PDF (Appendix 2: the Cope Rearrangement)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Download Answer
Question #72514, Chemistry / Organic Chemistry Explain the electrocyclic and sigmatropic reaction in detail. Answer: In organic chemistry, an electrocyclic reaction is a type of pericyclic rearrangement where the net result is one pi bond being converted into one sigma bond or vice versa. The Diels-Alder reaction is a common example. The especially interesting thing about this reaction is its stereochemistry. When performing an electrocyclic reaction, it is often desirable to predict the cis/trans geometry of the reaction's product. The first step in this process is to determine whether a reaction proceeds through conrotation or disrotation. Reactions can be either photochemical or thermal and their mexanisms are different. Mechanism of thermal reactions: Correlation diagrams, which connect the molecular orbitals of the reactant to those of the product having the same symmetry, can then be constructed for the two processes. Mechanism of photochemical reactions: If the ring opening of 3,4-dimethylcyclobutene were carried out under photochemical conditions the resulting electrocyclization would be occur via a disrotatory mode instead of a conrotatory mode as can be seen by the correlation diagram for the allowed excited state ring opening reaction. A sigmatropic reaction in organic chemistry is a pericyclic reaction wherein the net result is one σ-bond is changed to another σ-bond in an uncatalyzed intramolecular process. The most well-known of the sigmatropic rearrangements are the [3,3] Cope rearrangement, Claisen rearrangement, Carroll rearrangement [3,3] sigmatropic shifts are well studied sigmatropic rearrangements. The Woodward– Hoffman rules predict that these six electron reactions would proceed suprafacially, via a Huckel topology transition state. -

Strategic Applications of Tandem Reactions in Complex Natural Product Synthesis
Strategic Applications of Tandem Reactions in Complex Natural Product Synthesis: Rapid Access to the (Iso)Cyclocitrinol Core Christopher W. Plummer Submitted in partial fulfillment of the Requirement for the degree of Doctor of Philosophy in the Graduate School of Arts and Sciences Columbia University 2012 © 2012 Christopher Wainwright Plummer All Rights Reserved Abstract Strategic Applications of Tandem Reactions in Complex Natural Product Synthesis: Rapid Access to the (Iso)Cyclocitrinol Core Christopher W. Plummer This thesis describes the efforts of Professor James Leighton and myself toward the synthesis of the tetracyclic core of a class of steroidal natural products known as the cyclocitrinols. Our initial work in this area was performed on racemic model systems in order to validate our ring contraction-Cope rearrangement strategy. Novel chemistry was then identified to access the functionalized core in enantio-enriched form. Finally, in line with our efforts to probe the transition state of our key tandem Claisen-Cope reaction, additional substrates were prepared supporting our proposed transition state and improving the efficiency of this transformation. Table of Contents Chapter I. Background………………………………………………………....……………1 1.1: Isolation and Characterization of the Cyclocitrinols 1 1.2: Biosynthesis and Previous Synthetic Approach 2 1.3: Retrosynthetic Analysis 5 1.4: Previous Synthetic Efforts in the Leighton Laboratory 9 Chapter II. Model Studies……………………………………………………………….….13 2.1: 1st Generation Macrolactone Approach 13 2.2: -

Hydrogen Bond-Directed Stereospecific Interactions in (A) General Synthesis of Chiral Vicinal Diamines and (B) Generation of Helical Chirality with Amino Acids
HYDROGEN BOND-DIRECTED STEREOSPECIFIC INTERACTIONS IN (A) GENERAL SYNTHESIS OF CHIRAL VICINAL DIAMINES AND (B) GENERATION OF HELICAL CHIRALITY WITH AMINO ACIDS by Hyunwoo Kim A thesis submitted in conformity with the requirements for the degree of Doctor of Philosophy Graduate Department of Chemistry University of Toronto © Copyright by Hyunwoo Kim, 2009 Hydrogen Bond-Directed Stereospecific Interactions in (A) General Synthesis of Chiral Vicinal Diamines and (B) Generation of Helical Chirality with Amino Acids Hyunwoo Kim Doctor of Philosophy 2009 Department of Chemistry University of Toronto ABSTRACT Hydrogen bonding interactions have been applied to the synthesis of chiral vicinal diamines and the generation of helical chirality. A stereospecific synthesis of vicinal diamines was developed by using the diaza-Cope rearrangement reaction driven by resonance-assisted hydrogen bonds (RAHBs). This process for making a wide variety of chiral diamines requires only a single starting chiral diamine, 1,2-bis(2-hydroxyphenyl)-1,2-diaminoethane (HPEN) and aldehydes. Experimental and computational studies reveal that this process provides one of the simplest and most versatile approaches to preparing chiral vicinal diamines including not only C2 symmetric diaryl and dialkyl diamines but also unsymmetrical alkyl-aryl and aryl-aryl diamines with excellent yields and enantiopurities. Weak forces affecting kinetics and thermodynamics of the diaza-Cope rearrangement were systematically studied by combining experimental and computational approaches. These forces include hydrogen bonding effects, electronic effects, steric effects, and oxyanion effects. As an example of tuning diamine catalysts, a vicinal diamine-catalyzed synthesis of warfarin is described. Detailed mechanistic studies lead to a new mechanism involving diimine intermediates. -

R. B. Woodward Precipitation of Barium in the Copper-Tin Group Of
HETEROCYCLES, Vol. 7, No. 1. 1977 R. B. Woodward Precipitation of barium in the copper-tin group of qualitative analysis. W.J.Hal1 and RBW, -Ind. Eng. Chem., Anal. Ed., 6, 478 (1934). A new pressure regulator for vacuum distillation. R.L.Emerson and RBW, g.,2, 347 (1937). The Staling of coffee. II. S.C.Prescott, R.L.Emerson, RBW, and R. Heggie, Food Re- --search, 2, 165 (1937). Pyrolysis of organomagnesium compounds. I. A new agent for the reduction of bemophenone. D.B.Clapp, and RBW, 1. ---Am. Chem. Soc., -60, 1019 (1938). The direct introduction of the angular methyl group. RBW, Ibid., 62, 1208 (1940). Experiments on the synthesis of oestrone. I. 2-(8-phenylethy1)-furans as components in the diene synthesis. RBW, Ibid., 62, 1478 (1940). The formation of Reissert's compounds in non-aqueous media. RBW, Ibid., 62, 1626 (1940). A new optically active reagent for carbonyl compounds. The resolution of -dl-camphor. RBW, T. P. Kohman, and G. C. Harris, Ibid., 63, lu) (1941). The isolation and properties of 1, 1-dineopentylethylene, a component of triisobutylene. P.D.Bartlett, G.L. Fraser, and RBW, x.,3, 495 (1941). Structure and absorption spectra of a,p-unsaturated ketones. RBW, Ibid., 3, 1123 (1941). 5 Structure and absorption spectra. II. 3-Acetoxy-A -(6)-~cholestene-7-carboxylicacid. RBW, and A.F.Clifiord, -Ibid., -63, 2727 (1941). The structure of cantharidine and the synthesis of desoxycantharidine. RLW, and R.B. Loftfield, Ibid., 3, 3167 (1941). -t-Butyllithium. P.D.Bardett, C .G.Swain, and RBW, --Ibid., 63, 3229 (1941). -

Anion Relay Cyclopropanation and Aryl Vinyl Cyclopropane Cope Rearrangements By
Anion Relay Cyclopropanation and Aryl Vinyl Cyclopropane Cope Rearrangements By Kevin Michael Allegre B.Sc., The University of Kansas, 2013 Submitted to the graduate degree program in Chemistry and the Graduate Faculty of the University of Kansas in partial fulfillment of the requirements for the degree of Doctor of Philosophy. Chair: Jon A. Tunge Paul R. Hanson Helena Malinakova Michael Rubin Ryan A. Altman Date Defended: 18 July 2019 The dissertation committee for Kevin M. Allegre certifies that this is the approved version of the following dissertation: Anion Relay Cyclopropanation and Aryl Vinyl Cyclopropane Cope Rearrangements Chair: Jon A. Tunge Date Approved: 24 July 2019 ii Abstract Kevin M. Allegre and Jon A. Tunge Department of Chemistry, July 2019 University of Kansas Anion Relay Chemistry is a powerful tool for the rapid development of molecular complexity in an operationally simple manner. Much of the work in this field has been pioneered and developed by the Smith group, whose work has primarily focused on silicon and phosphorus Brook rearrangements to effect anion relay. Presented herein is the development of a retro-Claisen condensation protocol to effect anion relay in the synthesis of vinyl cyclopropanes, and subsequent aromatic Cope rearrangement of those vinyl cyclopropanes. This protocol provides a supplementary method of anion relay utilizing readily accessible nucleophiles, which obviates the need for synthesis of alkyl silanes or phosphines as starting materials. Chapter 1 is a review of anion relay chemistry, which focuses on through-space anion relay over 3 or more bonds. It covers both new developments and applications to total synthesis of through-space anion relay more than three bonds since the field was last reviewed by Smith in 2008. -

Robert Burns Woodward 1917–1979
NATIONAL ACADEMY OF SCIENCES ROBERT BURNS WOODWARD 1917–1979 A Biographical Memoir by ELKAN BLOUT Any opinions expressed in this memoir are those of the author and do not necessarily reflect the views of the National Academy of Sciences. Biographical Memoirs, VOLUME 80 PUBLISHED 2001 BY THE NATIONAL ACADEMY PRESS WASHINGTON, D.C. ROBERT BURNS WOODWARD April 10, 1917–July 8, 1979 BY ELKAN BLOUT OBERT BURNS WOODWARD was the preeminent organic chemist Rof the twentieth century. This opinion is shared by his colleagues, students, and by other distinguished chemists. Bob Woodward was born in Boston, Massachusetts, and was an only child. His father died when Bob was less than two years old, and his mother had to work hard to support her son. His early education was in the Quincy, Massachusetts, public schools. During this period he was allowed to skip three years, thus enabling him to finish grammar and high schools in nine years. In 1933 at the age of 16, Bob Woodward enrolled in the Massachusetts Institute of Technology to study chemistry, although he also had interests at that time in mathematics, literature, and architecture. His unusual talents were soon apparent to the MIT faculty, and his needs for individual study and intensive effort were met and encouraged. Bob did not disappoint his MIT teachers. He received his B.S. degree in 1936 and completed his doctorate in the spring of 1937, at which time he was only 20 years of age. Immediately following his graduation Bob taught summer school at the University of Illinois, but then returned to Harvard’s Department of Chemistry to start a productive period with an assistantship under Professor E. -
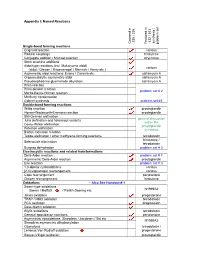
Appendix I: Named Reactions Single-Bond Forming Reactions Co
Appendix I: Named Reactions 235 / 335 432 / 533 synthesis / / synthesis Covered in Covered Featured in problem set problem Single-bond forming reactions Grignard reaction various Radical couplings hirstutene Conjugate addition / Michael reaction strychnine Stork enamine additions Aldol-type reactions (incl. Mukaiyama aldol) various (aldol / Claisen / Knoevenagel / Mannich / Henry etc.) Asymmetric aldol reactions: Evans / Carreira etc. saframycin A Organocatalytic asymmetric aldol saframycin A Pseudoephedrine glycinamide alkylation saframycin A Prins reaction Prins-pinacol reaction problem set # 2 Morita-Baylis-Hillman reaction McMurry condensation Gabriel synthesis problem set #3 Double-bond forming reactions Wittig reaction prostaglandin Horner-Wadsworth-Emmons reaction prostaglandin Still-Gennari olefination general discussion Julia olefination and heteroaryl variants within the Corey-Winter olefination prostaglandin Peterson olefination synthesis Barton extrusion reaction Tebbe olefination / other methylene-forming reactions tetrodotoxin hirstutene / Selenoxide elimination tetrodotoxin Burgess dehydration problem set # 3 Electrocyclic reactions and related transformations Diels-Alder reaction problem set # 1 Asymmetric Diels-Alder reaction prostaglandin Ene reaction problem set # 3 1,3-dipolar cycloadditions various [2,3] sigmatropic rearrangement various Cope rearrangement periplanone Claisen rearrangement hirstutene Oxidations – Also See Handout # 1 Swern-type oxidations (Swern / Moffatt / Parikh-Doering etc. N1999A2 Jones oxidation -

Elimination Reactions Are Described
Introduction In this module, different types of elimination reactions are described. From a practical standpoint, elimination reactions widely used for the generation of double and triple bonds in compounds from a saturated precursor molecule. The presence of a good leaving group is a prerequisite in most elimination reactions. Traditional classification of elimination reactions, in terms of the molecularity of the reaction is employed. How the changes in the nature of the substrate as well as reaction conditions affect the mechanism of elimination are subsequently discussed. The stereochemical requirements for elimination in a given substrate and its consequence in the product stereochemistry is emphasized. ELIMINATION REACTIONS Objective and Outline beta-eliminations E1, E2 and E1cB mechanisms Stereochemical considerations of these reactions Examples of E1, E2 and E1cB reactions Alpha eliminations and generation of carbene I. Basics Elimination reactions involve the loss of fragments or groups from a molecule to generate multiple bonds. A generalized equation is shown below for 1,2-elimination wherein the X and Y from two adjacent carbon atoms are removed, elimination C C C C -XY X Y Three major types of elimination reactions are: α-elimination: two atoms or groups are removed from the same atom. It is also known as 1,1-elimination. H R R C X C + HX R Both H and X are removed from carbon atom here R Carbene β-elimination: loss of atoms or groups on adjacent atoms. It is also H H known as 1,2- elimination. R C C R R HC CH R X H γ-elimination: loss of atoms or groups from the 1st and 3rd positions as shown below. -

2 Results and Discussion
22 2 Results and Discussion 2 Results and Discussion 2.1 Syntheses of Azoninones - Unsaturated Nine-Membered Ring Lactams 2.1.1 Syntheses of Vinyl pyrrolidines N-Vinyl pyrrolidines are important reactants for the preparation of azoninones by zwitterionic aza- Claisen rearrangement. They can be obtained by derivatisation reactions starting from chiral molecules such as L-proline and 2S,4R-4-hydroxyproline or by metal catalysed cyclisation reactions of N- substituted allenic amines.66 Since large amounts of vinyl pyrrolidines have to be synthetically available for the systematic examination of the aza-Claisen rearrangement and its application in a total synthesis, especially the ex-chiral pool synthesis of these precursors has been extensively investigated by our group.67 The following two schemes represent the synthesis of the vinyl pyrrolidines [6] and [11] that have been used in this work as precursors for the aza-Claisen rearrangement. This synthesis allows the preparation of 50-100g of vinyl pyrrolidines. HO HO HO BnCl, Et3N TBSCl, imid. AcCl, MeOH CH Cl CH2Cl2 2 2 N COOH CO Me 100% N CO2Me 88% N 2 95% H H Bn [1] [2] [3] TBSO TBSO 1) Oxalyl chloride TBSO DMSO, Et3N, CH2Cl2 DIBALH, THF 2) Ph3P=CH2, THF N CO2Me N 89% N 70% Bn Bn OH Bn [4] [5] [6] Scheme 19 Synthesis of vinyl pyrrolidine [6] Allylamine [6] was efficiently generated via a six-step sequence starting from trans-4-hydroxy-L-(-)- proline [1] the overall yield was about 50% (Scheme 19). After esterification,68 the N-benzyl group 66 (a) Huby, N. -

Synthesis and Reactions of 4,5-Homotropone and 4,5-Dimethylenetropone Richard Anthony Fugiel Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 1974 Synthesis and reactions of 4,5-homotropone and 4,5-dimethylenetropone Richard Anthony Fugiel Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Organic Chemistry Commons Recommended Citation Fugiel, Richard Anthony, "Synthesis and reactions of 4,5-homotropone and 4,5-dimethylenetropone " (1974). Retrospective Theses and Dissertations. 5986. https://lib.dr.iastate.edu/rtd/5986 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. INFORIVIATIOIM TO USERS This material was produced from a microfilm copy of the original document. While the most advanced technological means to photograph and reproduce this document have been used, the quality is heavily dependent upon the quality of the original submitted. The following explanation of techniques is provided to help you understand markings or patterns which may appear on this reproduction. 1.The sign or "target" for pages apparently lacking from the document photographed is "Missing Page(s)". If it was possible to obtain the missing page(s) or section, they are spliced into the film along with adjacent pages. This may have necessitated cutting thru an image and duplicating adjacent pages to insure you complete continuity. 2. When an image on the film is obliterated with a large round black mark, it is an indication that the photographer suspected that the copy may have moved during exposure and thus cause a blurred image. -

University of California
UC Riverside UC Riverside Electronic Theses and Dissertations Title Towards a Catalytic Asymmetric Cope Rearrangement and the Synthesis and Self-Assembly of Metal-Coordinated Hosts Permalink https://escholarship.org/uc/item/4gc654st Author Moehlig, Melissa Padilla Publication Date 2013 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA RIVERSIDE Towards a Catalytic Asymmetric Cope Rearrangement and the Synthesis and Self- Assembly of Metal-Coordinated Hosts A Dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Chemistry by Melissa Padilla Moehlig December 2013 Dissertation Committee: Dr. Richard J. Hooley, Chairperson Dr. Catharine H. Larsen Dr. Michael C. Pirrung Copyright by Melissa Padilla Moehlig 2013 The Dissertation of Melissa Padilla Moehlig is approved: Committee Chairperson University of California, Riverside ACKNOWLEDGEMENTS Graduate school has been one of the most rewarding and yet the most exhausting and stressful times of my life. It would not have survived without the help of several people. I would like to thank Dr. Courtney Meyet, Dr. Katherine Djernes, and Yoo-Jin Ghang for their friendship and all the laughs that were necessary to keep me sane. I would like to thank Michael Young, Hou Ung, and Jay-Ar Bendo for our morning coffee breaks, they were crucial to get my day started. I would like to thank Prof. Larsen for teaching me to be independent. I would like to thank Prof. Hooley for all his guidance over the past five years. You are truly a great mentor and I don’t think I would have survived graduate school without your help and advice. -

Pericyclic Reactions Notes
- 1 - PERICYCLIC REACTIONS NOTES Pericyclic reactions cannot be treated adequately by “curly-arrow” formalisms and a knowledge of molecular orbital theory is crucial to their understanding. They are reactions in which “all first order changes in bonding relationships takes place in concert on a closed curve” (Woodward & Hoffmann). More simply, the term “pericyclic” covers all concerted reactions involving a cyclic flow of electrons through a single transition state. Pericyclic reactions can be predicted and controlled to a great degree, which makes them very useful in synthesis. There are broadly four classes of pericyclic reaction: Sigmatropic – These are unimolecular isomerisations, and involve the movement of a σ-bond from one position to another. An illustration would be the first step of the Claisen Rearrangement: Note the nomenclature of this reaction, being described as a [i,j] shift. For example, this following is a [1,7] shift: Electrocyclic – These are unimolecular. They are characterised by ring opening or closing with a σ- bond forming at one end. Ring closing is more common, since this is formation of a σ-bond at the expense of a π-bond, but ring strain can lead to opening. Two examples are: Cycloaddition – This is the largest class of pericyclic reaction. It is characterised by two fragments coming together to form two new σ-bonds in a ring. Some examples are Diels-Alder and Ozonolysis reactions, which are described below. Chelotropic reactions are a specific type of cycloaddition, where the two bonds are made or broken at the same atom. The classic example of this is carbene addition to a double bond.