Improved Hematopoietic Differentiation of Human Pluripotent Stem Cells Via
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Prox1regulates the Subtype-Specific Development of Caudal Ganglionic
The Journal of Neuroscience, September 16, 2015 • 35(37):12869–12889 • 12869 Development/Plasticity/Repair Prox1 Regulates the Subtype-Specific Development of Caudal Ganglionic Eminence-Derived GABAergic Cortical Interneurons X Goichi Miyoshi,1 Allison Young,1 Timothy Petros,1 Theofanis Karayannis,1 Melissa McKenzie Chang,1 Alfonso Lavado,2 Tomohiko Iwano,3 Miho Nakajima,4 Hiroki Taniguchi,5 Z. Josh Huang,5 XNathaniel Heintz,4 Guillermo Oliver,2 Fumio Matsuzaki,3 Robert P. Machold,1 and Gord Fishell1 1Department of Neuroscience and Physiology, NYU Neuroscience Institute, Smilow Research Center, New York University School of Medicine, New York, New York 10016, 2Department of Genetics & Tumor Cell Biology, St. Jude Children’s Research Hospital, Memphis, Tennessee 38105, 3Laboratory for Cell Asymmetry, RIKEN Center for Developmental Biology, Kobe 650-0047, Japan, 4Laboratory of Molecular Biology, Howard Hughes Medical Institute, GENSAT Project, The Rockefeller University, New York, New York 10065, and 5Cold Spring Harbor Laboratory, Cold Spring Harbor, New York 11724 Neurogliaform (RELNϩ) and bipolar (VIPϩ) GABAergic interneurons of the mammalian cerebral cortex provide critical inhibition locally within the superficial layers. While these subtypes are known to originate from the embryonic caudal ganglionic eminence (CGE), the specific genetic programs that direct their positioning, maturation, and integration into the cortical network have not been eluci- dated. Here, we report that in mice expression of the transcription factor Prox1 is selectively maintained in postmitotic CGE-derived cortical interneuron precursors and that loss of Prox1 impairs the integration of these cells into superficial layers. Moreover, Prox1 differentially regulates the postnatal maturation of each specific subtype originating from the CGE (RELN, Calb2/VIP, and VIP). -

Clinical Utility of Recently Identified Diagnostic, Prognostic, And
Modern Pathology (2017) 30, 1338–1366 1338 © 2017 USCAP, Inc All rights reserved 0893-3952/17 $32.00 Clinical utility of recently identified diagnostic, prognostic, and predictive molecular biomarkers in mature B-cell neoplasms Arantza Onaindia1, L Jeffrey Medeiros2 and Keyur P Patel2 1Instituto de Investigacion Marques de Valdecilla (IDIVAL)/Hospital Universitario Marques de Valdecilla, Santander, Spain and 2Department of Hematopathology, MD Anderson Cancer Center, Houston, TX, USA Genomic profiling studies have provided new insights into the pathogenesis of mature B-cell neoplasms and have identified markers with prognostic impact. Recurrent mutations in tumor-suppressor genes (TP53, BIRC3, ATM), and common signaling pathways, such as the B-cell receptor (CD79A, CD79B, CARD11, TCF3, ID3), Toll- like receptor (MYD88), NOTCH (NOTCH1/2), nuclear factor-κB, and mitogen activated kinase signaling, have been identified in B-cell neoplasms. Chronic lymphocytic leukemia/small lymphocytic lymphoma, diffuse large B-cell lymphoma, follicular lymphoma, mantle cell lymphoma, Burkitt lymphoma, Waldenström macroglobulinemia, hairy cell leukemia, and marginal zone lymphomas of splenic, nodal, and extranodal types represent examples of B-cell neoplasms in which novel molecular biomarkers have been discovered in recent years. In addition, ongoing retrospective correlative and prospective outcome studies have resulted in an enhanced understanding of the clinical utility of novel biomarkers. This progress is reflected in the 2016 update of the World Health Organization classification of lymphoid neoplasms, which lists as many as 41 mature B-cell neoplasms (including provisional categories). Consequently, molecular genetic studies are increasingly being applied for the clinical workup of many of these neoplasms. In this review, we focus on the diagnostic, prognostic, and/or therapeutic utility of molecular biomarkers in mature B-cell neoplasms. -

Genome-Wide DNA Methylation Analysis of KRAS Mutant Cell Lines Ben Yi Tew1,5, Joel K
www.nature.com/scientificreports OPEN Genome-wide DNA methylation analysis of KRAS mutant cell lines Ben Yi Tew1,5, Joel K. Durand2,5, Kirsten L. Bryant2, Tikvah K. Hayes2, Sen Peng3, Nhan L. Tran4, Gerald C. Gooden1, David N. Buckley1, Channing J. Der2, Albert S. Baldwin2 ✉ & Bodour Salhia1 ✉ Oncogenic RAS mutations are associated with DNA methylation changes that alter gene expression to drive cancer. Recent studies suggest that DNA methylation changes may be stochastic in nature, while other groups propose distinct signaling pathways responsible for aberrant methylation. Better understanding of DNA methylation events associated with oncogenic KRAS expression could enhance therapeutic approaches. Here we analyzed the basal CpG methylation of 11 KRAS-mutant and dependent pancreatic cancer cell lines and observed strikingly similar methylation patterns. KRAS knockdown resulted in unique methylation changes with limited overlap between each cell line. In KRAS-mutant Pa16C pancreatic cancer cells, while KRAS knockdown resulted in over 8,000 diferentially methylated (DM) CpGs, treatment with the ERK1/2-selective inhibitor SCH772984 showed less than 40 DM CpGs, suggesting that ERK is not a broadly active driver of KRAS-associated DNA methylation. KRAS G12V overexpression in an isogenic lung model reveals >50,600 DM CpGs compared to non-transformed controls. In lung and pancreatic cells, gene ontology analyses of DM promoters show an enrichment for genes involved in diferentiation and development. Taken all together, KRAS-mediated DNA methylation are stochastic and independent of canonical downstream efector signaling. These epigenetically altered genes associated with KRAS expression could represent potential therapeutic targets in KRAS-driven cancer. Activating KRAS mutations can be found in nearly 25 percent of all cancers1. -

At the X-Roads of Sex and Genetics in Pulmonary Arterial Hypertension
G C A T T A C G G C A T genes Review At the X-Roads of Sex and Genetics in Pulmonary Arterial Hypertension Meghan M. Cirulis 1,2,* , Mark W. Dodson 1,2, Lynn M. Brown 1,2, Samuel M. Brown 1,2, Tim Lahm 3,4,5 and Greg Elliott 1,2 1 Division of Pulmonary, Critical Care and Occupational Medicine, University of Utah, Salt Lake City, UT 84132, USA; [email protected] (M.W.D.); [email protected] (L.M.B.); [email protected] (S.M.B.); [email protected] (G.E.) 2 Division of Pulmonary and Critical Care Medicine, Intermountain Medical Center, Salt Lake City, UT 84107, USA 3 Division of Pulmonary, Critical Care, Sleep and Occupational Medicine, Department of Medicine, Indiana University School of Medicine, Indianapolis, IN 46202, USA; [email protected] 4 Richard L. Roudebush Veterans Affairs Medical Center, Indianapolis, IN 46202, USA 5 Department of Anatomy, Cell Biology & Physiology, Indiana University School of Medicine, Indianapolis, IN 46202, USA * Correspondence: [email protected]; Tel.: +1-801-581-7806 Received: 29 September 2020; Accepted: 17 November 2020; Published: 20 November 2020 Abstract: Group 1 pulmonary hypertension (pulmonary arterial hypertension; PAH) is a rare disease characterized by remodeling of the small pulmonary arteries leading to progressive elevation of pulmonary vascular resistance, ultimately leading to right ventricular failure and death. Deleterious mutations in the serine-threonine receptor bone morphogenetic protein receptor 2 (BMPR2; a central mediator of bone morphogenetic protein (BMP) signaling) and female sex are known risk factors for the development of PAH in humans. -

Myt1l Safeguards Neuronal Identity by Actively Repressing Many Non-Neuronal Fates Moritz Mall1, Michael S
LETTER doi:10.1038/nature21722 Myt1l safeguards neuronal identity by actively repressing many non-neuronal fates Moritz Mall1, Michael S. Kareta1†, Soham Chanda1,2, Henrik Ahlenius3, Nicholas Perotti1, Bo Zhou1,2, Sarah D. Grieder1, Xuecai Ge4†, Sienna Drake3, Cheen Euong Ang1, Brandon M. Walker1, Thomas Vierbuchen1†, Daniel R. Fuentes1, Philip Brennecke5†, Kazuhiro R. Nitta6†, Arttu Jolma6, Lars M. Steinmetz5,7, Jussi Taipale6,8, Thomas C. Südhof2 & Marius Wernig1 Normal differentiation and induced reprogramming require human Myt1l (Extended Data Fig. 1). Chromatin immunoprecipita- the activation of target cell programs and silencing of donor cell tion followed by DNA sequencing (ChIP–seq) of endogenous Myt1l programs1,2. In reprogramming, the same factors are often used to in fetal neurons (embryonic day (E) 13.5) and ectopic Myt1l in mouse reprogram many different donor cell types3. As most developmental embryonic fibroblasts (MEFs) two days after induction identified 3,325 repressors, such as RE1-silencing transcription factor (REST) and high-confidence Myt1l peaks that overlapped remarkably well between Groucho (also known as TLE), are considered lineage-specific neurons and MEFs (Fig. 1a, Extended Data Fig. 2, Supplementary repressors4,5, it remains unclear how identical combinations of Table 1). Thus, similar to the pioneer factor Ascl1, Myt1l can access transcription factors can silence so many different donor programs. the majority of its cognate DNA binding sites even in a distantly related Distinct lineage repressors would have to be induced in different cell type. However, unlike Ascl1 targets8, the chromatin at Myt1l donor cell types. Here, by studying the reprogramming of mouse fibroblasts to neurons, we found that the pan neuron-specific a Myt1l Myt1l b Myt1l Ascl1 Random Myt1l 6 Ascl1 + Brn2 endogenous transcription factor Myt1-like (Myt1l) exerts its pro-neuronal Closed 0.030 function by direct repression of many different somatic lineage k programs except the neuronal program. -

An Activin A/BMP2 Chimera, AB215, Blocks Estrogen Signaling Via Induction of ID Proteins in Breast Cancer Cells
Jung et al. BMC Cancer 2014, 14:549 http://www.biomedcentral.com/1471-2407/14/549 RESEARCH ARTICLE Open Access An Activin A/BMP2 chimera, AB215, blocks estrogen signaling via induction of ID proteins in breast cancer cells Jae Woo Jung1,3, Sun Young Shim1, Dong Kun Lee1, Witek Kwiatkowski2 and Senyon Choe1,2* Abstract Background: One in eight women will be affected by breast cancer in her lifetime. Approximately 75% of breast cancers express estrogen receptor alpha (ERα) and/or progesterone receptor and these receptors are markers for tumor dependence on estrogen. Anti-estrogenic drugs such as tamoxifen are commonly used to block estrogen-mediated signaling in breast cancer. However, many patients either do not respond to these therapies (de novo resistance) or develop resistance to them following prolonged treatment (acquired resistance). Therefore, it is imperative to continue efforts aimed at developing new efficient and safe methods of targeting ER activity in breast cancer. Methods: AB215 is a chimeric ligand assembled from sections of Activin A and BMP2. BMP2’sandAB215’s inhibition of breast cancer cells growth was investigated. In vitro luciferase and MTT proliferation assays together with western blot, RT_PCR, and mRNA knockdown methods were used to determine the mechanism of inhibition of estrogen positive breast cancer cells growth by BMP2 and AB215. Additionally in vivo xenograft tumor model was used to investigate anticancer properties of AB215. Results: Here we report that AB215, a chimeric ligand assembled from sections of Activin A and BMP2 with BMP2-like signaling, possesses stronger anti-proliferative effects on ERα positive breast cancer cells than BMP2. -
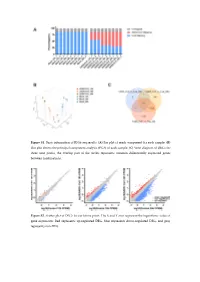
Figure S1. Basic Information of RNA-Seq Results. (A) Bar Plot of Reads Component for Each Sample
Figure S1. Basic information of RNA-seq results. (A) Bar plot of reads component for each sample. (B) Dot plot shows the principal component analysis (PCA) of each sample. (C) Venn diagram of DEGs for three time points, the overlap part of the circles represents common differentially expressed genes between combinations. Figure S2. Scatter plot of DEGs for each time point. The X and Y axes represent the logarithmic value of gene expression. Red represents up-regulated DEG, blue represents down-regulated DEG, and gray represents non-DEG. Table S1. Primers used for quantitative real-time PCR analysis of DEGs. Gene Primer Sequence Forward 5’-CTACGAGTGGATGGTCAAGAGC-3’ FOXO1 Reverse 5’-CCAGTTCCTTCATTCTGCACACG-3’ Forward 5’-GACGTCCGGCATCAGAGAAA-3’ IRS2 Reverse 5’-TCCACGGCTAATCGTCACAG-3’ Forward 5’-CACAACCAGGACCTCACACC-3’ IRS1 Reverse 5’-CTTGGCACGATAGAGAGCGT-3’ Forward 5’-AGGATACCACTCCCAACAGACCT-3’ IL6 Reverse 5’-CAAGTGCATCATCGTTGTTCATAC-3’ Forward 5’-TCACGTTGTACGCAGCTACC-3’ CCL5 Reverse 5’-CAGTCCTCTTACAGCCTTTGG-3’ Forward 5’-CTGTGCAGCCGCAGTGCCTACC-3’ BMP7 Reverse 5’-ATCCCTCCCCACCCCACCATCT-3’ Forward 5’-CTCTCCCCCTCGACTTCTGA-3’ BCL2 Reverse 5’-AGTCACGCGGAACACTTGAT-3’ Forward 5’-CTGTCGAACACAGTGGTACCTG-3’ FGF7 Reverse 5’-CCAACTGCCACTGTCCTGATTTC-3’ Forward 5’-GGGAGCCAAAAGGGTCATCA-3’ GAPDH Reverse 5’-CGTGGACTGTGGTCATGAGT-3’ Supplementary material: Differentially expressed genes log2(SADS-CoV_12h/ Qvalue (SADS-CoV _12h/ Gene Symbol Control_12h) Control_12h) PTGER4 -1.03693 6.79E-04 TMEM72 -3.08132 3.66E-04 IFIT2 -1.02918 2.11E-07 FRAT2 -1.09282 4.66E-05 -

Identifying the Risky SNP of Osteoporosis with ID3-PEP Decision Tree Algorithm
Hindawi Complexity Volume 2017, Article ID 9194801, 8 pages https://doi.org/10.1155/2017/9194801 Research Article Identifying the Risky SNP of Osteoporosis with ID3-PEP Decision Tree Algorithm Jincai Yang,1 Huichao Gu,1 Xingpeng Jiang,1 Qingyang Huang,2 Xiaohua Hu,1 and Xianjun Shen1 1 School of Computer Science, Central China Normal University, Wuhan 430079, China 2School of Life Science, Central China Normal University, Wuhan 430079, China Correspondence should be addressed to Jincai Yang; [email protected] Received 31 March 2017; Revised 26 May 2017; Accepted 8 June 2017; Published 7 August 2017 Academic Editor: Fang-Xiang Wu Copyright © 2017 Jincai Yang et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In the past 20 years, much progress has been made on the genetic analysis of osteoporosis. A number of genes and SNPs associated with osteoporosis have been found through GWAS method. In this paper, we intend to identify the suspected risky SNPs of osteoporosis with computational methods based on the known osteoporosis GWAS-associated SNPs. The process includes two steps. Firstly, we decided whether the genes associated with the suspected risky SNPs are associated with osteoporosis by using random walk algorithm on the PPI network of osteoporosis GWAS-associated genes and the genes associated with the suspected risky SNPs. In order to solve the overfitting problem in ID3 decision tree algorithm, we then classified the SNPs with positive results based on their features of position and function through a simplified classification decision tree which was constructed by ID3 decision tree algorithm with PEP (Pessimistic-Error Pruning). -

Analysis of Estrogen-Regulated Genes in Mouse Uterus Using Cdna Microarray and Laser Capture Microdissection
157 Analysis of estrogen-regulated genes in mouse uterus using cDNA microarray and laser capture microdissection Seok Ho Hong1,2, Hee Young Nah2, Ji Yoon Lee2, Myung Chan Gye1, Chung Hoon Kim2 and Moon Kyoo Kim1 1Department of Life Science, College of Natural Sciences, Hanyang University, Seoul 133-791, Korea 2Department of Obstetrics and Gynecology, College of Medicine, Ulsan University, Asan Medical Center, Seoul 138-746, Korea (Requests for offprints should be addressed toMKKim;Email: [email protected]) Abstract The steroid hormone, estrogen, plays an important role in protein 2, integral membrane protein 2B and chemokine various physiological events which are mediated via its ligand 12. The expression patterns of several selected genes nuclear estrogen receptors, ER and ER. However, the identified by the microarray analysis were confirmed by molecular mechanisms that are regulated by estrogen in RT-PCR. In addition, laser capture microdissection the uterus remain largely unknown. To identify genes that (LCM) was conducted to determine the expression of are regulated by estrogen, the ovariectomized mouse selected genes in specific uterine cell types. Analysis of uterus was exposed to 17-estradiol (E2) for 6 h and 12 h, early and late responsive genes using LCM and cDNA and the data were analyzed by cDNA microarray. The microarray not only suggests direct and indirect effects of present study confirms previous findings and identifies E2 on uterine physiological events, but also demonstrates several genes with expressions not previously known to be differential regulation of E2 in specific uterine cell types. influenced by estrogen. These genes include small proline- These results provide a basic background on global gene rich protein 2A, receptor-activity-modifying protein 3, alterations or genetic pathways in the uterus during the inhibitor of DNA binding-1, eukaryotic translation initia- estrous cycle and the implantation period. -

Xo PANEL DNA GENE LIST
xO PANEL DNA GENE LIST ~1700 gene comprehensive cancer panel enriched for clinically actionable genes with additional biologically relevant genes (at 400 -500x average coverage on tumor) Genes A-C Genes D-F Genes G-I Genes J-L AATK ATAD2B BTG1 CDH7 CREM DACH1 EPHA1 FES G6PC3 HGF IL18RAP JADE1 LMO1 ABCA1 ATF1 BTG2 CDK1 CRHR1 DACH2 EPHA2 FEV G6PD HIF1A IL1R1 JAK1 LMO2 ABCB1 ATM BTG3 CDK10 CRK DAXX EPHA3 FGF1 GAB1 HIF1AN IL1R2 JAK2 LMO7 ABCB11 ATR BTK CDK11A CRKL DBH EPHA4 FGF10 GAB2 HIST1H1E IL1RAP JAK3 LMTK2 ABCB4 ATRX BTRC CDK11B CRLF2 DCC EPHA5 FGF11 GABPA HIST1H3B IL20RA JARID2 LMTK3 ABCC1 AURKA BUB1 CDK12 CRTC1 DCUN1D1 EPHA6 FGF12 GALNT12 HIST1H4E IL20RB JAZF1 LPHN2 ABCC2 AURKB BUB1B CDK13 CRTC2 DCUN1D2 EPHA7 FGF13 GATA1 HLA-A IL21R JMJD1C LPHN3 ABCG1 AURKC BUB3 CDK14 CRTC3 DDB2 EPHA8 FGF14 GATA2 HLA-B IL22RA1 JMJD4 LPP ABCG2 AXIN1 C11orf30 CDK15 CSF1 DDIT3 EPHB1 FGF16 GATA3 HLF IL22RA2 JMJD6 LRP1B ABI1 AXIN2 CACNA1C CDK16 CSF1R DDR1 EPHB2 FGF17 GATA5 HLTF IL23R JMJD7 LRP5 ABL1 AXL CACNA1S CDK17 CSF2RA DDR2 EPHB3 FGF18 GATA6 HMGA1 IL2RA JMJD8 LRP6 ABL2 B2M CACNB2 CDK18 CSF2RB DDX3X EPHB4 FGF19 GDNF HMGA2 IL2RB JUN LRRK2 ACE BABAM1 CADM2 CDK19 CSF3R DDX5 EPHB6 FGF2 GFI1 HMGCR IL2RG JUNB LSM1 ACSL6 BACH1 CALR CDK2 CSK DDX6 EPOR FGF20 GFI1B HNF1A IL3 JUND LTK ACTA2 BACH2 CAMTA1 CDK20 CSNK1D DEK ERBB2 FGF21 GFRA4 HNF1B IL3RA JUP LYL1 ACTC1 BAG4 CAPRIN2 CDK3 CSNK1E DHFR ERBB3 FGF22 GGCX HNRNPA3 IL4R KAT2A LYN ACVR1 BAI3 CARD10 CDK4 CTCF DHH ERBB4 FGF23 GHR HOXA10 IL5RA KAT2B LZTR1 ACVR1B BAP1 CARD11 CDK5 CTCFL DIAPH1 ERCC1 FGF3 GID4 HOXA11 -

Effect of Transcriptional Regulator ID3 on Pulmonary Arterial Hypertension and Hereditary Hemorrhagic Telangiectasia
Florida International University FIU Digital Commons Robert Stempel College of Public Health & Environmental Health Sciences Social Work 7-2019 Effect of Transcriptional Regulator ID3 on Pulmonary Arterial Hypertension and Hereditary Hemorrhagic Telangiectasia Vincent Avecilla Follow this and additional works at: https://digitalcommons.fiu.edu/eoh_fac Part of the Medicine and Health Sciences Commons This work is brought to you for free and open access by the Robert Stempel College of Public Health & Social Work at FIU Digital Commons. It has been accepted for inclusion in Environmental Health Sciences by an authorized administrator of FIU Digital Commons. For more information, please contact [email protected]. Hindawi International Journal of Vascular Medicine Volume 2019, Article ID 2123906, 8 pages https://doi.org/10.1155/2019/2123906 Review Article Effect of Transcriptional Regulator ID3 on Pulmonary Arterial Hypertension and Hereditary Hemorrhagic Telangiectasia Vincent Avecilla 1,2 Department of Environmental Health Sciences, Robert Stempel College of Public Health & Social Work, Florida International University, Miami, FL , USA Celgene Corporation, Summit, NJ , USA Correspondence should be addressed to Vincent Avecilla; [email protected] Received 12 March 2019; Accepted 26 June 2019; Published 11 July 2019 Academic Editor: Robert M. Schainfeld Copyright © 2019 Vincent Avecilla. Tis is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Pulmonary arterial hypertension (PAH) can be discovered in patients who have a loss of function mutation of activin A receptor-like type 1 (ACVRL) gene, a bone morphogenetic protein (BMP) type 1 receptor. -

Intracellular Enhancement of BMP Signaling by LIM-Domain Protein FHL3 Controls Spatiotemporal Emergence of the Neural Crest Driven by WNT Signaling
bioRxiv preprint doi: https://doi.org/10.1101/711192; this version posted July 22, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 1 Intracellular enhancement of BMP signaling by LIM-domain protein FHL3 controls spatiotemporal emergence of the neural crest driven by WNT signaling. Mansour Alkobtawi1,2, Patrick Pla1,2§ and Anne H. Monsoro-Burq1,2,3§*. 1, Univ. Paris Sud, Université Paris Saclay, Centre Universitaire, 15, rue GeorGes Clémenceau, F-91405, Orsay, France. 2, Institut Curie Research Division, PSL Research University, CNRS UMR 3347, INSERM U1021, F-91405, Orsay, France. 3, Institut Universitaire de France, F-75005, Paris. §co-last authors. * correspondence: [email protected] bioRxiv preprint doi: https://doi.org/10.1101/711192; this version posted July 22, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. 2 Abstract How multiple morphoGen siGnals are coordinated in space and time to position key embryonic tissues remains elusive. DurinG neural crest formation, bone morphogenetic protein (BMP), fibroblast Growth factor (FGF) and WNT siGnalinG cooperate by actinG either on the paraxial mesoderm or directly on the neural border ectoderm, but how each tissue interprets this complex information remains poorly understood. Here we show that Fhl3, a scaffold LIM domain protein of previously unknown developmental function, is essential for neural crest formation by linkinG BMP and WNT siGnalinG thereby positioninG the neural crest-inducinG siGnalinG center in the paraxial mesoderm.