) gene (MYH7). To of total MYH7-mRNA in soleus and myocardium, respec- address potential molecular mechanisms underlying the tively. For mutations I736T, R719W and V606M, fractions family-specific prognosis, we determined the relative of mutated MYH7-mRNA in M. soleus were 39, 57 and expression of mutant versus wild-type MYH7-mRNA. We 29%, respectively. For all mutations, unequal abundance found a hitherto unknown mutation-dependent unequal was similar at the protein level. Importantly, fractions of expression of mutant to wild-type MYH7-mRNA, which is mutated transcripts were comparable among siblings, in paralleled by similar unequal expression of b-MHC at the younger relatives and unrelated carriers of the same protein level. -

Transcriptional Control of Tissue-Resident Memory T Cell Generation
Transcriptional control of tissue-resident memory T cell generation Filip Cvetkovski Submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the Graduate School of Arts and Sciences COLUMBIA UNIVERSITY 2019 © 2019 Filip Cvetkovski All rights reserved ABSTRACT Transcriptional control of tissue-resident memory T cell generation Filip Cvetkovski Tissue-resident memory T cells (TRM) are a non-circulating subset of memory that are maintained at sites of pathogen entry and mediate optimal protection against reinfection. Lung TRM can be generated in response to respiratory infection or vaccination, however, the molecular pathways involved in CD4+TRM establishment have not been defined. Here, we performed transcriptional profiling of influenza-specific lung CD4+TRM following influenza infection to identify pathways implicated in CD4+TRM generation and homeostasis. Lung CD4+TRM displayed a unique transcriptional profile distinct from spleen memory, including up-regulation of a gene network induced by the transcription factor IRF4, a known regulator of effector T cell differentiation. In addition, the gene expression profile of lung CD4+TRM was enriched in gene sets previously described in tissue-resident regulatory T cells. Up-regulation of immunomodulatory molecules such as CTLA-4, PD-1, and ICOS, suggested a potential regulatory role for CD4+TRM in tissues. Using loss-of-function genetic experiments in mice, we demonstrate that IRF4 is required for the generation of lung-localized pathogen-specific effector CD4+T cells during acute influenza infection. Influenza-specific IRF4−/− T cells failed to fully express CD44, and maintained high levels of CD62L compared to wild type, suggesting a defect in complete differentiation into lung-tropic effector T cells. -

Genome Editing for the Understanding and Treatment of Inherited Cardiomyopathies
International Journal of Molecular Sciences Review Genome Editing for the Understanding and Treatment of Inherited Cardiomyopathies 1, 1, 1,2, Quynh Nguyen y , Kenji Rowel Q. Lim y and Toshifumi Yokota * 1 Department of Medical Genetics, Faculty of Medicine and Dentistry, University of Alberta, Edmonton, AB T6G2H7, Canada; [email protected] (Q.N.); [email protected] (K.R.Q.L.) 2 The Friends of Garrett Cumming Research & Muscular Dystrophy Canada, HM Toupin Neurological Science Research Chair, Edmonton, AB T6G2H7, Canada * Correspondence: [email protected]; Tel.: +1-780-492-1102 These authors contributed equally to the work. y Received: 28 December 2019; Accepted: 19 January 2020; Published: 22 January 2020 Abstract: Cardiomyopathies are diseases of heart muscle, a significant percentage of which are genetic in origin. Cardiomyopathies can be classified as dilated, hypertrophic, restrictive, arrhythmogenic right ventricular or left ventricular non-compaction, although mixed morphologies are possible. A subset of neuromuscular disorders, notably Duchenne and Becker muscular dystrophies, are also characterized by cardiomyopathy aside from skeletal myopathy. The global burden of cardiomyopathies is certainly high, necessitating further research and novel therapies. Genome editing tools, which include zinc finger nucleases (ZFNs), transcription activator-like effector nucleases (TALENs) and clustered regularly interspaced short palindromic repeats (CRISPR) systems have emerged as increasingly important technologies in studying -

RESEARCH ARTICLE Body Weight-Dependent Troponin T Alternative Splicing Is Evolutionarily Conserved from Insects to Mammals and I
1523 The Journal of Experimental Biology 214, 1523-1532 © 2011. Published by The Company of Biologists Ltd doi:10.1242/jeb.051763 RESEARCH ARTICLE Body weight-dependent troponin T alternative splicing is evolutionarily conserved from insects to mammals and is partially impaired in skeletal muscle of obese rats Rudolf J. Schilder1,*, Scot R. Kimball1, James H. Marden2 and Leonard S. Jefferson1 1Department of Cellular and Molecular Physiology, The Pennsylvania State University College of Medicine, 500 University Drive, Hershey, PA 17033, USA and 2Department of Biology, The Pennsylvania State University, 208 Mueller Lab, University Park, PA 16802, USA *Author for correspondence ([email protected]) Accepted 19 January 2011 SUMMARY Do animals know at a physiological level how much they weigh, and, if so, do they make homeostatic adjustments in response to changes in body weight? Skeletal muscle is a likely tissue for such plasticity, as weight-bearing muscles receive mechanical feedback regarding body weight and consume ATP in order to generate forces sufficient to counteract gravity. Using rats, we examined how variation in body weight affected alternative splicing of fast skeletal muscle troponin T (Tnnt3), a component of the thin filament that regulates the actin–myosin interaction during contraction and modulates force output. In response to normal growth and experimental body weight increases, alternative splicing of Tnnt3 in rat gastrocnemius muscle was adjusted in a quantitative fashion. The response depended on weight per se, as externally attached loads had the same effect as an equal change in actual body weight. Examining the association between Tnnt3 alternative splicing and ATP consumption rate, we found that the Tnnt3 splice form profile had a significant association with nocturnal energy expenditure, independently of effects of weight. -

TNNT3 Rabbit Pab
Leader in Biomolecular Solutions for Life Science TNNT3 Rabbit pAb Catalog No.: A15323 Basic Information Background Catalog No. The binding of Ca(2+) to the trimeric troponin complex initiates the process of muscle A15323 contraction. Increased Ca(2+) concentrations produce a conformational change in the troponin complex that is transmitted to tropomyosin dimers situated along actin Observed MW filaments. The altered conformation permits increased interaction between a myosin 37kDa head and an actin filament which, ultimately, produces a muscle contraction. The troponin complex has protein subunits C, I, and T. Subunit C binds Ca(2+) and subunit I Calculated MW binds to actin and inhibits actin-myosin interaction. Subunit T binds the troponin 29kDa/30kDa/31kDa complex to the tropomyosin complex and is also required for Ca(2+)-mediated activation of actomyosin ATPase activity. There are 3 different troponin T genes that Category encode tissue-specific isoforms of subunit T for fast skeletal-, slow skeletal-, and cardiac-muscle. This gene encodes fast skeletal troponin T protein; also known as Primary antibody troponin T type 3. Alternative splicing results in multiple transcript variants encoding additional distinct troponin T type 3 isoforms. A developmentally regulated switch Applications between fetal/neonatal and adult troponin T type 3 isoforms occurs. Additional splice WB variants have been described but their biological validity has not been established. Mutations in this gene may cause distal arthrogryposis multiplex congenita type 2B Cross-Reactivity (DA2B). Mouse, Rat Recommended Dilutions Immunogen Information WB 1:200 - 1:2000 Gene ID Swiss Prot 7140 P45378 Immunogen Recombinant fusion protein containing a sequence corresponding to amino acids 147-256 of human TNNT3 (NP_001036246.1). -
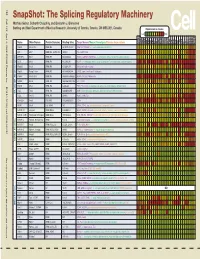
Snapshot: the Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J
192 Cell SnapShot: The Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J. Blencowe 133 Banting and Best Department of Medical Research, University of Toronto, Toronto, ON M5S 3E1, Canada Expression in mouse , April4, 2008©2008Elsevier Inc. Low High Name Other Names Protein Domains Binding Sites Target Genes/Mouse Phenotypes/Disease Associations Amy Ceb Hip Hyp OB Eye SC BM Bo Ht SM Epd Kd Liv Lu Pan Pla Pro Sto Spl Thy Thd Te Ut Ov E6.5 E8.5 E10.5 SRp20 Sfrs3, X16 RRM, RS GCUCCUCUUC SRp20, CT/CGRP; −/− early embryonic lethal E3.5 9G8 Sfrs7 RRM, RS, C2HC Znf (GAC)n Tau, GnRH, 9G8 ASF/SF2 Sfrs1 RRM, RS RGAAGAAC HipK3, CaMKIIδ, HIV RNAs; −/− embryonic lethal, cond. KO cardiomyopathy SC35 Sfrs2 RRM, RS UGCUGUU AChE; −/− embryonic lethal, cond. KO deficient T-cell maturation, cardiomyopathy; LS SRp30c Sfrs9 RRM, RS CUGGAUU Glucocorticoid receptor SRp38 Fusip1, Nssr RRM, RS ACAAAGACAA CREB, type II and type XI collagens SRp40 Sfrs5, HRS RRM, RS AGGAGAAGGGA HipK3, PKCβ-II, Fibronectin SRp55 Sfrs6 RRM, RS GGCAGCACCUG cTnT, CD44 DOI 10.1016/j.cell.2008.03.010 SRp75 Sfrs4 RRM, RS GAAGGA FN1, E1A, CD45; overexpression enhances chondrogenic differentiation Tra2α Tra2a RRM, RS GAAARGARR GnRH; overexpression promotes RA-induced neural differentiation SR and SR-Related Proteins Tra2β Sfrs10 RRM, RS (GAA)n HipK3, SMN, Tau SRm160 Srrm1 RS, PWI AUGAAGAGGA CD44 SWAP Sfrs8 RS, SWAP ND SWAP, CD45, Tau; possible asthma susceptibility gene hnRNP A1 Hnrnpa1 RRM, RGG UAGGGA/U HipK3, SMN2, c-H-ras; rheumatoid arthritis, systemic lupus -

NICU Gene List Generator.Xlsx
Neonatal Crisis Sequencing Panel Gene List Genes: A2ML1 - B3GLCT A2ML1 ADAMTS9 ALG1 ARHGEF15 AAAS ADAMTSL2 ALG11 ARHGEF9 AARS1 ADAR ALG12 ARID1A AARS2 ADARB1 ALG13 ARID1B ABAT ADCY6 ALG14 ARID2 ABCA12 ADD3 ALG2 ARL13B ABCA3 ADGRG1 ALG3 ARL6 ABCA4 ADGRV1 ALG6 ARMC9 ABCB11 ADK ALG8 ARPC1B ABCB4 ADNP ALG9 ARSA ABCC6 ADPRS ALK ARSL ABCC8 ADSL ALMS1 ARX ABCC9 AEBP1 ALOX12B ASAH1 ABCD1 AFF3 ALOXE3 ASCC1 ABCD3 AFF4 ALPK3 ASH1L ABCD4 AFG3L2 ALPL ASL ABHD5 AGA ALS2 ASNS ACAD8 AGK ALX3 ASPA ACAD9 AGL ALX4 ASPM ACADM AGPS AMELX ASS1 ACADS AGRN AMER1 ASXL1 ACADSB AGT AMH ASXL3 ACADVL AGTPBP1 AMHR2 ATAD1 ACAN AGTR1 AMN ATL1 ACAT1 AGXT AMPD2 ATM ACE AHCY AMT ATP1A1 ACO2 AHDC1 ANK1 ATP1A2 ACOX1 AHI1 ANK2 ATP1A3 ACP5 AIFM1 ANKH ATP2A1 ACSF3 AIMP1 ANKLE2 ATP5F1A ACTA1 AIMP2 ANKRD11 ATP5F1D ACTA2 AIRE ANKRD26 ATP5F1E ACTB AKAP9 ANTXR2 ATP6V0A2 ACTC1 AKR1D1 AP1S2 ATP6V1B1 ACTG1 AKT2 AP2S1 ATP7A ACTG2 AKT3 AP3B1 ATP8A2 ACTL6B ALAS2 AP3B2 ATP8B1 ACTN1 ALB AP4B1 ATPAF2 ACTN2 ALDH18A1 AP4M1 ATR ACTN4 ALDH1A3 AP4S1 ATRX ACVR1 ALDH3A2 APC AUH ACVRL1 ALDH4A1 APTX AVPR2 ACY1 ALDH5A1 AR B3GALNT2 ADA ALDH6A1 ARFGEF2 B3GALT6 ADAMTS13 ALDH7A1 ARG1 B3GAT3 ADAMTS2 ALDOB ARHGAP31 B3GLCT Updated: 03/15/2021; v.3.6 1 Neonatal Crisis Sequencing Panel Gene List Genes: B4GALT1 - COL11A2 B4GALT1 C1QBP CD3G CHKB B4GALT7 C3 CD40LG CHMP1A B4GAT1 CA2 CD59 CHRNA1 B9D1 CA5A CD70 CHRNB1 B9D2 CACNA1A CD96 CHRND BAAT CACNA1C CDAN1 CHRNE BBIP1 CACNA1D CDC42 CHRNG BBS1 CACNA1E CDH1 CHST14 BBS10 CACNA1F CDH2 CHST3 BBS12 CACNA1G CDK10 CHUK BBS2 CACNA2D2 CDK13 CILK1 BBS4 CACNB2 CDK5RAP2 -

Hypertrophic Cardiomyopathy (HCM)
case example: Hypertrophic Cardiomyopathy (HCM) who is the patient? • 24 year-old male with no cardiac symptoms; assessed due to family history • Normal ECG (no left ventricular hypertrophy or conduction disease), cardiac echocardiogram, cardiac MRI • No prior cardiovascular genetic testing what is the SCA 60 family history? • Family history of sudden cardiac arrest d.60 (SCA) and hypertrophic obstructive cardiomyopathy N • Father died at age 45 from SCA: HCM found on requested autopsy report No other SCA 45 reported d.45 • Paternal grandfather died at age 60 SCA HCM dx autopsy from SCA • No prior cardiovascular genetic testing - + done on family members MYH7 variant - 24 MYH7 variant + what happened with genetic testing? • Cardiologist ordered HCMFirst panel (MYH7 and MYBPC3 genes) with reflex option on patient (clinical rationale below): - Up to 50% of HCM due to a mutation in one of the HCMFirst genes, which represent ~80% of known genetic causes of HCM - Tiered approach: HCMFirst panel reflexes to larger HCMNext panel, only if needed • Positive finding: MYH7 variant, likely pathogenic: p.G584S • This alteration is reported in multiple patients with HCM.1,2,3 MYH7 mutations account for ~40% of HCM and 5-8% of dilated cardiomyopathy (DCM). MYH7 mutations can also cause left ventricular non-compaction (LVNC) and skeletal myopathies, with/ without cardiac involvement.4,5 how did genetic testing help the patient and family? • Confirmed patient to be at risk for HCM and sudden cardiac arrest, despite negative clinical presentation • Tiered -

A Missense Mutation in the RSRSP Stretch of Rbm20 Causes Dilated
www.nature.com/scientificreports OPEN A missense mutation in the RSRSP stretch of Rbm20 causes dilated cardiomyopathy and atrial fbrillation in mice Kensuke Ihara1,2*, Tetsuo Sasano2, Yuichi Hiraoka3, Marina Togo‑Ohno4, Yurie Soejima5, Motoji Sawabe5, Megumi Tsuchiya6, Hidesato Ogawa6, Tetsushi Furukawa1 & Hidehito Kuroyanagi4* Dilated cardiomyopathy (DCM) is a fatal heart disease characterized by left ventricular dilatation and cardiac dysfunction. Recent genetic studies on DCM have identifed causative mutations in over 60 genes, including RBM20, which encodes a regulator of heart‑specifc splicing. DCM patients with RBM20 mutations have been reported to present with more severe cardiac phenotypes, including impaired cardiac function, atrial fbrillation (AF), and ventricular arrhythmias leading to sudden cardiac death, compared to those with mutations in the other genes. An RSRSP stretch of RBM20, a hotspot of missense mutations found in patients with idiopathic DCM, functions as a crucial part of its nuclear localization signals. However, the relationship between mutations in the RSRSP stretch and cardiac phenotypes has never been assessed in an animal model. Here, we show that Rbm20 mutant mice harboring a missense mutation S637A in the RSRSP stretch, mimicking that in a DCM patient, demonstrated severe cardiac dysfunction and spontaneous AF and ventricular arrhythmias mimicking the clinical state in patients. In contrast, Rbm20 mutant mice with frame‑shifting deletion demonstrated less severe phenotypes, although loss of RBM20‑dependent alternative splicing was indistinguishable. RBM20S637A protein cannot be localized to the nuclear speckles, but accumulated in cytoplasmic, perinuclear granule‑like structures in cardiomyocytes, which might contribute to the more severe cardiac phenotypes. Dilated cardiomyopathy (DCM) is a fatal cardiac disease characterized by enlargement of the cardiac chambers and impaired systolic function1. -

Cardiogenetics Testing Reference Guide December 2018
Cardiogenetics Testing reference guide December 2018 Why Choose Ambry More than 1 in 200 people have an inherited cardiovascular condition. Ambry’s mission is to provide the most advanced genetic testing information available to help you identity those at-risk and determine the best treatment options. If we know a patient has a disease-causing genetic change, not only does it mean better disease management, it also indicates that we can test others in the family and provide them with potentially life-saving information. Diseases and Testing Options cardiomyopathies arrhythmias Hypertrophic Cardiomyopathy (HCMNext) Catecholaminergic Polymorphic Ventricular Dilated Cardiomyopathy (DCMNext) Tachycardia (CPVTNext) Arrhythmogenic Right Ventricular Long QT Syndrome, Short QT Syndrome, Cardiomyopathy (ARVCNext) Brugada Syndrome (LongQTNext, RhythmNext) Cardiomyopathies (CMNext, CardioNext) Arrhythmias (RhythmNext, CardioNext) other cardio conditions Transthyretin Amyloidosis (TTR) familial hypercholesterolemia Noonan Syndrome (NoonanNext) and lipid disorders Hereditary Hemorrhagic Telangiectasia Familial Hypercholesterolemia (FHNext) (HHTNext) Sitosterolemia (Sitosterolemia Panel) Comprehensive Lipid Menu thoracic aortic aneurysms (CustomNext-Cardio) and dissections Familial Chylomicronemia Syndrome (FCSNext) Thoracic Aneurysms and Dissections, aortopathies (TAADNext) Marfan Syndrome (TAADNext) Ehlers-Danlos Syndrome (TAADNext) Targeted Panels Gene Comparison ALL PANELS HAVE A TURNAROUND TIME OF 2-3 WEEKS arrhythmias CPVTNext CPVTNext CASQ2,