A Role for Alternative Splicing in Circadian Control of Exocytosis and Glucose Homeostasis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Manual: Pathdetect Reporter Cell Lines
PathDetect Reporter Cell Lines INSTRUCTION MANUAL Catalog #800050 (PathDetect HLR Cell Line) #800055 (HLR-Elk1 Cell Line) #800060 (HLR-CHOP Cell Line) #800065 (HLR-CREB Cell Line) Revision A BN #800050-12 For In Vitro Use Only 800050-12 LIMITED PRODUCT WARRANTY This warranty limits our liability to replacement of this product. No other warranties of any kind, express or implied, including without limitation, implied warranties of merchantability or fitness for a particular purpose, are provided by Agilent. Agilent shall have no liability for any direct, indirect, consequential, or incidental damages arising out of the use, the results of use, or the inability to use this product. ORDERING INFORMATION AND TECHNICAL SERVICES United States and Canada Agilent Technologies Stratagene Products Division 11011 North Torrey Pines Road La Jolla, CA 92037 Telephone (858) 373-6300 Order Toll Free (800) 424-5444 Technical Services (800) 894-1304 Internet [email protected] World Wide Web www.stratagene.com Europe Location Telephone Fax Technical Services Austria 0800 292 499 0800 292 496 0800 292 498 Belgium 00800 7000 7000 00800 7001 7001 00800 7400 7400 0800 15775 0800 15740 0800 15720 France 00800 7000 7000 00800 7001 7001 00800 7400 7400 0800 919 288 0800 919 287 0800 919 289 Germany 00800 7000 7000 00800 7001 7001 00800 7400 7400 0800 182 8232 0800 182 8231 0800 182 8234 Netherlands 00800 7000 7000 00800 7001 7001 00800 7400 7400 0800 023 0446 +31 (0)20 312 5700 0800 023 0448 Switzerland 00800 7000 7000 00800 7001 7001 00800 7400 7400 0800 563 080 0800 563 082 0800 563 081 United Kingdom 00800 7000 7000 00800 7001 7001 00800 7400 7400 0800 917 3282 0800 917 3283 0800 917 3281 All Other Countries Please contact your local distributor. -
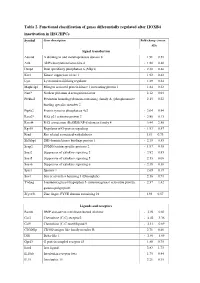
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

Expression of Oncogenes ELK1 and ELK3 in Cancer
Review Article Annals of Colorectal Cancer Research Published: 11 Nov, 2019 Expression of Oncogenes ELK1 and ELK3 in Cancer Akhlaq Ahmad and Asif Hayat* College of Chemistry, Fuzhou University, China Abstract Cancer is the uncontrolled growth of abnormal cells anywhere in a body, ELK1 and ELK3 is a member of the Ets-domain transcription factor family and the TCF (Ternary Complex Factor) subfamily. Proteins in this subfamily regulate transcription when recruited by SRF (Serum Response Factor) to bind to serum response elements. ELK1 and ELK3 transcription factors are known as oncogenes. Both transcription factors are proliferated in a different of type of cancer. Herein, we summarized the expression of transcription factor ELK1 and ELK3 in cancer cells. Keywords: ETS; ELK1; ELK3; Transcription factor; Cancer Introduction The ETS, a transcription factor of E twenty-six family based on a dominant ETS amino acids that integrated with a ~10-basepair element arrange in highly mid core sequence 5′-GGA(A/T)-3′ [1-2]. The secular family alter enormous 28/29 members which has been assigned in human and mouse and similarly the family description are further sub-divided into nine sub-families according to their homology and domain factor [3]. More importantly, one of the subfamily members such as ELK (ETS-like) adequate an N-terminal ETS DNA-binding domain along with a B-box domain that transmit the response of serum factor upon the formation of ternary complex and therefore manifested as ternary complex factors [4]. Further the ELK sub-divided into Elk1, Elk3 (Net, Erp or Sap2) and Elk4 (Sap1) proteins [3,4], which simulated varied proportional of potential protein- protein interactions [4,5]. -

TRANSCRIPTIONAL REGULATION of Hur in RENAL STRESS
TRANSCRIPTIONAL REGULATION OF HuR IN RENAL STRESS DISSERTATION Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy in the Graduate School of The Ohio State University By Sudha Suman Govindaraju Graduate Program in Biochemistry The Ohio State University 2014 Dissertation Committee: Dr. Beth S. Lee, Ph.D., Advisor Dr. Kathleen Boris-Lawrie, Ph.D. Dr. Sissy M. Jhiang, Ph.D. Dr. Arthur R. Strauch, Ph.D Abstract HuR is a ubiquitously expressed RNA-binding protein that affects the post- transcriptional life of thousands of cellular mRNAs by regulating transcript stability and translation. HuR can post-transcriptionally regulate gene expression and modulate cellular responses to stress, differentiation, proliferation, apoptosis, senescence, inflammation, and the immune response. It is an important mediator of survival during cellular stress, but when inappropriately expressed, can promote oncogenic transformation. Not surprisingly, the expression of HuR itself is tightly regulated at multiple transcriptional and post-transcriptional levels. Previous studies demonstrated the existence of two alternate HuR transcripts that differ in their 5’ untranslated regions and have markedly different translatabilities. These forms were also found to be reciprocally expressed following cellular stress in kidney proximal tubule cell lines, and the shorter, more readily translatable variant was shown to be regulated by Smad 1/5/8 pathway and bone morphogenetic protein-7 (BMP-7) signaling. In this study, the factors that promote transcription of the longer alternate form were identified. NF-κB was shown to be important for expression of the long HuR mRNA, as was a newly identified region with potential for binding the Sp/KLF families of transcription factors. -

Molecular Profile of Tumor-Specific CD8+ T Cell Hypofunction in a Transplantable Murine Cancer Model
Downloaded from http://www.jimmunol.org/ by guest on September 25, 2021 T + is online at: average * The Journal of Immunology , 34 of which you can access for free at: 2016; 197:1477-1488; Prepublished online 1 July from submission to initial decision 4 weeks from acceptance to publication 2016; doi: 10.4049/jimmunol.1600589 http://www.jimmunol.org/content/197/4/1477 Molecular Profile of Tumor-Specific CD8 Cell Hypofunction in a Transplantable Murine Cancer Model Katherine A. Waugh, Sonia M. Leach, Brandon L. Moore, Tullia C. Bruno, Jonathan D. Buhrman and Jill E. Slansky J Immunol cites 95 articles Submit online. Every submission reviewed by practicing scientists ? is published twice each month by Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts http://jimmunol.org/subscription Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html http://www.jimmunol.org/content/suppl/2016/07/01/jimmunol.160058 9.DCSupplemental This article http://www.jimmunol.org/content/197/4/1477.full#ref-list-1 Information about subscribing to The JI No Triage! Fast Publication! Rapid Reviews! 30 days* Why • • • Material References Permissions Email Alerts Subscription Supplementary The Journal of Immunology The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2016 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. This information is current as of September 25, 2021. The Journal of Immunology Molecular Profile of Tumor-Specific CD8+ T Cell Hypofunction in a Transplantable Murine Cancer Model Katherine A. -

TBX15/Mir-152/KIF2C Pathway Regulates Breast Cancer Doxorubicin Resistance Via Preventing PKM2 Ubiquitination
TBX15/miR-152/KIF2C Pathway Regulates Breast Cancer Doxorubicin Resistance via Preventing PKM2 Ubiquitination Cheng-Fei Jiang Nanjing Medical University Yun-Xia Xie Zhengzhou University Ying-Chen Qian Nanjing Medical University Min Wang Nanjing Medical University Ling-Zhi Liu Thomas Jefferson University - Center City Campus: Thomas Jefferson University Yong-Qian Shu Nanjing Medical University Xiao-Ming Bai ( [email protected] ) Nanjing Medical University Bing-Hua Jiang Thomas Jefferson University Research Keywords: TBX15, miR-152, KIF2C, PKM2, Doxorubicin resistance, breast cancer Posted Date: December 28th, 2020 DOI: https://doi.org/10.21203/rs.3.rs-130874/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/29 Abstract Background Chemoresistance is a critical risk problem for breast cancer treatment. However, mechanisms by which chemoresistance arises remains to be elucidated. The expression of T-box transcription factor 15 (TBX- 15) was found downregulated in some cancer tissues. However, role and mechanism of TBX15 in breast cancer chemoresistance is unknown. Here we aimed to identify the effects and mechanisms of TBX15 in doxorubicin resistance in breast cancer. Methods As measures of Drug sensitivity analysis, MTT and IC50 assays were used in DOX-resistant breast cancer cells. ECAR and OCR assays were used to analyze the glycolysis level, while Immunoblotting and Immunouorescence assays were used to analyze the autophagy levels in vitro. By using online prediction software, luciferase reporter assays, co-Immunoprecipitation, Western blotting analysis and experimental animals models, we further elucidated the mechanisms. Results We found TBX15 expression levels were decreased in Doxorubicin (DOX)-resistant breast cancer cells. -

The Krüppel-Like Factors in Female Reproductive System Pathologies
R C M SIMMEN and others KLFs in female reproductive 54:2 R89–R101 Review diseases The Kru¨ppel-like factors in female reproductive system pathologies Rosalia C M Simmen, Melissa E Heard, Angela M Simmen1, Maria Theresa M Montales, Meera Marji, Samantha Scanlon and John Mark P Pabona2 Department of Physiology and Biophysics, University of Arkansas for Medical Sciences, Little Rock, Correspondence Arkansas 72205, USA should be addressed 1Department of Obstetrics and Gynecology, University of Michigan Health System, Ann Arbor, Michigan 48109, USA to R C M Simmen 2Department of Internal Medicine, Harlem Hospital Center, Columbia University Medical Center, Email New York, New York 10037, USA [email protected] Abstract Female reproductive tract pathologies arise largely from dysregulation of estrogen and Key Words progesterone receptor signaling, leading to aberrant cell proliferation, survival, and " KLF differentiation. The signaling pathways orchestrated by these nuclear receptors are complex, " endometrial pathologies require the participation of many nuclear proteins serving as key binding partners or targets, " progesterone and involve a range of paracrine and autocrine regulatory circuits. The members of the " Notch Kru¨ ppel-like factor (KLF) family of transcription factors are ubiquitously expressed in " Wnt reproductive tissues and have been increasingly implicated as critical co-regulators and integrators of steroid hormone actions. Herein, we explore the involvement of KLF family members in uterine pathology, describe their currently known molecular mechanisms, Journal of Molecular and discuss their potential as targets for therapeutic intervention. Endocrinology (2015) 54, R89–R101 Journal of Molecular Endocrinology Introduction The human uterus has a unique role in the successful transcriptional pathways involve interactions with transmission of germ line DNA to guarantee the numerous nuclear co-regulators (Dasgupta & O’Malley propagation of the human species. -

New Mechanism Based Approaches for Treating Prostate Cancer Rayna Rosati Wayne State University
Wayne State University Wayne State University Dissertations 1-1-2017 New Mechanism Based Approaches For Treating Prostate Cancer Rayna Rosati Wayne State University, Follow this and additional works at: https://digitalcommons.wayne.edu/oa_dissertations Part of the Oncology Commons Recommended Citation Rosati, Rayna, "New Mechanism Based Approaches For Treating Prostate Cancer" (2017). Wayne State University Dissertations. 1865. https://digitalcommons.wayne.edu/oa_dissertations/1865 This Open Access Dissertation is brought to you for free and open access by DigitalCommons@WayneState. It has been accepted for inclusion in Wayne State University Dissertations by an authorized administrator of DigitalCommons@WayneState. NEW MECHANISM BASED APPROACHES FOR TREATING PROSTATE CANCER by RAYNA C. ROSATI DISSERTATION Submitted to the Graduate School of Wayne State University, Detroit, Michigan in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY 2017 MAJOR: CANCER BIOLOGY Approved By: Advisor Date DEDICATION This dissertation is dedicated to my family, who made me who I am today with all of their love and support. To my grandmother, Lucy, who just recently lost her battle with lung cancer and who would send me newspaper clippings in the mail about new topics of prostate cancer research. To my siblings, who have always been my best friends. To my father, Daniel, who has been my greatest mentor in life. To my mother, Nanci, who has been there for me through everything and who gave me my creative bone. ii ACKNOWLEDGEMENTS First off, I would like to thank my mentor, Dr. Manohar Ratnam, whose enthusiasm for science is undeniable. Thank you for always being so optimistic and believing I could accomplish this very challenging project. -

Mediator of DNA Damage Checkpoint 1 (MDC1) Is a Novel Estrogen Receptor Co-Regulator in Invasive 6 Lobular Carcinoma of the Breast 7 8 Evelyn K
bioRxiv preprint doi: https://doi.org/10.1101/2020.12.16.423142; this version posted December 16, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. 1 Running Title: MDC1 co-regulates ER in ILC 2 3 Research article 4 5 Mediator of DNA damage checkpoint 1 (MDC1) is a novel estrogen receptor co-regulator in invasive 6 lobular carcinoma of the breast 7 8 Evelyn K. Bordeaux1+, Joseph L. Sottnik1+, Sanjana Mehrotra1, Sarah E. Ferrara2, Andrew E. Goodspeed2,3, James 9 C. Costello2,3, Matthew J. Sikora1 10 11 +EKB and JLS contributed equally to this project. 12 13 Affiliations 14 1Dept. of Pathology, University of Colorado Anschutz Medical Campus 15 2Biostatistics and Bioinformatics Shared Resource, University of Colorado Comprehensive Cancer Center 16 3Dept. of Pharmacology, University of Colorado Anschutz Medical Campus 17 18 Corresponding author 19 Matthew J. Sikora, PhD.; Mail Stop 8104, Research Complex 1 South, Room 5117, 12801 E. 17th Ave.; Aurora, 20 CO 80045. Tel: (303)724-4301; Fax: (303)724-3712; email: [email protected]. Twitter: 21 @mjsikora 22 23 Authors' contributions 24 MJS conceived of the project. MJS, EKB, and JLS designed and performed experiments. JLS developed models 25 for the project. EKB, JLS, SM, and AEG contributed to data analysis and interpretation. SEF, AEG, and JCC 26 developed and performed informatics analyses. MJS wrote the draft manuscript; all authors read and revised the 27 manuscript and have read and approved of this version of the manuscript. -

Transcriptome Sequencing and Genome-Wide Association Analyses Reveal Lysosomal Function and Actin Cytoskeleton Remodeling in Schizophrenia and Bipolar Disorder
Molecular Psychiatry (2015) 20, 563–572 © 2015 Macmillan Publishers Limited All rights reserved 1359-4184/15 www.nature.com/mp ORIGINAL ARTICLE Transcriptome sequencing and genome-wide association analyses reveal lysosomal function and actin cytoskeleton remodeling in schizophrenia and bipolar disorder Z Zhao1,6,JXu2,6, J Chen3,6, S Kim4, M Reimers3, S-A Bacanu3,HYu1, C Liu5, J Sun1, Q Wang1, P Jia1,FXu2, Y Zhang2, KS Kendler3, Z Peng2 and X Chen3 Schizophrenia (SCZ) and bipolar disorder (BPD) are severe mental disorders with high heritability. Clinicians have long noticed the similarities of clinic symptoms between these disorders. In recent years, accumulating evidence indicates some shared genetic liabilities. However, what is shared remains elusive. In this study, we conducted whole transcriptome analysis of post-mortem brain tissues (cingulate cortex) from SCZ, BPD and control subjects, and identified differentially expressed genes in these disorders. We found 105 and 153 genes differentially expressed in SCZ and BPD, respectively. By comparing the t-test scores, we found that many of the genes differentially expressed in SCZ and BPD are concordant in their expression level (q ⩽ 0.01, 53 genes; q ⩽ 0.05, 213 genes; q ⩽ 0.1, 885 genes). Using genome-wide association data from the Psychiatric Genomics Consortium, we found that these differentially and concordantly expressed genes were enriched in association signals for both SCZ (Po10 − 7) and BPD (P = 0.029). To our knowledge, this is the first time that a substantially large number of genes show concordant expression and association for both SCZ and BPD. Pathway analyses of these genes indicated that they are involved in the lysosome, Fc gamma receptor-mediated phagocytosis, regulation of actin cytoskeleton pathways, along with several cancer pathways. -

Ten Commandments for a Good Scientist
Unravelling the mechanism of differential biological responses induced by food-borne xeno- and phyto-estrogenic compounds Ana María Sotoca Covaleda Wageningen 2010 Thesis committee Thesis supervisors Prof. dr. ir. Ivonne M.C.M. Rietjens Professor of Toxicology Wageningen University Prof. dr. Albertinka J. Murk Personal chair at the sub-department of Toxicology Wageningen University Thesis co-supervisor Dr. ir. Jacques J.M. Vervoort Associate professor at the Laboratory of Biochemistry Wageningen University Other members Prof. dr. Michael R. Muller, Wageningen University Prof. dr. ir. Huub F.J. Savelkoul, Wageningen University Prof. dr. Everardus J. van Zoelen, Radboud University Nijmegen Dr. ir. Toine F.H. Bovee, RIKILT, Wageningen This research was conducted under the auspices of the Graduate School VLAG Unravelling the mechanism of differential biological responses induced by food-borne xeno- and phyto-estrogenic compounds Ana María Sotoca Covaleda Thesis submitted in fulfillment of the requirements for the degree of doctor at Wageningen University by the authority of the Rector Magnificus Prof. dr. M.J. Kropff, in the presence of the Thesis Committee appointed by the Academic Board to be defended in public on Tuesday 14 September 2010 at 4 p.m. in the Aula Unravelling the mechanism of differential biological responses induced by food-borne xeno- and phyto-estrogenic compounds. Ana María Sotoca Covaleda Thesis Wageningen University, Wageningen, The Netherlands, 2010, With references, and with summary in Dutch. ISBN: 978-90-8585-707-5 “Caminante no hay camino, se hace camino al andar. Al andar se hace camino, y al volver la vista atrás se ve la senda que nunca se ha de volver a pisar” - Antonio Machado – A mi madre. -

Mechanisms of Synaptic Plasticity Mediated by Clathrin Adaptor-Protein Complexes 1 and 2 in Mice
Mechanisms of synaptic plasticity mediated by Clathrin Adaptor-protein complexes 1 and 2 in mice Dissertation for the award of the degree “Doctor rerum naturalium” at the Georg-August-University Göttingen within the doctoral program “Molecular Biology of Cells” of the Georg-August University School of Science (GAUSS) Submitted by Ratnakar Mishra Born in Birpur, Bihar, India Göttingen, Germany 2019 1 Members of the Thesis Committee Prof. Dr. Peter Schu Institute for Cellular Biochemistry, (Supervisor and first referee) University Medical Center Göttingen, Germany Dr. Hans Dieter Schmitt Neurobiology, Max Planck Institute (Second referee) for Biophysical Chemistry, Göttingen, Germany Prof. Dr. med. Thomas A. Bayer Division of Molecular Psychiatry, University Medical Center, Göttingen, Germany Additional Members of the Examination Board Prof. Dr. Silvio O. Rizzoli Department of Neuro-and Sensory Physiology, University Medical Center Göttingen, Germany Dr. Roland Dosch Institute of Developmental Biochemistry, University Medical Center Göttingen, Germany Prof. Dr. med. Martin Oppermann Institute of Cellular and Molecular Immunology, University Medical Center, Göttingen, Germany Date of oral examination: 14th may 2019 2 Table of Contents List of abbreviations ................................................................................. 5 Abstract ................................................................................................... 7 Chapter 1: Introduction ............................................................................