Genetic and Neurodevelopmental Spectrum Of
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Translational Regulation of Syngap1 by FMRP Modulates NMDAR Mediated Signalling
bioRxiv preprint doi: https://doi.org/10.1101/345058; this version posted June 26, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Translational regulation of Syngap1 by FMRP modulates NMDAR mediated signalling Abhik Paul1#, Bharti Nawalpuri2#, Shruthi Sateesh1, Ravi S Muddashetty2, James P Clement1* 1 Neuroscience Unit, Jawaharlal Nehru Centre for Advanced Scientific Research, Jakkur, Bangalore 560064. India Phone: +91-80-22082613 2 The Institute for Stem Cell Biology and Regenerative Medicine, GKVK post, Bellary Road, Bangalore 560065. India # Equal contribution * Corresponding author: Email: [email protected] Phone: +91-80-22082613 Number of Pages: 34 including figures Number of Figure: 5 main and 4 extended Figures Number of words for: Abstract:157 Introduction: 452 Discussion: 854 Conflict of Interest: The authors declare no competing financial interests Acknowledgement: This work was supported by grants to JPC by DST-SERB (SB/YS/LS- 215/2013), and to RSM in part, by Dept. of Biotechnology, India (BT/PR8723/AGR/36/776/2013, and BT/IN/Denmark/07/RSM/2015-2016), and intramural funds from both the Institutes. We thank Bhavana Kayyar, Utsa Bhaduri, and Vijay Kumar M J for technical support in our bioinformatics analysis, and Sudhriti Ghosh Dastidar for technical advice on Figure 4. Further, we thank Dr Ravi Manjithaya and his group, and Prof. Kaustuv Sanyal’s group for technical assistance. We also thank Prof MRS Rao, Prof Tapas K Kundu, 1 bioRxiv preprint doi: https://doi.org/10.1101/345058; this version posted June 26, 2018. -

SYNGAP1 Resource Guide
"Building Bridges of Hope to a Cure" An Overview of SYNGAP1 Basic Biology and Clinical Description 1012 14th Street NW, Suite 500 (240) 347-0302 Washington D.C. 20005 www.bridgesyngap.org Production and distribution of this resource are made possible by an educational grant from Building Bridges of Hope to a Cure" About Us 3 Our History 4 SYNGAP1-Normal Function 5 Uniqueness in SYNGAP1 6 Common Symptoms of SYNGAP1 7 Facts about SYNGAP1 Syndrome 8 Common Indications of SYNGAP1 Cognition, development and behavior 9 Characteristics of SYNGAP1 Epilepsy 10 What About CBD? 11 SYNGAP1 Centers of Excellence 12 R e f e r e n c e s 13 References Continued 14 CBD Cited References 15 Connect with Us 16 "Building Bridges of Hope to a Cure" Bridge the Gap – SYNGAP Education and Research Foundation is the leading organization advocating and raising funds for research and treatments for SYNGAP1. The Foundation has its origins in the USA, and now with international outreach, gathers critical information from SYNGAP1 patients worldwide. Bridge the Gap-SYNGAP Education and Research Foundation's mission is to improve the quality of life for people affected by SYNGAP1, provide family support, accelerating research and raising awareness Our Mission To raise awareness and educate the public about SYNGAP1 (MRD5), unite patient families while building a robust data registry and providing meaningful information to researchers. Our Vision To increase the diagnosis rate of SYNGAP1 patients worldwide and provide the expert care, improving the quality of life for our SYNGAP1 community while searching for treatments. Our Goals Increase Diagnosis Rate will improve patient experience and quality of life a standard of care and SYNGAP1 disease profile. -

Endogenous Sirnas and Noncoding RNA-Derived Small Rnas Are Expressed in Adult Mouse Hippocampus and Are Up-Regulated in Olfactory Discrimination Training
Downloaded from rnajournal.cshlp.org on September 27, 2021 - Published by Cold Spring Harbor Laboratory Press Endogenous siRNAs and noncoding RNA-derived small RNAs are expressed in adult mouse hippocampus and are up-regulated in olfactory discrimination training NEIL R. SMALHEISER,1 GIOVANNI LUGLI,1 JYOTHI THIMMAPURAM,2 EDWIN H. COOK,1 and JOHN LARSON1 1Department of Psychiatry, University of Illinois at Chicago, Chicago, Illinois 60612, USA 2W.M. Keck Center for Comparative and Functional Genomics, University of Illinois at Urbana-Champaign, Urbana, Illinois 61801, USA ABSTRACT We previously proposed that endogenous siRNAs may regulate synaptic plasticity and long-term gene expression in the mammalian brain. Here, a hippocampal-dependent task was employed in which adult mice were trained to execute a nose-poke in a port containing one of two simultaneously present odors in order to obtain a reward. Mice demonstrating olfactory discrimination training were compared to pseudo-training and nose-poke control groups; size-selected hippocampal RNA was subjected to Illumina deep sequencing. Sequences that aligned uniquely and exactly to the genome without uncertain nucleotide assignments, within exons or introns of MGI annotated genes, were examined further. The data confirm that small RNAs having features of endogenous siRNAs are expressed in brain; that many of them derive from genes that regulate synaptic plasticity (and have been implicated in neuropsychiatric diseases); and that hairpin-derived endo-siRNAs and the 20- to 23-nt size class of small RNAs show a significant increase during an early stage of training. The most abundant putative siRNAs arose from an intronic inverted repeat within the SynGAP1 locus; this inverted repeat was a substrate for dicer in vitro, and SynGAP1 siRNA was specifically associated with Argonaute proteins in vivo. -

Syngap Families Welcome Packet Welcome
Syngap Families Welcome Packet Welcome We would like to extend you a warm welcome to the Syngap community. Getting a diagnosis is a big step! You finally have answers to the questions you have been asking. It could be that your child is young and you were not expecting the diagnosis… or you could have an older child and are just now finding answers after all these years. Feeling confused or overwhelmed is normal and expected. Please know that things are going to get better and we are here for you. So where do you go from here? Below are several steps we encourage you to take to learn more about Syngap. Given that Syngap is a rare neurological condition there is a great probability that your team of doctors have little or no knowledge about Syngap and how to treat it. You will soon become the Syngap specialist on your loved one’s care team and we are here to give you all the tools you need to be the best advocate. Where to start? Step 1: Basic understanding of Syngap and how it affects your loved one 1. Basic Introduction Syngap1 Syndrome is a rare genetic disorder caused by a mutation on the SYNGAP1 gene. It leads to several neurological issues in Syngap patients. Syngap1 was first diagnosed in 2009 by Dr Michaud at St Justine Hospital in Montreal. 2. Basic Genetics Syngap1 Syndrome is caused by a mutation on the SYNGAP1 gene (6p.21.32).The human body is made of trillions of cells. Each cell contains 23 pairs of chromosomes (46 total). -
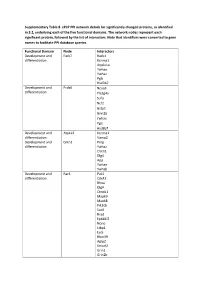
Supplementary Table 8. Cpcp PPI Network Details for Significantly Changed Proteins, As Identified in 3.2, Underlying Each of the Five Functional Domains
Supplementary Table 8. cPCP PPI network details for significantly changed proteins, as identified in 3.2, underlying each of the five functional domains. The network nodes represent each significant protein, followed by the list of interactors. Note that identifiers were converted to gene names to facilitate PPI database queries. Functional Domain Node Interactors Development and Park7 Rack1 differentiation Kcnma1 Atp6v1a Ywhae Ywhaz Pgls Hsd3b7 Development and Prdx6 Ncoa3 differentiation Pla2g4a Sufu Ncf2 Gstp1 Grin2b Ywhae Pgls Hsd3b7 Development and Atp1a2 Kcnma1 differentiation Vamp2 Development and Cntn1 Prnp differentiation Ywhaz Clstn1 Dlg4 App Ywhae Ywhab Development and Rac1 Pak1 differentiation Cdc42 Rhoa Dlg4 Ctnnb1 Mapk9 Mapk8 Pik3cb Sod1 Rrad Epb41l2 Nono Ltbp1 Evi5 Rbm39 Aplp2 Smurf2 Grin1 Grin2b Xiap Chn2 Cav1 Cybb Pgls Ywhae Development and Hbb-b1 Atp5b differentiation Hba Kcnma1 Got1 Aldoa Ywhaz Pgls Hsd3b4 Hsd3b7 Ywhae Development and Myh6 Mybpc3 differentiation Prkce Ywhae Development and Amph Capn2 differentiation Ap2a2 Dnm1 Dnm3 Dnm2 Atp6v1a Ywhab Development and Dnm3 Bin1 differentiation Amph Pacsin1 Grb2 Ywhae Bsn Development and Eef2 Ywhaz differentiation Rpgrip1l Atp6v1a Nphp1 Iqcb1 Ezh2 Ywhae Ywhab Pgls Hsd3b7 Hsd3b4 Development and Gnai1 Dlg4 differentiation Development and Gnao1 Dlg4 differentiation Vamp2 App Ywhae Ywhab Development and Psmd3 Rpgrip1l differentiation Psmd4 Hmga2 Development and Thy1 Syp differentiation Atp6v1a App Ywhae Ywhaz Ywhab Hsd3b7 Hsd3b4 Development and Tubb2a Ywhaz differentiation Nphp4 -

Artificial Intelligence-Based Computational Framework for Drug
Tsuji et al. Alzheimer’s Research & Therapy (2021) 13:92 https://doi.org/10.1186/s13195-021-00826-3 RESEARCH Open Access Artificial intelligence-based computational framework for drug-target prioritization and inference of novel repositionable drugs for Alzheimer’s disease Shingo Tsuji1*† , Takeshi Hase2,3,4,5†, Ayako Yachie-Kinoshita2,4, Taiko Nishino2,SamikGhosh2, Masataka Kikuchi6, Kazuro Shimokawa7, Hiroyuki Aburatani1, Hiroaki Kitano2 and Hiroshi Tanaka3 Abstract Background: Identifying novel therapeutic targets is crucial for the successful development of drugs. However, the cost to experimentally identify therapeutic targets is huge and only approximately 400 genes are targets for FDA-approved drugs. As a result, it is inevitable to develop powerful computational tools that can identify potential novel therapeutic targets. Fortunately, the human protein-protein interaction network (PIN) could be a useful resource to achieve this objective. Methods: In this study, we developed a deep learning-based computational framework that extracts low-dimensional representations of high-dimensional PIN data. Our computational framework uses latent features and state-of-the-art machine learning techniques to infer potential drug target genes. Results: We applied our computational framework to prioritize novel putative target genes for Alzheimer’s disease and successfully identified key genes that may serve as novel therapeutic targets (e.g., DLG4, EGFR, RAC1, SYK, PTK2B, SOCS1). Furthermore, based on these putative targets, we could infer repositionable -

The First International Conference on SYNGAP1-Related Brain Disorders
Weldon et al. Journal of Neurodevelopmental Disorders (2018) 10:6 DOI 10.1186/s11689-018-9225-1 REVIEW Open Access The first international conference on SYNGAP1-related brain disorders: a stakeholder meeting of families, researchers, clinicians, and regulators Monica Weldon1, Murat Kilinc2, J. Lloyd Holder Jr3* and Gavin Rumbaugh2,4* Abstract Background: Pathologic mutations in SYNGAP1 cause a genetically defined form of intellectual disability (ID) with comorbid epilepsy and autistic features. While only recently discovered, pathogenicity of this gene is a relatively frequent genetic cause of classically undefined developmental delay that progresses to ID with commonly occurring comorbidities. Main body: A meeting of 150 people was held that included affected individuals and their caregivers, clinicians that treat this and related brain disorders, neuroscientists that study SYNGAP1 biology or the function of related genes, and representatives from government agencies that fund science and approve new medical treatments. The meeting focused on developing a consensus among all stakeholders as to how best to achieve a more fundamental and profound understanding of SYNGAP1 biology and its role in human disease. Short conclusion: From all of these proceedings, several areas of consensus emerged. The clinicians and geneticists agreed that the prevalence of epilepsy and sensory processing impairments in SYNGAP1-related brain disorders approached 100%. The neurobiologists agreed that more basic research is needed to better understand the molecular and cellular functions of the Syngap1 gene, which will lead to targets for therapeutic intervention. Finally, everyone agreed that there is a pressing need to form a robust patient registry as an initial step toward a prospective natural history study of patients with pathogenic SYNGAP1 variants. -

47421 SAP102 (A7R8L) Rabbit Mab
Revision 1 C 0 2 - t SAP102 (A7R8L) Rabbit mAb a e r o t S Orders: 877-616-CELL (2355) [email protected] 1 Support: 877-678-TECH (8324) 2 4 Web: [email protected] 7 www.cellsignal.com 4 # 3 Trask Lane Danvers Massachusetts 01923 USA For Research Use Only. Not For Use In Diagnostic Procedures. Applications: Reactivity: Sensitivity: MW (kDa): Source/Isotype: UniProt ID: Entrez-Gene Id: WB, IP H M R Endogenous 102 Rabbit Q92796 1741 Product Usage Information 1. Cuthbert, P.C. et al. (2007) J Neurosci 27, 2673-82. 2. Makino, K. et al. (1997) Oncogene 14, 2425-33. Application Dilution 3. Masuko, N. et al. (1999) J Biol Chem 274, 5782-90. 4. Lau, L.F. et al. (1996) J Biol Chem 271, 21622-8. Western Blotting 1:1000 5. Yan, J. et al. (2005) Mol Psychiatry 10, 329-32. Immunoprecipitation 1:50 Storage Supplied in 10 mM sodium HEPES (pH 7.5), 150 mM NaCl, 100 µg/ml BSA, 50% glycerol and less than 0.02% sodium azide. Store at –20°C. Do not aliquot the antibody. Specificity / Sensitivity SAP102 (A7R8L) Rabbit mAb recognizes endogenous levels of total SAP102 protein. Species Reactivity: Human, Mouse, Rat Source / Purification Monoclonal antibody is produced by immunizing animals with a synthetic peptide corresponding to residues near the carboxy terminus of human SAP102 protein. Background Synapse-associated protein 102 (SAP102, DLG3) belongs to the membrane-associated guanylate kinase (MAGUK) protein family and is a homolog of the Drosophila disc large (dlg) tumor suppressor protein. SAP102 consists of three PDZ domains, a Src homology 3 (SH3) domain, and a guanylate kinase (GK) domain (1). -

Characterization of Intellectual Disability and Autism Comorbidity Through Gene Panel Sequencing
bioRxiv preprint doi: https://doi.org/10.1101/545772; this version posted February 10, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Characterization of Intellectual disability and Autism comorbidity through gene panel sequencing Maria Cristina Aspromonte 1, 2, Mariagrazia Bellini 1, 2, Alessandra Gasparini 3, Marco Carraro 3, Elisa Bettella 1, 2, Roberta Polli 1, 2, Federica Cesca 1, 2, Stefania Bigoni 4, Stefania Boni 5, Ombretta Carlet 6, Susanna Negrin 6, Isabella Mammi 7, Donatella Milani 8 , Angela Peron 9, 10, Stefano Sartori 11, Irene Toldo 11, Fiorenza Soli 12, Licia Turolla 13, Franco Stanzial 14, Francesco Benedicenti 14, Cristina Marino-Buslje 15, Silvio C.E. Tosatto 3, 16, Alessandra Murgia 1, 2, Emanuela Leonardi 1, 2 1. Molecular Genetics of Neurodevelopment, Dept. of Woman and Child Health, University of Padova, Padova, Italy 2. Fondazione Istituto di Ricerca Pediatrica (IRP), Città della Speranza, Padova, Italy 3. Dept. of Biomedical Sciences, University of Padova, Padova, Italy 4. Medical Genetics Unit, Ospedale Universitario S. Anna, Ferrara, Italy 5. Medical Genetics Unit, S. Martino Hospital, Belluno, Italy 6. Child Neuropsychiatry Unit, IRCCS Eugenio Medea, Conegliano, Italy 7. Medical Genetics Unit, Dolo General Hospital, Venezia, Italy 8. Pediatric Highly Intensive Care Unit, Department of Pathophysiology and Transplantation, University of Milano, Fondazione IRCCS, Ca' Granda Ospedale Maggiore Policlinico, Milan, Italy 9. Child Neuropsychiatry Unit, Epilepsy Center, Santi Paolo-Carlo Hospital, Dept. of Health Sciences, University of Milano, Milano, Italy 10. Department of Pediatrics, Division of Medical Genetics, University of Utah School of Medicine, Salt Lake City, UT, USA 11. -

Twenty Years of Syngap Research: from Synapses to Cognition
1596 • The Journal of Neuroscience, February 19, 2020 • 40(8):1596–1605 Review Twenty Years of SynGAP Research: From Synapses to Cognition X Timothy R. Gamache,1,2 XYoichi Araki,1,2 and XRichard L. Huganir1,2 1Solomon H. Snyder Department of Neuroscience, and 2Kavli Neuroscience Discovery Institute, Johns Hopkins University, Baltimore, Maryland 21205 SynGAP is a potent regulator of biochemical signaling in neurons and plays critical roles in neuronal function. It was first identified in 1998, and has since been extensively characterized as a mediator of synaptic plasticity. Because of its involvement in synaptic plasticity, SynGAP has emergedasacriticalproteinfornormalcognitivefunction.Inrecentyears,mutationsintheSYNGAP1genehavebeenshowntocauseintellectual disabilityinhumansandhavebeenlinkedtootherneurodevelopmentaldisorders,suchasautismspectrumdisordersandschizophrenia.While the structure and biochemical function of SynGAP have been well characterized, a unified understanding of the various roles of SynGAP at the synapse and its contributions to neuronal function remains to be achieved. In this review, we summarize and discuss the current understanding of the multifactorial role of SynGAP in regulating neuronal function gathered over the last two decades. Introduction SynGAP structure and function SynGAP was first identified, cloned, and characterized in 1998 by SynGAP is an exceedingly abundant constituent of the PSD. In- two independent laboratories (Chen et al., 1998; Kim et al., deed, quantitative proteomic analyses have revealed SynGAP to 1998). One study identified SynGAP following a yeast two-hybrid be one of the most highly abundant proteins in the PSD, reaching screen for novel PDZ-interacting proteins. The screen specifically copy numbers that are surpassed only by CaMKII␣ and the identified proteins from a hippocampal cDNA library that inter- PSD-95 family proteins (Sugiyama et al., 2005; Cheng et al., 2006; act with the third PDZ domain of SAP102, a member of the Sheng and Kim, 2011). -

LGI1–ADAM22–MAGUK Configures Transsynaptic Nanoalignment for Synaptic Transmission and Epilepsy Prevention
LGI1–ADAM22–MAGUK configures transsynaptic nanoalignment for synaptic transmission and epilepsy prevention Yuko Fukataa,b,1, Xiumin Chenc,d,1, Satomi Chikenb,e, Yoko Hiranoa,f, Atsushi Yamagatag, Hiroki Inahashia, Makoto Sanboh, Hiromi Sanob,e, Teppei Gotoh, Masumi Hirabayashib,h, Hans-Christian Kornaui,j, Harald Prüssi,k, Atsushi Nambub,e, Shuya Fukail, Roger A. Nicollc,d,2, and Masaki Fukataa,b,2 aDivision of Membrane Physiology, Department of Molecular and Cellular Physiology, National Institute for Physiological Sciences, National Institutes of Natural Sciences, Aichi 444-8787, Japan; bDepartment of Physiological Sciences, School of Life Science, SOKENDAI (The Graduate University for Advanced Studies), Aichi 444-8585, Japan; cDepartment of Cellular and Molecular Pharmacology, University of California, San Francisco, CA 94158; dDepartment of Physiology, University of California, San Francisco, CA 94158; eDivision of System Neurophysiology, Department of System Neuroscience, National Institute for Physiological Sciences, National Institutes of Natural Sciences, Aichi 444-8585, Japan; fDepartment of Pediatrics, Graduate School of Medicine, The University of Tokyo, Tokyo 113-8655, Japan; gLaboratory for Protein Functional and Structural Biology, RIKEN Center for Biosystems Dynamics Research, Kanagawa 230-0045, Japan; hCenter for Genetic Analysis of Behavior, National Institute for Physiological Sciences, National Institutes of Natural Sciences, Okazaki 444-8787, Japan; iGerman Center for Neurodegenerative Diseases (DZNE), 10117 Berlin, Germany; jNeuroscience Research Center, Cluster NeuroCure, Charité-Universitätsmedizin Berlin, 10117 Berlin, Germany; kDepartment of Neurology and Experimental Neurology, Charité-Universitätsmedizin Berlin, 10117 Berlin, Germany; and lDepartment of Chemistry, Graduate School of Science, Kyoto University, 606-8502 Kyoto, Japan Contributed by Roger A. Nicoll, December 1, 2020 (sent for review October 29, 2020; reviewed by David S. -

Tiratha Raj Singh.Pdf
GENE-40307; No. of pages: 10; 4C: Gene xxx (2015) xxx–xxx Contents lists available at ScienceDirect Gene journal homepage: www.elsevier.com/locate/gene 1Q1 Unc-51 like kinase 1 (ULK1) in silico analysis for biomarker 2 identification: A vital component of autophagy 3Q2 Rohit Randhawa a, Manika Sehgal a, Tiratha Raj Singh a, Ajay Duseja b, Harish Changotra a,⁎ 4 a Department of Biotechnology and Bioinformatics, Jaypee University of Information Technology, Waknaghat, Solan 1732 34 Himachal Pradesh, India 5 b Department of Hepatology, Postgraduate Institute of Medical Education and Research, Chandigarh 160 012, India 6 article info abstract 7 Article history: Autophagy is a degradation pathway involving lysosomal machinery for degradation of damaged organelles like 19 8 Received 19 October 2014 the endoplasmic reticulum and mitochondria into their building blocks to maintain homeostasis within the cell. 20 9 Received in revised form 3 February 2015 ULK1, a serine/threonine kinase, is conserved across species, from yeasts to mammals, and plays a central role in 21 10 Accepted 5 February 2015 autophagy pathway. It receives signals from upstream modulators such as TIP60, mTOR and AMPK and relays 22 11 Available online xxxx them to its downstream substrates like Ambra1 and ZIP kinase. The activity of this complex is regulated through 23 – fi fi 24 12 Keywords: protein protein interactions and post-translational modi cations. Applying in silico analysis we identi ed 25 13Q3 Autophagy (i) conserved patterns of ULK1 that showed its evolutionary relationship between the species which were closely 14 ULK1 related in a family compared to others.