Degeneracy and All That = ∫
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Unit 1 Old Quantum Theory
UNIT 1 OLD QUANTUM THEORY Structure Introduction Objectives li;,:overy of Sub-atomic Particles Earlier Atom Models Light as clectromagnetic Wave Failures of Classical Physics Black Body Radiation '1 Heat Capacity Variation Photoelectric Effect Atomic Spectra Planck's Quantum Theory, Black Body ~diation. and Heat Capacity Variation Einstein's Theory of Photoelectric Effect Bohr Atom Model Calculation of Radius of Orbits Energy of an Electron in an Orbit Atomic Spectra and Bohr's Theory Critical Analysis of Bohr's Theory Refinements in the Atomic Spectra The61-y Summary Terminal Questions Answers 1.1 INTRODUCTION The ideas of classical mechanics developed by Galileo, Kepler and Newton, when applied to atomic and molecular systems were found to be inadequate. Need was felt for a theory to describe, correlate and predict the behaviour of the sub-atomic particles. The quantum theory, proposed by Max Planck and applied by Einstein and Bohr to explain different aspects of behaviour of matter, is an important milestone in the formulation of the modern concept of atom. In this unit, we will study how black body radiation, heat capacity variation, photoelectric effect and atomic spectra of hydrogen can be explained on the basis of theories proposed by Max Planck, Einstein and Bohr. They based their theories on the postulate that all interactions between matter and radiation occur in terms of definite packets of energy, known as quanta. Their ideas, when extended further, led to the evolution of wave mechanics, which shows the dual nature of matter -

Quantum Theory of the Hydrogen Atom
Quantum Theory of the Hydrogen Atom Chemistry 35 Fall 2000 Balmer and the Hydrogen Spectrum n 1885: Johann Balmer, a Swiss schoolteacher, empirically deduced a formula which predicted the wavelengths of emission for Hydrogen: l (in Å) = 3645.6 x n2 for n = 3, 4, 5, 6 n2 -4 •Predicts the wavelengths of the 4 visible emission lines from Hydrogen (which are called the Balmer Series) •Implies that there is some underlying order in the atom that results in this deceptively simple equation. 2 1 The Bohr Atom n 1913: Niels Bohr uses quantum theory to explain the origin of the line spectrum of hydrogen 1. The electron in a hydrogen atom can exist only in discrete orbits 2. The orbits are circular paths about the nucleus at varying radii 3. Each orbit corresponds to a particular energy 4. Orbit energies increase with increasing radii 5. The lowest energy orbit is called the ground state 6. After absorbing energy, the e- jumps to a higher energy orbit (an excited state) 7. When the e- drops down to a lower energy orbit, the energy lost can be given off as a quantum of light 8. The energy of the photon emitted is equal to the difference in energies of the two orbits involved 3 Mohr Bohr n Mathematically, Bohr equated the two forces acting on the orbiting electron: coulombic attraction = centrifugal accelleration 2 2 2 -(Z/4peo)(e /r ) = m(v /r) n Rearranging and making the wild assumption: mvr = n(h/2p) n e- angular momentum can only have certain quantified values in whole multiples of h/2p 4 2 Hydrogen Energy Levels n Based on this model, Bohr arrived at a simple equation to calculate the electron energy levels in hydrogen: 2 En = -RH(1/n ) for n = 1, 2, 3, 4, . -

Degenerate Eigenvalue Problem 32.1 Degenerate Perturbation
Physics 342 Lecture 32 Degenerate Eigenvalue Problem Lecture 32 Physics 342 Quantum Mechanics I Wednesday, April 23rd, 2008 We have the matrix form of the first order perturbative result from last time. This carries over pretty directly to the Schr¨odingerequation, with only minimal replacement (the inner product and finite vector space change, but notationally, the results are identical). Because there are a variety of quantum mechanical systems with degenerate spectra (like the Hydrogen 2 eigenstates, each En has n associated eigenstates) and we want to be able to predict the energy shift associated with perturbations in these systems, we can copy our arguments for matrices to cover matrices with more than one eigenvector per eigenvalue. The punch line of that program is that we can use the non-degenerate perturbed energies, provided we start with the \correct" degenerate linear combinations. 32.1 Degenerate Perturbation N×N Going back to our symmetric matrix example, we have A IR , and 2 again, a set of eigenvectors and eigenvalues: A xi = λi xi. This time, suppose that the eigenvalue λi has a set of M associated eigenvectors { that is, suppose a set of eigenvectors yj satisfy: A yj = λi yj j = 1 M (32.1) −! 1 of 9 32.1. DEGENERATE PERTURBATION Lecture 32 (so this represents M separate equations) that are themselves orthonormal1. Clearly, any linear combination of these vectors is also an eigenvector: M M X X A βk yk = λi βk yk: (32.2) k=1 k=1 M PM Define the general combination of yi to be z βk yk, also an f gi=1 ≡ k=1 eigenvector of A with eigenvalue λi. -

Vibrational Quantum Number
Fundamentals in Biophotonics Quantum nature of atoms, molecules – matter Aleksandra Radenovic [email protected] EPFL – Ecole Polytechnique Federale de Lausanne Bioengineering Institute IBI 26. 03. 2018. Quantum numbers •The four quantum numbers-are discrete sets of integers or half- integers. –n: Principal quantum number-The first describes the electron shell, or energy level, of an atom –ℓ : Orbital angular momentum quantum number-as the angular quantum number or orbital quantum number) describes the subshell, and gives the magnitude of the orbital angular momentum through the relation Ll2 ( 1) –mℓ:Magnetic (azimuthal) quantum number (refers, to the direction of the angular momentum vector. The magnetic quantum number m does not affect the electron's energy, but it does affect the probability cloud)- magnetic quantum number determines the energy shift of an atomic orbital due to an external magnetic field-Zeeman effect -s spin- intrinsic angular momentum Spin "up" and "down" allows two electrons for each set of spatial quantum numbers. The restrictions for the quantum numbers: – n = 1, 2, 3, 4, . – ℓ = 0, 1, 2, 3, . , n − 1 – mℓ = − ℓ, − ℓ + 1, . , 0, 1, . , ℓ − 1, ℓ – –Equivalently: n > 0 The energy levels are: ℓ < n |m | ≤ ℓ ℓ E E 0 n n2 Stern-Gerlach experiment If the particles were classical spinning objects, one would expect the distribution of their spin angular momentum vectors to be random and continuous. Each particle would be deflected by a different amount, producing some density distribution on the detector screen. Instead, the particles passing through the Stern–Gerlach apparatus are deflected either up or down by a specific amount. -

The Quantum Mechanical Model of the Atom
The Quantum Mechanical Model of the Atom Quantum Numbers In order to describe the probable location of electrons, they are assigned four numbers called quantum numbers. The quantum numbers of an electron are kind of like the electron’s “address”. No two electrons can be described by the exact same four quantum numbers. This is called The Pauli Exclusion Principle. • Principle quantum number: The principle quantum number describes which orbit the electron is in and therefore how much energy the electron has. - it is symbolized by the letter n. - positive whole numbers are assigned (not including 0): n=1, n=2, n=3 , etc - the higher the number, the further the orbit from the nucleus - the higher the number, the more energy the electron has (this is sort of like Bohr’s energy levels) - the orbits (energy levels) are also called shells • Angular momentum (azimuthal) quantum number: The azimuthal quantum number describes the sublevels (subshells) that occur in each of the levels (shells) described above. - it is symbolized by the letter l - positive whole number values including 0 are assigned: l = 0, l = 1, l = 2, etc. - each number represents the shape of a subshell: l = 0, represents an s subshell l = 1, represents a p subshell l = 2, represents a d subshell l = 3, represents an f subshell - the higher the number, the more complex the shape of the subshell. The picture below shows the shape of the s and p subshells: (notice the electron clouds) • Magnetic quantum number: All of the subshells described above (except s) have more than one orientation. -

4 Nuclear Magnetic Resonance
Chapter 4, page 1 4 Nuclear Magnetic Resonance Pieter Zeeman observed in 1896 the splitting of optical spectral lines in the field of an electromagnet. Since then, the splitting of energy levels proportional to an external magnetic field has been called the "Zeeman effect". The "Zeeman resonance effect" causes magnetic resonances which are classified under radio frequency spectroscopy (rf spectroscopy). In these resonances, the transitions between two branches of a single energy level split in an external magnetic field are measured in the megahertz and gigahertz range. In 1944, Jevgeni Konstantinovitch Savoiski discovered electron paramagnetic resonance. Shortly thereafter in 1945, nuclear magnetic resonance was demonstrated almost simultaneously in Boston by Edward Mills Purcell and in Stanford by Felix Bloch. Nuclear magnetic resonance was sometimes called nuclear induction or paramagnetic nuclear resonance. It is generally abbreviated to NMR. So as not to scare prospective patients in medicine, reference to the "nuclear" character of NMR is dropped and the magnetic resonance based imaging systems (scanner) found in hospitals are simply referred to as "magnetic resonance imaging" (MRI). 4.1 The Nuclear Resonance Effect Many atomic nuclei have spin, characterized by the nuclear spin quantum number I. The absolute value of the spin angular momentum is L =+h II(1). (4.01) The component in the direction of an applied field is Lz = Iz h ≡ m h. (4.02) The external field is usually defined along the z-direction. The magnetic quantum number is symbolized by Iz or m and can have 2I +1 values: Iz ≡ m = −I, −I+1, ..., I−1, I. -

DEGENERACY CURVES, GAPS, and DIABOLICAL POINTS in the SPECTRA of NEUMANN PARALLELOGRAMS P Overfelt
DEGENERACY CURVES, GAPS, AND DIABOLICAL POINTS IN THE SPECTRA OF NEUMANN PARALLELOGRAMS P Overfelt To cite this version: P Overfelt. DEGENERACY CURVES, GAPS, AND DIABOLICAL POINTS IN THE SPECTRA OF NEUMANN PARALLELOGRAMS. 2020. hal-03017250 HAL Id: hal-03017250 https://hal.archives-ouvertes.fr/hal-03017250 Preprint submitted on 20 Nov 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. DEGENERACY CURVES, GAPS, AND DIABOLICAL POINTS IN THE SPECTRA OF NEUMANN PARALLELOGRAMS P. L. OVERFELT Abstract. In this paper we consider the problem of solving the Helmholtz equation over the space of all parallelograms subject to Neumann boundary conditions and determining the degeneracies occurring in their spectra upon changing the two parameters, angle and side ratio. This problem is solved numerically using the finite element method (FEM). Specifically for the lowest eleven normalized eigenvalue levels of the family of Neumann parallelograms, the intersection of two (or more) adjacent eigen- value level surfaces occurs in one of three ways: either as an isolated point associated with the special geometries, i.e., the rectangle, the square, or the rhombus, as part of a degeneracy curve which appears to contain an infinite number of points, or as a diabolical point in the Neumann parallelogram spec- trum. -

A Singular One-Dimensional Bound State Problem and Its Degeneracies
A Singular One-Dimensional Bound State Problem and its Degeneracies Fatih Erman1, Manuel Gadella2, Se¸cil Tunalı3, Haydar Uncu4 1 Department of Mathematics, Izmir˙ Institute of Technology, Urla, 35430, Izmir,˙ Turkey 2 Departamento de F´ısica Te´orica, At´omica y Optica´ and IMUVA. Universidad de Valladolid, Campus Miguel Delibes, Paseo Bel´en 7, 47011, Valladolid, Spain 3 Department of Mathematics, Istanbul˙ Bilgi University, Dolapdere Campus 34440 Beyo˘glu, Istanbul,˙ Turkey 4 Department of Physics, Adnan Menderes University, 09100, Aydın, Turkey E-mail: [email protected], [email protected], [email protected], [email protected] October 20, 2017 Abstract We give a brief exposition of the formulation of the bound state problem for the one-dimensional system of N attractive Dirac delta potentials, as an N N matrix eigenvalue problem (ΦA = ωA). The main aim of this paper is to illustrate that the non-degeneracy× theorem in one dimension breaks down for the equidistantly distributed Dirac delta potential, where the matrix Φ becomes a special form of the circulant matrix. We then give elementary proof that the ground state is always non-degenerate and the associated wave function may be chosen to be positive by using the Perron-Frobenius theorem. We also prove that removing a single center from the system of N delta centers shifts all the bound state energy levels upward as a simple consequence of the Cauchy interlacing theorem. Keywords. Point interactions, Dirac delta potentials, bound states. 1 Introduction Dirac delta potentials or point interactions, or sometimes called contact potentials are one of the exactly solvable classes of idealized potentials, and are used as a pedagogical tool to illustrate various physically important phenomena, where the de Broglie wavelength of the particle is much larger than the range of the interaction. -

A Relativistic Electron in a Coulomb Potential
A Relativistic Electron in a Coulomb Potential Alfred Whitehead Physics 518, Fall 2009 The Problem Solve the Dirac Equation for an electron in a Coulomb potential. Identify the conserved quantum numbers. Specify the degeneracies. Compare with solutions of the Schrödinger equation including relativistic and spin corrections. Approach My approach follows that taken by Dirac in [1] closely. A few modifications taken from [2] and [3] are included, particularly in regards to the final quantum numbers chosen. The general strategy is to first find a set of transformations which turn the Hamiltonian for the system into a form that depends only on the radial variables r and pr. Once this form is found, I solve it to find the energy eigenvalues and then discuss the energy spectrum. The Radial Dirac Equation We begin with the electromagnetic Hamiltonian q H = p − cρ ~σ · ~p − A~ + ρ mc2 (1) 0 1 c 3 with 2 0 0 1 0 3 6 0 0 0 1 7 ρ1 = 6 7 (2) 4 1 0 0 0 5 0 1 0 0 2 1 0 0 0 3 6 0 1 0 0 7 ρ3 = 6 7 (3) 4 0 0 −1 0 5 0 0 0 −1 1 2 0 1 0 0 3 2 0 −i 0 0 3 2 1 0 0 0 3 6 1 0 0 0 7 6 i 0 0 0 7 6 0 −1 0 0 7 ~σ = 6 7 ; 6 7 ; 6 7 (4) 4 0 0 0 1 5 4 0 0 0 −i 5 4 0 0 1 0 5 0 0 1 0 0 0 i 0 0 0 0 −1 We note that, for the Coulomb potential, we can set (using cgs units): Ze2 p = −eΦ = − o r A~ = 0 This leads us to this form for the Hamiltonian: −Ze2 H = − − cρ ~σ · ~p + ρ mc2 (5) r 1 3 We need to get equation 5 into a form which depends not on ~p, but only on the radial variables r and pr. -

11.-HYPERBOLA-THEORY.Pdf
12. HYPERBOLA 1. INTRODUCTION A hyperbola is the locus of a point which moves in the plane in such a way that Z the ratio of its distance from a fixed point in the same plane to its distance X’ P from a fixed line is always constant which is always greater than unity. M The fixed point is called the focus, the fixed line is called the directrix. The constant ratio is generally denoted by e and is known as the eccentricity of the Directrix hyperbola. A hyperbola can also be defined as the locus of a point such that S (focus) the absolute value of the difference of the distances from the two fixed points Z’ (foci) is constant. If S is the focus, ZZ′ is the directrix and P is any point on the hyperbola as show in figure. Figure 12.1 SP Then by definition, we have = e (e > 1). PM Note: The general equation of a conic can be taken as ax22+ 2hxy + by + 2gx + 2fy += c 0 This equation represents a hyperbola if it is non-degenerate (i.e. eq. cannot be written into two linear factors) ahg ∆ ≠ 0, h2 > ab. Where ∆=hb f gfc MASTERJEE CONCEPTS 1. The general equation ax22+ 2hxy + by + 2gx + 2fy += c 0 can be written in matrix form as ahgx ah x x y + 2gx + 2fy += c 0 and xy1hb f y = 0 hb y gfc1 Degeneracy condition depends on the determinant of the 3x3 matrix and the type of conic depends on the determinant of the 2x2 matrix. -
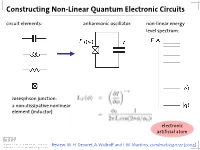
Constructing Non-Linear Quantum Electronic Circuits Circuit Elements: Anharmonic Oscillator: Non-Linear Energy Level Spectrum
Constructing Non-Linear Quantum Electronic Circuits circuit elements: anharmonic oscillator: non-linear energy level spectrum: Josesphson junction: a non-dissipative nonlinear element (inductor) electronic artificial atom Review: M. H. Devoret, A. Wallraff and J. M. Martinis, condmat/0411172 (2004) A Classification of Josephson Junction Based Qubits How to make use in of Jospehson junctions in a qubit? Common options of bias (control) circuits: phase qubit charge qubit flux qubit (Cooper Pair Box, Transmon) current bias charge bias flux bias How is the control circuit important? The Cooper Pair Box Qubit A Charge Qubit: The Cooper Pair Box discrete charge on island: continuous gate charge: total box capacitance Hamiltonian: electrostatic part: charging energy magnetic part: Josephson energy Hamilton Operator of the Cooper Pair Box Hamiltonian: commutation relation: charge number operator: eigenvalues, eigenfunctions completeness orthogonality phase basis: basis transformation Solving the Cooper Pair Box Hamiltonian Hamilton operator in the charge basis N : solutions in the charge basis: Hamilton operator in the phase basis δ : transformation of the number operator: solutions in the phase basis: Energy Levels energy level diagram for EJ=0: • energy bands are formed • bands are periodic in Ng energy bands for finite EJ • Josephson coupling lifts degeneracy • EJ scales level separation at charge degeneracy Charge and Phase Wave Functions (EJ << EC) courtesy CEA Saclay Charge and Phase Wave Functions (EJ ~ EC) courtesy CEA Saclay Tuning the Josephson Energy split Cooper pair box in perpendicular field = ( ) , cos cos 2 � � − − ̂ 0 SQUID modulation of Josephson energy consider two state approximation J. Clarke, Proc. IEEE 77, 1208 (1989) Two-State Approximation Restricting to a two-charge Hilbert space: Shnirman et al., Phys. -

1 Does Consciousness Really Collapse the Wave Function?
Does consciousness really collapse the wave function?: A possible objective biophysical resolution of the measurement problem Fred H. Thaheld* 99 Cable Circle #20 Folsom, Calif. 95630 USA Abstract An analysis has been performed of the theories and postulates advanced by von Neumann, London and Bauer, and Wigner, concerning the role that consciousness might play in the collapse of the wave function, which has become known as the measurement problem. This reveals that an error may have been made by them in the area of biology and its interface with quantum mechanics when they called for the reduction of any superposition states in the brain through the mind or consciousness. Many years later Wigner changed his mind to reflect a simpler and more realistic objective position, expanded upon by Shimony, which appears to offer a way to resolve this issue. The argument is therefore made that the wave function of any superposed photon state or states is always objectively changed within the complex architecture of the eye in a continuous linear process initially for most of the superposed photons, followed by a discontinuous nonlinear collapse process later for any remaining superposed photons, thereby guaranteeing that only final, measured information is presented to the brain, mind or consciousness. An experiment to be conducted in the near future may enable us to simultaneously resolve the measurement problem and also determine if the linear nature of quantum mechanics is violated by the perceptual process. Keywords: Consciousness; Euglena; Linear; Measurement problem; Nonlinear; Objective; Retina; Rhodopsin molecule; Subjective; Wave function collapse. * e-mail address: [email protected] 1 1.