Seedling Emergence Patterns
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Plant Physiology
PLANT PHYSIOLOGY Vince Ördög Created by XMLmind XSL-FO Converter. PLANT PHYSIOLOGY Vince Ördög Publication date 2011 Created by XMLmind XSL-FO Converter. Table of Contents Cover .................................................................................................................................................. v 1. Preface ............................................................................................................................................ 1 2. Water and nutrients in plant ............................................................................................................ 2 1. Water balance of plant .......................................................................................................... 2 1.1. Water potential ......................................................................................................... 3 1.2. Absorption by roots .................................................................................................. 6 1.3. Transport through the xylem .................................................................................... 8 1.4. Transpiration ............................................................................................................. 9 1.5. Plant water status .................................................................................................... 11 1.6. Influence of extreme water supply .......................................................................... 12 2. Nutrient supply of plant ..................................................................................................... -

Botany for Gardeners Offers a Clear Explanation of How Plants Grow
BotGar_Cover (5-8-2004) 11/8/04 11:18 AM Page 1 $19.95/ £14.99 GARDENING & HORTICULTURE/Reference Botany for Gardeners offers a clear explanation of how plants grow. • What happens inside a seed after it is planted? Botany for Gardeners Botany • How are plants structured? • How do plants adapt to their environment? • How is water transported from soil to leaves? • Why are minerals, air, and light important for healthy plant growth? • How do plants reproduce? The answers to these and other questions about complex plant processes, written in everyday language, allow gardeners and horticulturists to understand plants “from the plant’s point of view.” A bestseller since its debut in 1990, Botany for Gardeners has now been expanded and updated, and includes an appendix on plant taxonomy and a comprehensive index. Twodozen new photos and illustrations Botany for Gardeners make this new edition even more attractive than its predecessor. REVISED EDITION Brian Capon received a ph.d. in botany Brian Capon from the University of Chicago and was for thirty years professor of botany at California State University, Los Angeles. He is the author of Plant Survival: Adapting to a Hostile Brian World, also published by Timber Press. Author photo by Dan Terwilliger. Capon For details on other Timber Press books or to receive our catalog, please visit our Web site, www.timberpress.com. In the United States and Canada you may also reach us at 1-800-327-5680, and in the United Kingdom at [email protected]. ISBN 0-88192-655-8 ISBN 0-88192-655-8 90000 TIMBER PRESS 0 08819 26558 0 9 780881 926552 UPC EAN 001-033_Botany 11/8/04 11:20 AM Page 1 Botany for Gardeners 001-033_Botany 11/8/04 11:21 AM Page 2 001-033_Botany 11/8/04 11:21 AM Page 3 Botany for Gardeners Revised Edition Written and Illustrated by BRIAN CAPON TIMBER PRESS Portland * Cambridge 001-033_Botany 11/8/04 11:21 AM Page 4 Cover photographs by the author. -
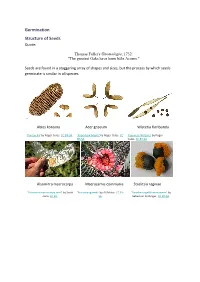
Germination Structure of Seeds Quote
Germination Structure of Seeds Quote: Thomas Fuller's Gnomologia, 1732: "The greatest Oaks have been little Acorns." Seeds are found in a staggering array of shapes and sizes, but the process by which seeds germinate is similar in all species. Abies koreana Acer griseum Wisteria floribunda 'Korean Fir' by Roger Culos. CC BY-SA. 'Paperbark Maple' by Roger Culos. CC 'Japanese Wisteria' by Roger BY-SA. Culos. CC BY-SA. Alsomitra macrocarpa Macrozamia communis Strelitzia reginae 'Alsomitra macrocarpa seed' by Scott 'BurrawangSeeds' by AYArktos. CC BY- 'Paradiesvogelblumensamen' by Zona. CC BY. SA. Sebastian Stabinger. CC BY-SA. Taraxicum officinale Stephanotis floribunda Phleum pratense 'Achane of Taraxacum sect. 'Stephanotis seed' by L. Marie"/Lenore 'Timoteegras vruchten Phleum Ruderalia' by Didier Edman, Sunnyvale, CA. CC BY. pratense' by Rasbak. CC BY-SA. Descouens. CC BY-SA. Dicotyledon seeds testa epicotyl plumule hypocotyl cotyledon radicle 'Aesculus hippocastanum seed section' by Boronian. CC BY. plumule epicotyl hypocotyl testa hilum radicle cotyledon micropyle endosperm Monocotyledon seeds endosperm epicotyl testa hypocotyl cotyledon radicle Parts of a seed Testa The seed coat. A protective layer which is tough and hard and it protects the seed from attack by insects, fungi and bacteria. Cotyledon Dicotyledons have 2 cotyledons Monocotyledons have 1 cotyledon A cotyledon is an embryonic leaf. It is the first leaf to appear when a seedling grows. They often contain reserves of food which the developing seedling can use to grow. Epicotyl The section of stem between the cotyledon(s) and the plumule. In a seedling it is the section of stem between the cotyledons and the first true leaves. -

Drupe. Fruit with a Hard Endocarp (Figs. 67 and 71-73); E.G., and Sterculiaceae (Helicteres Guazumaefolia, Sterculia)
Fig. 71. Fig. 72. Fig. 73. Drupe. Fruit with a hard endocarp (figs. 67 and 71-73); e.g., and Sterculiaceae (Helicteres guazumaefolia, Sterculia). Anacardiaceae (Spondias purpurea, S. mombin, Mangifera indi- Desmopsis bibracteata (Annonaceae) has aggregate follicles ca, Tapirira), Caryocaraceae (Caryocar costaricense), Chrysobal- with constrictions between successive seeds, similar to those anaceae (Licania), Euphorbiaceae (Hyeronima), Malpighiaceae found in loments. (Byrsonima crispa), Olacaceae (Minquartia guianensis), Sapin- daceae (Meliccocus bijugatus), and Verbenaceae (Vitex cooperi). Samaracetum. Aggregate of samaras (fig. 74); e.g., Aceraceae (Acer pseudoplatanus), Magnoliaceae (Liriodendron tulipifera Hesperidium. Septicidal berry with a thick pericarp (fig. 67). L.), Sapindaceae (Thouinidium dodecandrum), and Tiliaceae Most of the fruit is derived from glandular trichomes. It is (Goethalsia meiantha). typical of the Rutaceae (Citrus). Multiple Fruits Aggregate Fruits Multiple fruits are found along a single axis and are usually coalescent. The most common types follow: Several types of aggregate fruits exist (fig. 74): Bibacca. Double fused berry; e.g., Lonicera. Achenacetum. Cluster of achenia; e.g., the strawberry (Fra- garia vesca). Sorosis. Fruits usually coalescent on a central axis; they derive from the ovaries of several flowers; e.g., Moraceae (Artocarpus Baccacetum or etaerio. Aggregate of berries; e.g., Annonaceae altilis). (Asimina triloba, Cananga odorata, Uvaria). The berries can be aggregate and syncarpic as in Annona reticulata, A. muricata, Syconium. Syncarp with many achenia in the inner wall of a A. pittieri and other species. hollow receptacle (fig. 74); e.g., Ficus. Drupacetum. Aggregate of druplets; e.g., Bursera simaruba THE GYMNOSPERM FRUIT (Burseraceae). Fertilization stimulates the growth of young gynostrobiles Folliacetum. Aggregate of follicles; e.g., Annonaceae which in species such as Pinus are more than 1 year old. -

Tree Seedling Availability, Planting, and Initial Care
F-5024 Tree Seedling Availability, Planting, and Initial Care William G. Ross Assistant Professor Oklahoma Cooperative Extension Fact Sheets Extension Forestry Specialistst are also available on our website at: http://www.osuextra.com Introduction This is the second in a series of three publications addressing the topic of forest stand establishment. If one is Bareroot not sure of what seedlings to plant or of what forest product to Most nurseries that sell seedlings produce some form of produce, it may be beneficial to read the first publication in this bareroot stock. Bareroot seedlings, as the name describes, is series, titled “Tree Planting Objectives and the Seedling a seedling with only the stem and the root supplied to the Selection Process.” landowner for planting. The major advantages of bareroot There are four points toconsider to identify management stock are the relative ease of production in the nurseries, the objectives. These are listed in brief form below. relative low cost to landowner, the relative ease of planting, and the ability to mechanize many of the operations in the 1. Identify the growing region where land is located. seedling production method. 2. Identify the soil types and moisture regimes located on the Shortcomings of bareroot seedlings include the vulner- land. ability of the scedling to the uncontrollable nursery environ- 3. Identify those objectives feasible and within the bounds of ment, exposure of the root system to harsh environmental the above environmental constraints. conditions after lifting, and the requirement of a large, sea- 4. Identify those tree species that, when managed correctly, sonal labor force to plant the seedlings. -

Life Cycles of Plants
Life Cycles of Plants Flowering plants produce flowers. The flowers later become fruit. Inside the fruit, we can find seeds. The fruit protect the seeds. Seeds can develop into new plants. The life cycle of flowering plants follows the three stages of seed-seedling-adult. Seed Seedling (Young plant) Stage 1: The plant begins Stage 2: The young plant that grows is known as a its life as a seed. With seedling. enough air, food, water and the right temperature, At first, the seedling obtains its food from the seed the seed will begin to leaves. germinate or grow. When the true leaves start to grow, the plant is ready to make its own food by the process of photosynthesis. The seed leaves will shrivel and fall off. Roots grow deep down into the ground to obtain water and mineral salts needed for the plant. The plant will grow towards the sunlight. The shoot grows upwards to obtain maximum sunlight. When the true leaves appear, the plant is ready to make its own food. Seed leaves provide the plant with food before the true leaves develop. Roots grow downwards to absorb water and mineral salts from the ground. They hold the plant firmly in the soil. Adult plant Stage 3: As the plant grows, it develops flowers which later become fruit. There are seeds inside the fruit. The seeds will fall to the ground and develop into new plants. The life cycle then repeats itself. Life cycle of a flowering plant Adapted: PSLE Science Partner A Complete Guide to L&U Block © Singapore Asia Publishers Pte Ltd. -

Genetic Suppression of Plant Development and Chloroplast Biogenesis Via the Snowy Cotyledon 3 and Phytochrome B Pathways
CSIRO PUBLISHING Functional Plant Biology, 2015, 42, 676–686 http://dx.doi.org/10.1071/FP15026 Genetic suppression of plant development and chloroplast biogenesis via the Snowy Cotyledon 3 and Phytochrome B pathways Diep Ganguly A, Peter Crisp A, Klaus Harter B, Barry J. Pogson A and Verónica Albrecht-Borth A,C AARC (Australian Research Council) Centre of Excellence in Plant Energy Biology, Research School of Biology, Australian National University Canberra, Acton, ACT 0200, Australia. BZentrum für Molekularbiologie der Pflanzen, Plant Physiology, University of Tübingen, 72076 Tübingen, Germany. CCorresponding author. Email: [email protected] Abstract. Plant development is regulated by external and internal factors such as light and chloroplast development. A revertant of the Arabidopsis thaliana (L.) Heyhn. chloroplast biogenesis mutant snowy cotyledon 3 (sco3–1) was isolated partially recovering the impaired chloroplast phenotype. The mutation was identified in the Phytochrome B (PhyB) gene and is a result of an amino acid change within the PAS repeat domain required for light-induced nuclear localisation. An independent phyB-9 mutation was crossed into sco3–1 mutants, resulting in the same partial reversion of sco3–1. Further analysis demonstrated that SCO3 and PhyB influence the greening process of seedlings and rosette leaves, embryogenesis, rosette formation and flowering. Interestingly, the functions of these proteins are interwoven in various ways, suggesting a complex genetic interaction. Whole-transcriptome profiling of sco3–1phyB-9 indicated that a completely distinct set of genes was differentially regulated in the double mutant compared with the single sco3–1 or phyB-9 mutants. Thus, we hypothesise that PhyB and SCO3 genetically suppress each other in plant and chloroplast development. -

New Insights on the Main Factors That Guide Seed Dormancy and Germination
G C A T T A C G G C A T genes Review From the Outside to the Inside: New Insights on the Main Factors That Guide Seed Dormancy and Germination Chiara Longo †, Soyanni Holness †, Veronica De Angelis, Andrea Lepri, Sara Occhigrossi, Veronica Ruta and Paola Vittorioso * Department Biology and Biotechnology C. Darwin, Sapienza Università di Roma, P.le Aldo Moro 5, 00185 Rome, Italy; [email protected] (C.L.); [email protected] (S.H.); [email protected] (V.D.A.); [email protected] (A.L.); [email protected] (S.O.); [email protected] (V.R.) * Correspondence: [email protected]; Tel.: +39-064-991-2265 † These authors contributed equally to this work. Abstract: The transition from a dormant to a germinating seed represents a crucial developmental switch in the life cycle of a plant. Subsequent transition from a germinating seed to an autotrophic organism also requires a robust and multi-layered control. Seed germination and seedling growth are multistep processes, involving both internal and external signals, which lead to a fine-tuning control network. In recent years, numerous studies have contributed to elucidate the molecular mechanisms underlying these processes: from light signaling and light-hormone crosstalk to the effects of abiotic stresses, from epigenetic regulation to translational control. However, there are still many open questions and molecular elements to be identified. This review will focus on the different aspects of the molecular control of seed dormancy and germination, pointing out new molecular elements and how these integrate in the signaling pathways already known. -

1.3 Propagating Crops from Seed and Greenhouse Management
1.3 Propagating Crops from Seed and Greenhouse Management Introduction 3 Instructor’s Lecture 1 Outline: Seed and Seedling Biology and Cultural Requirements 5 Detailed Lecture 1 Outline for Students 7 Instructor’s Lecture 2 Outline: Propagation Media and Container Formats 11 Detailed Lecture 2 Outline for Students 15 Demonstration 1: Greenhouse Management 21 Instructor’s Demonstration Outline 21 Demonstration 2: Propagation Media 23 Instructor’s Demonstration Outline 23 Demonstration 3: Sowing Seed 25 Instructor’s Demonstration Outline 26 Demonstration 4: Transplanting or “Pricking Out” 27 Instructor’s Demonstration Outline 28 Demonstration 5: Irrigation in Propagation Facilities 29 Instructor’s Demonstration Outline 29 Demonstration 6: Seedling Development and the “Hardening Off” Process 31 Instructor’s Demonstration Outline 31 Assessment Questions and Key 33 Resources 37 Glossary 39 Appendices 1. Seed Viability Chart 41 2. Soil Temperature Conditions for Vegetable Seed Germination 42 3. Examples of Propagation Containers 43 4. Days Required for Seedling Emergence at Various Soil Temperatures 44 5. Approximate Monthly Temperatures for Best Growth and Quality of Vegetable Crops 45 6. Propagation Media— Ingredients and Properties Imparted 46 7. Sample Soil Mix Recipes 47 8. “Pricking Out” Technique, Depth of Planting 48 9. Flat-Grown and Cell-Grown Seedlings 49 10. Propagation and Crop Performance Records Sheet 50 11. Greenhouse Records Sheet 51 2 | Unit 1.3 Propagation/Greenhouse Management Introduction: Propagation/Greenhouse Management UNIT OVERVIEW MODES OF INSTRUCTION Getting plants off to a healthy > LECTURE (2 LECTURES, 2 HOURS EACH) start is critical to successful crop Lecture 1 covers seed biology, and the cultural require- production.This unit introduces ments for germination. -

Physiological and Biochemical Response to Fusarium Culmorum Infection in Three Durum Wheat Genotypes at Seedling and Full Anthesis Stage
International Journal of Molecular Sciences Article Physiological and Biochemical Response to Fusarium culmorum Infection in Three Durum Wheat Genotypes at Seedling and Full Anthesis Stage Jakub Pastuszak 1,* , Anna Szczerba 1, Michał Dziurka 2 , Marta Hornyák 1,3, Przemysław Kope´c 2 , Marek Szklarczyk 4 and Agnieszka Płazek˙ 1 1 Department of Plant Breeding, Physiology and Seed Science, University of Agriculture, Podłuzna˙ 3, 30-239 Kraków, Poland; [email protected] (A.S.); [email protected] (M.H.); [email protected] (A.P.) 2 Franciszek Górski Institute of Plant Physiology, Polish Academy of Sciences, Niezapominajek 21, 30-239 Kraków, Poland; [email protected] (M.D.); [email protected] (P.K.) 3 Polish Academy of Sciences, W. Szafer Institute of Botany, Lubicz 46, 31-512 Kraków, Poland 4 Faculty of Biotechnology and Horticulture, University of Agriculture, 29 Listopada 54, 31-425 Kraków, Poland; [email protected] * Correspondence: [email protected] Abstract: Fusarium culmorum is a worldwide, soil-borne plant pathogen. It causes diseases of cereals, reduces their yield, and fills the grain with toxins. The main direction of modern breeding is to select wheat genotypes the most resistant to Fusarium diseases. This study uses seedlings and plants at the anthesis stage to analyze total soluble carbohydrates, total and cell-wall bound phenolics, Citation: Pastuszak, J.; Szczerba, A.; chlorophyll content, antioxidant activity, hydrogen peroxide content, mycotoxin accumulation, visual Dziurka, M.; Hornyák, M.; Kope´c,P.; symptoms of the disease, and Fusarium head blight index (FHBi). These results determine the Szklarczyk, M.; Płazek,˙ A. -

BIO 102 General Biology Lecture Outline Plantae I. Introduction A. Taxonomy Domain
BIO 102 General Biology Lecture Outline Plantae I. Introduction A. Taxonomy Domain: Eukarya Kingdom: Plantae B. General characteristics Multicellular Autotrophic / Photosynthetic Cellulose cell wall Reproduction – alternation of generations C. Life cycle Alternation of generations Haploid = gametophyte, which produces gametes via mitosis Diploid = sporophyte, which produces spores via meiosis Meiosis = cell division from 2n to n Mitosis = cell division either from 2n to 2n, OR from n to n Fertilization = combine gametes: n + n = 2n Homospory vs. heterospory II. Major Groups of Plants A. Characterized by 4 criteria in branching manner Vascular vs. non-vascular Seed production or lack thereof Types of seeds produced Development & morphology B. Non-tracheophytes (nonvascular plants) General characteristics No vascular tissue Gametophyte (haploid) generation dominant Requires water for reproduction Examples Liverworts Hornworts Mosses Moss reproductive cycle C. Tracheophytes General characteristics Vascular tissue: Xylem and Phloem Specialized tissues: Leaves Roots Stems Cuticles Stomata Sporophyte (diploid) generation dominant Divisions based on seed-bearing vs. non-seed bearing (Seed vs. spore) D. Seedless plants BIO 102 General Biology Lecture Outline General characteristics Have vascular tissue but lack seeds Examples Ferns Whisk ferns Club mosses Horsetails E. Seed-bearing plants General characteristics Have vascular tissue and seeds Divisions based on whether seeds are “covered” or not F. Gymnosperms General characteristics Vascular plants with “naked” seeds = i.e., no fruit Examples Conifers Gingkos Cycads Gnetophytes Gymnosperm life cycle G. Angiosperms General characteristics Vascular plants with “covered” seeds = i.e., seeds within fruits Fruits are derived from flowers Angiosperm = flowering plant Divisions are based on development and plant morphology (i.e., structures) Angiosperm life cycle H. -

Eudicots Monocots Stems Embryos Roots Leaf Venation Pollen Flowers
Monocots Eudicots Embryos One cotyledon Two cotyledons Leaf venation Veins Veins usually parallel usually netlike Stems Vascular tissue Vascular tissue scattered usually arranged in ring Roots Root system usually Taproot (main root) fibrous (no main root) usually present Pollen Pollen grain with Pollen grain with one opening three openings Flowers Floral organs usually Floral organs usually in in multiples of three multiples of four or five © 2014 Pearson Education, Inc. 1 Reproductive shoot (flower) Apical bud Node Internode Apical bud Shoot Vegetative shoot system Blade Leaf Petiole Axillary bud Stem Taproot Lateral Root (branch) system roots © 2014 Pearson Education, Inc. 2 © 2014 Pearson Education, Inc. 3 Storage roots Pneumatophores “Strangling” aerial roots © 2014 Pearson Education, Inc. 4 Stolon Rhizome Root Rhizomes Stolons Tubers © 2014 Pearson Education, Inc. 5 Spines Tendrils Storage leaves Stem Reproductive leaves Storage leaves © 2014 Pearson Education, Inc. 6 Dermal tissue Ground tissue Vascular tissue © 2014 Pearson Education, Inc. 7 Parenchyma cells with chloroplasts (in Elodea leaf) 60 µm (LM) © 2014 Pearson Education, Inc. 8 Collenchyma cells (in Helianthus stem) (LM) 5 µm © 2014 Pearson Education, Inc. 9 5 µm Sclereid cells (in pear) (LM) 25 µm Cell wall Fiber cells (cross section from ash tree) (LM) © 2014 Pearson Education, Inc. 10 Vessel Tracheids 100 µm Pits Tracheids and vessels (colorized SEM) Perforation plate Vessel element Vessel elements, with perforated end walls Tracheids © 2014 Pearson Education, Inc. 11 Sieve-tube elements: 3 µm longitudinal view (LM) Sieve plate Sieve-tube element (left) and companion cell: Companion cross section (TEM) cells Sieve-tube elements Plasmodesma Sieve plate 30 µm Nucleus of companion cell 15 µm Sieve-tube elements: longitudinal view Sieve plate with pores (LM) © 2014 Pearson Education, Inc.