Is the Use of DPP-4 Inhibitors Associated with an Increased Risk for Heart Failure?
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Treatment of Diabetes Mellitus
TREATMENT OF DIABETES MELLITUS DIABETES is a condition that affects how the body makes energy from food. Food is broken down into sugar (glucose) in the body and released into the blood. When the blood sugar level rises after a meal, insulin responds to let the sugar into the cells to be used as energy. In diabetes, the body either does not make enough insulin or it stops responding to insulin as well as it should. This results in sugar staying in the blood and leads to serious health problems over time. DIAGNOSIS OF DIABETES1 • A1C Test: Lab test measuring average blood sugar over past two to three months • Fasting Blood Sugar Test: Lab test measuring blood sugar after eight hours of no food or drink • Oral Glucose Tolerance Test (OGTT): Measures blood sugar before and two hours after drinking a specific sugary liquid • Random Blood Sugar Test: Measures blood sugar at a moment in time, without any kind of preparation (like fasting) FASTING BLOOD ORAL GLUCOSE TOLERANCE RANDOM BLOOD RESULT A1C TEST SUGAR TEST TEST SUGAR TEST Diabetes ≥ 6.5% ≥126 mg/dL ≥ 200 mg/dL ≥ 200 mg/dL Prediabetes 5.7 – 6.4% 100 – 125 mg/dL 140 – 199 mg/dL N/A Normal < 5.7% ≤99 mg/dL < 140 mg/dL N/A NON-DRUG TREATMENTS2 THERAPY COST WHAT TO EXPECT Diet (Mediterranean diet) and exercise (30 minutes a day, five days a week of moderate- Weight loss $-$$ intensity exercise); 7% weight loss decreases risk of diabetes3 Psychological intervention $$-$$$ Psychotherapy may reduce diabetic distress and improve glycemic control4,5 nationalcooperativerx.com PRESCRIPTION TREATMENTS -

Does Addition of Vildagliptin to Metformin Monotherapy Improve Glycemic Control in Patients with Type 2 Diabetes Mellitus?
PRACTICE pOINT www.nature.com/clinicalpractice/endmet Does addition of vildagliptin to metformin monotherapy improve glycemic control in patients with type 2 diabetes mellitus? Original article Bosi E et al. (2007) Effects of vildagliptin on OUTCOME MEASURES glucose control over 24 weeks in patients with type 2 diabetes The primary outcome measure was the change inadequately controlled with metformin. Diabetes Care 30: 890–895 in HbA1c level from baseline to study end. Secondary outcome measures included FPG SYNOPSIS levels, lipid profiles, β-cell function, and the KEYWORDS dipeptidyl peptidase 4 inhibitor, incidence and severity of adverse events. glycemic control, HbA1c, type 2 diabetes mellitus, vildagliptin RESULTS BACKGROUND The 50 mg vildagliptin, 100 mg vildagliptin Metformin is frequently prescribed as the and placebo groups comprised 177, 185, and first-line treatment for type 2 diabetes mellitus 182 patients, respectively. Participants were (T2DM); however, additional antidiabetic agents predominantly white and obese, with a mean might be required when glycemic control is age of 54 years. The mean duration of T2DM poor. was 6.2 years, the mean duration of metformin monotherapy was 17 months, and the mean OBJECTIVE metformin dose was 2,100 mg daily. The study To assess the efficacy and safety of the dipeptidyl was completed by >83% of patients in each peptidase 4 (DPP4) inhibitor vildagliptin as group. The mean baseline HbA1c level was 8.4% an add-on to metformin monotherapy. and the mean baseline FPG level was 9.9 mmol/l. Treatment with vildagliptin improved both of DESIGN AND INTERVENTION these parameters. The between-treatment This was a 24-week, multicenter, double- difference (vildagliptin minus placebo) in HbA1c blind, placebo-controlled, randomized study at study end was –0.7% and –1.1% with 50 mg of patients with T2DM inadequately controlled and 100 mg vildagliptin, respectively (P <0.001 with metformin monotherapy. -

Galvus Data Sheet
1 NEW ZEALAND DATA SHEET 1 GALVUS (50 mg tablets) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Vildagliptin: 1-[(3-Hydroxy-adamant-1-ylamino)-acetyl]-pyrrolidine-2(S)-carbonitrile. One tablet of Galvus® contains 50 mg of vildagliptin. For the full list of excipients, see section 6.1. 3 PHARMACEUTICAL FORM Galvus 50 mg: white to light yellowish, round (8 mm diameter) flat faced with beveled edges, unscored tablet. One side is debossed with "NVR", and the other side with "FB". 4 CLINICAL PARTICULARS 4.1 Therapeutic indications Galvus is indicated as an adjunct to diet and exercise to improve glycaemic control in patients with type 2 diabetes mellitus. • as monotherapy. • in dual combination with metformin, a sulphonylurea (SU), or a thiazolidinedione (TZD) when diet, exercise and a single antidiabetic agent do not result in adequate glycaemic control. • In triple combination with a sulphonylurea and metformin when diet and exercise plus dual therapy with these agents do not provide adequate glycaemic control. • In combination with insulin (with or without metformin) when diet, exercise and a stable dose of insulin do not result in adequate glycaemic control. 4.2 Dosage and method of administration Dose The management of antidiabetic therapy should be individualized. The recommended dose of Galvus is 50 mg once or twice daily. The maximum daily dose of Galvus is 100 mg. For monotherapy, and for combination with metformin, with a TZD or with insulin (with or without metformin), the recommended dose of Galvus is 50 mg or 100 mg daily. When used in dual combination with a sulphonylurea, the recommended dose of vildagliptin is 50 mg once daily. -

Januvia (Sitagliptin) Tablets
CENTER FOR DRUG EVALUATION AND RESEARCH Approval Package for: APPLICATION NUMBER: NDA 021995/S-013 Trade Name: JANUVIA Generic Name: Sitagliptin Sponsor: Merck & Co., Inc. Approval Date: 12/28/2009 Indications: JANUVIA is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: NDA 021995/S-013 CONTENTS Reviews / Information Included in this NDA Review. Approval Letter X Other Action Letters X Labeling X Summary Review Officer/Employee List Office Director Memo Cross Discipline Team Leader Review Medical Review(s) X Chemistry Review(s) Environmental Assessment Pharmacology Review(s) X Statistical Review(s) Microbiology Review(s) Clinical Pharmacology/Biopharmaceutics Review(s) Risk Assessment and Risk Mitigation Review(s) X Proprietary Name Review(s) Other Review(s) X Administrative/Correspondence Document(s) X CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: NDA 021995/S-013 APPROVAL LETTER DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration Silver Spring MD 20993 NDA 021995/S-013 SUPPLEMENT APPROVAL Merck & Co., Inc. Attention: Richard J. Swanson, Ph.D. Director, Regulatory Affairs P.O. Box 1000, UG2C-50 North Wales, PA 19454-1099 Dear Dr. Swanson: Please refer to your supplemental new drug application (S-013) dated and received March 5, 2009, submitted under section 505(b) of the Federal Food, Drug, and Cosmetic Act (FDCA) for Januvia (sitagliptin) tablets. We also refer to your supplemental new drug application (b) (4) dated and received November 13, 2009. Your submission of November 13, 2009, also constitutes a complete response to our October 16, 2009, action letter for supplemental application S-013. -
Onglyza (Saxagliptin) Uses, Dosage, Side Effects - Drugs.Com 04/07/2015 Insulin Pump – Tubeless Feel the Freedom for Yourself! Learn About the Omnipod®
Onglyza (saxagliptin) Uses, Dosage, Side Effects - Drugs.com 04/07/2015 Insulin Pump – Tubeless Feel The Freedom For Yourself! Learn About The OmniPod®. Browse all medications A B C D E F G H I J K L M N O P Q R S T U V W X Y Z Advanced Search Phonetic Search Drugs A-Z Pill Identifier Interactions Checker News Health Professionals Q & A Mednotes Apps Home → Conditions → Diabetes, Type 2 → Onglyza Print Share Sign In or Register Onglyza Related Information Availability Pregnancy Category Generic Name: saxagliptin (SAX a GLIP tin) Prescription only No proven risk in Brand Names: Onglyza humans CSA Schedule Not a controlled drug Approval History Drug history at FDA Overview Side Effects Dosage Interactions For Professionals More Reviews Average User Rating Insulin Pump – Tubeless Feel The Freedom For Yourself! Learn About The 9 User Reviews 7.6 Rate it! OmniPod® Drug Class What is Onglyza? Dipeptidyl peptidase 4 inhibitors Onglyza (saxagliptin) is an oral diabetes medicine that helps control blood sugar levels . It works by regulating Related Drugs the levels of insulin your body produces after Diabetes, Type 2 eating. metformin Onglyza is for people with type 2 diabetes. It is insulin aspart Januvia sometimes used in combination with other glipizide diabetes medications, but is not for treating glimepiride type 1 diabetes . Lantus Onglyza may also be used for purposes not Invokana listed in this medication guide. Victoza glyburide Levemir Humalog Actos Important information Janumet You should not use Onglyza if you are in a state of diabetic ketoacidosis (call your doctor for Glucophage treatment with insulin). -

Komboglyze, INN-Saxagliptin, Metformin
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Komboglyze 2.5 mg/850 mg film-coated tablets Komboglyze 2.5 mg/1,000 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Komboglyze 2.5 mg/850 mg film-coated tablets Each tablet contains 2.5 mg of saxagliptin (as hydrochloride) and 850 mg of metformin hydrochloride. Komboglyze 2.5 mg/1,000 mg film-coated tablets Each tablet contains 2.5 mg of saxagliptin (as hydrochloride) and 1,000 mg of metformin hydrochloride. For the full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM Film-coated tablet (tablet). Komboglyze 2.5 mg/850 mg film-coated tablets Light brown to brown, biconvex, round, film-coated tablets, with “2.5/850” printed on one side and “4246” printed on the other side, in blue ink. Komboglyze 2.5 mg/1,000 mg film-coated tablets Pale yellow to light yellow, biconvex, oval shaped, film-coated tablets, with “2.5/1000” printed on one side and “4247” printed on the other side, in blue ink. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Komboglyze is indicated in adults with type 2 diabetes mellitus as an adjunct to diet and exercise to improve glycaemic control: in patients inadequately controlled on their maximally tolerated dose of metformin alone in combination with other medicinal products for the treatment of diabetes, including insulin, in patients inadequately controlled with metformin and these medicinal products (see sections 4.4, 4.5 and 5.1 for available data on different combinations) in patients already being treated with the combination of saxagliptin and metformin as separate tablets. -

Pharmacokinetics of Omarigliptin, a Once-Weekly Dipeptidyl Peptidase-4 Inhibitor
Available online a t www.derpharmachemica.com ISSN 0975-413X Der Pharma Chemica, 2016, 8(12):292-295 CODEN (USA): PCHHAX (http://derpharmachemica.com/archive.html) Mini-review: Pharmacokinetics of Omarigliptin, a Once-weekly Dipeptidyl Peptidase-4 Inhibitor Nermeen Ashoush a,b aClinical Pharmacy and Pharmacy Practice Department, Faculty of Pharmacy, British University in Egypt, El- Sherouk city, Cairo 11837, Egypt. bHead of Health Economics Unit, Center for Drug Research and Development (CDRD), Faculty of Pharmacy, British University in Egypt, El-Sherouk city, Cairo 11837, Egypt. _____________________________________________________________________________________________ ABSTRACT The dipeptidyl peptidase-4 (DPP-4) inhibitors are novel oral hypoglycemic drugs which have been in clinical use for the past 10 years. The drugs are safe, weight neutral and widely prescribed. There are currently many gliptins approved by FDA, namely sitagliptin, vildagliptin, saxagliptin, linagliptin, alogliptin with several more in advanced stages of development. The gliptins may possess cardiovascular protective effects and their administration may promote β-cell survival; claims currently being evaluated in clinical and preclinical studies. The gliptins are an optional second-line therapy after metformin; they are generally well tolerated with low risk of hypoglycemia. The various compounds differ with respect to their pharmacokinetic properties; however, their clinical efficacy appears to be similar. The clinical differences between the various compounds -

TREATMENT of TYPE 2 DIABETES with BIPHASIC INSULIN ANALOGUES *Ali A
TREATMENT OF TYPE 2 DIABETES WITH BIPHASIC INSULIN ANALOGUES *Ali A. Rizvi Professor of Medicine, Department of Medicine and Director, Division of Endocrinology, University of South Carolina School of Medicine, Columbia, South Carolina, USA *Correspondence to [email protected] Disclosure: The author has received grant support, as principal investigator at the University of South Carolina site, from the National Institutes of Health (NIH) for the SPRINT Trial (Contract Number: HHSN268200900040C, ClinicalTrials.gov Identifier: NCT01206062). The contents of this paper do not necessarily represent the views of the NIH. Received: 29.03.16 Accepted: 09.09.16 Citation: EMJ Diabet. 2016;4[1]:74-83. ABSTRACT The majority of patients with Type 2 diabetes require insulin therapy for treating hyperglycaemia. There are several regimens available for insulin initiation and maintenance. Insulin analogues have been developed to mimic normal physiology as closely as possible. Biphasic analogues can target both fasting and postprandial hyperglycaemia, with the added advantage of being premixed and thus convenient for the patient. A practical and feasible option is to initiate insulin with one or more biphasic preparations at mealtimes, thus providing both basal and prandial coverage. Individual titration of dose and frequency of daily injections with biphasic insulin preparations has the potential for improving glycaemic control with a high degree of patient acceptance. Drawbacks include a more rigid regimen, a relative lack of flexibility, and a somewhat higher degree of glycaemic variability and hypoglycaemia when compared to multiple daily basal-bolus injections. Awareness of the advantages and limitations of biphasic insulin analogues can assist clinicians in their appropriate use for the treatment of patients with Type 2 diabetes. -

Vildagliptin/Metformin Hydrochloride Novartis, INN-Vildagliptin/Metformin
European Medicines Agency Evaluation of Medicines for Human Use Doc.Ref.: EMEA/CHMP/656453/2008 ASSESSMENT REPORT FOR Vildagliptin/metformin hydrochloride Novartis International Nonproprietary Name: vildagliptin / metformin hydrochloride Procedure No. EMEA/H/C/001050 Assessment Report as adopted by the CHMP with all information of a commercially confidential nature deleted. 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20) 74 18 84 00 Fax (44-20) 74 18 84 16 E-mail: [email protected] http://www.emea.europa.eu © European Medicines Agency, 2008. Reproduction is authorised provided the source is acknowledged TABLE OF CONTENTS 1. BACKGROUND INFORMATION ON THE PROCEDURE........................................... 3 1.1 Submission of the dossier ........................................................................................................ 3 1.2 Steps taken for the assessment of the product.......................................................................... 3 2. SCIENTIFIC DISCUSSION................................................................................................. 4 2.1 Introduction.............................................................................................................................. 4 2.2 Quality aspects......................................................................................................................... 4 2.3 Non-clinical aspects................................................................................................................. 4 2.4 Clinical -

Sitagliptin: a New Class of Oral Drug for Type 2 Diabetes
JK SCIENCE DRUG REVIEW Sitagliptin: a New Class of Oral Drug for Type 2 Diabetes Dinesh K. Badyal, Jasleen Kaur Introduction Pharmacokinetics Type 2 diabetes is the most common form of the Bioavailability of sitagliptin is approximately 87%. Half- disease, accounting for about 90% to 95 % of all diagnosed life is between 8-14 hours. It is 38% bound to plasma cases of diabetes. In type 2 diabetes, the body does not proteins. It undergoes limited metabolism via CYP3A4 produce enough insulin or the cells ignore the insulin. and CYP2C8. Elimination is mainly through urine (5, 6). Over time, high blood sugar levels can increase the risk Clinical Use for serious complications, including heart disease, In October 2006, the U.S. Food and Drug blindness, nerve damage and kidney damage (1). Any Administration (FDA) approved sitagliptin as new oral hypoglycemic drug that can increase the control monotherapy and as add-on therapy to either of two other of blood glucose with fewer adverse effects in patients types of oral diabetes medications, metformin or with diabetes may be welcomed. Sitagliptin is the first thiazolidinediones to improve blood glucose control in and only prescription medication in a new class of oral patients with type 2 diabetes when diet and exercise are antihyperglycemic agents, which enhance the body's own not enough (5). In March, 2007 it was approved in ability to lower blood glucose when it is elevated (2). European Union. Sitagliptin is currently approved in 42 Mechanism of Action countries (7). The recommended dose of sitagliptin is Sitagliptin prolongs the activity of proteins that increase 100 mg once daily. -
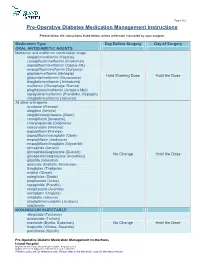
Pre-Operative Diabetes Medication Management Instructions
Page 1 of 2 Pre-Operative Diabetes Medication Management Instructions Please follow the instructions listed below unless otherwise instructed by your surgeon Medication Type Day Before Surgery Day of Surgery ORAL ANTIDIABETIC AGENTS Metformin and metformin combination drugs alogliptin/metformin (Kazano) canagliflozin/metformin (Invokamet) dapagliflozin/metformin (Xigduo XR) empagliflozin/metformin (Synjardy) glipizide/metformin (Metaglip) Hold Evening Dose Hold the Dose glyburide/metformin (Glucovance) linagliptin/metformin (Jentadueto) metformin (Glucophage, Riomet) pioglitazone/metformin (Actoplus Met) repaglidine/metformin (PrandiMet, Repaglin) sitagliptin/metformin (Janumet) All other oral agents acarbose (Precose) alogliptin (Nesina) alogliptin/pioglitazone (Oseni) canagliflozin (Invokana) chlorpropamide (Diabinese) colesevelam (Welchol) dapagliflozin (Farxiga) dapagliflozin/saxagliptin (Qtern) empagliflozin (Jardiance) empagliflozin/linagliptin (Glyxambi) glimepiride (Amaryl) glimepiride/pioglitazone (Duetact) No Change Hold the Dose glimepiride/rosiglitazone (Avandaryl) glipizide (Glucotrol) glyburide (DiaBeta, Micronase) linagliptan (Tradjenta) miglitol (Glyset) nateglinide (Starlix) pioglitazone (Actos) repaglinide (Prandin) rosiglitazone (Avandia) saxagliptin (Onglyza) sitagliptin (Januvia) sitagliptin/simvastatin (Juvisync) tolazamide NON-INSULIN INJECTABLE albiglutide (Tanzeum) dulaglutide (Trulicity) exenatide (Byetta, Bydureon) No Change Hold the Dose liraglutide (Victoza, Saxenda) pramlintide (Symlin) Pre-Operative Diabetic -

OSENI (Alogliptin and Pioglitazone) Tablets II May Increase Risk
HIGHLIGHTS OF PRESCRIBING INFORMATION -----------------------WARNINGS AND PRECAUTIONS--------------------- These highlights do not include all the information needed to use • Congestive heart failure: Fluid retention may occur and can OSENI safely and effectively. See full prescribing information for exacerbate or lead to congestive heart failure. Combination use OSENI. with insulin and use in congestive heart failure NYHA Class I and OSENI (alogliptin and pioglitazone) tablets II may increase risk. Monitor patients for signs and symptoms. Initial U.S. Approval: 2013 (5.1) • Acute pancreatitis: There have been postmarketing reports of WARNING: CONGESTIVE HEART FAILURE acute pancreatitis. If pancreatitis is suspected, promptly See full prescribing information for complete boxed warning discontinue OSENI. (5.2) • Thiazolidinediones, including pioglitazone, cause or • Hypersensitivity: There have been postmarketing reports of exacerbate congestive heart failure in some patients. (5.1) serious hypersensitivity reactions in patients treated with alogliptin • After initiation of OSENI and after dose increases, monitor such as anaphylaxis, angioedema and severe cutaneous adverse patients carefully for signs and symptoms of heart failure reactions. In such cases, promptly discontinue OSENI, assess for (e.g., excessive, rapid weight gain, dyspnea and/or other potential causes, institute appropriate monitoring and edema). If heart failure develops, it should be managed treatment and initiate alternative treatment for diabetes. (5.3) according to current standards of care and • Hepatic effects: Postmarketing reports of hepatic failure, discontinuation or dose reduction of pioglitazone in OSENI sometimes fatal. Causality cannot be excluded. If liver injury is must be considered. detected, promptly interrupt OSENI and assess patient for • OSENI is not recommended in patients with symptomatic probable cause, then treat cause if possible, to resolution or heart failure.