A Lysosomal Enigma CLN5 and Its Significance in Understanding
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The CLN5 Disease
Mia-Lisa Schmiedt Mia-Lisa Schmiedt Mia-Lisa Schmiedt The CLN5 disease − RESEARCH protein maturation, RESEARCH The CLN5 disease − protein maturation, trafficking and pathology trafficking and pathology The CLN5 disease −protein maturation, trafficking and pathology and trafficking maturation, The CLN5 disease −protein Neuronal ceroid lipofuscinoses (NCLs) are a group of hereditary neurode- generative disorders primarily affecting children. Characteristics for NCLs are accumulation of autofluorescent storage material, neuronal degenera- tion, motor disturbances, progressive loss of vision and premature death. One member of the NCL family is the CLN5 disease, a late infantile variant phenotype form, caused by mutations in the CLN5 gene. CLN5 encodes a lysosomal protein of unidentified function. This thesis work contributes to the basic understanding of the molecular and cell biological mechanisms underlying CLN5 disease. Real-time PCR studies indicated that Cln5 gene expression increases gradually in the mouse brain with age and its expres- sion is highest in microglia. This thesis project further presents that the CLN5 protein is cleaved in the ER, trimmed and finally traffics to lysosomes. CLN5 constructs carrying different disease causing mutations revealed that trafficking is disturbed with varying severity depending on the particular mutation. Also, this work provides novel aspects about the early events in the pathogenesis of CLN5 disease, late infantile variant, links Cln5 to lipid metabolism and strengthens the recently reported -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Palmitoyl-Protein Thioesterase 1 Deficiency in Drosophila Melanogaster Causes Accumulation
Genetics: Published Articles Ahead of Print, published on February 1, 2006 as 10.1534/genetics.105.053306 Palmitoyl-protein thioesterase 1 deficiency in Drosophila melanogaster causes accumulation of abnormal storage material and reduced lifespan Anthony J. Hickey*,†,1, Heather L. Chotkowski*, Navjot Singh*, Jeffrey G. Ault*, Christopher A. Korey‡,2, Marcy E. MacDonald‡, and Robert L. Glaser*,†,3 * Wadsworth Center, New York State Department of Health, Albany, NY 12201-2002 † Department of Biomedical Sciences, State University of New York, Albany, NY 12201-0509 ‡ Molecular Neurogenetics Unit, Center for Human Genetic Research, Massachusetts General Hospital, Boston, MA 02114 1 current address: Albany Medical College, Albany, NY 12208 2 current address: Department of Biology, College of Charleston, Charleston, SC 294243 3 corresponding author: Wadsworth Center, NYS Dept. Health, P. O. Box 22002, Albany, NY 12201-2002 E-mail: [email protected] 1 running title: Phenotypes of Ppt1-deficient Drosophila key words: Batten disease infantile neuronal ceroid lipofuscinosis palmitoyl-protein thioesterase CLN1 Drosophila corresponding author: Robert L. Glaser Wadsworth Center, NYS Dept. Health P. O. Box 22002 Albany, NY 12201-2002 E-mail: [email protected] phone: 518-473-4201 fax: 518-474-3181 2 ABSTRACT Human neuronal ceroid lipofuscinoses (NCLs) are a group of genetic neurodegenerative diseases characterized by progressive death of neurons in the central nervous system (CNS) and accumulation of abnormal lysosomal storage material. Infantile NCL (INCL), the most severe form of NCL, is caused by mutations in the Ppt1 gene, which encodes the lysosomal enzyme palmitoyl-protein thioesterase 1 (Ppt1). We generated mutations in the Ppt1 ortholog of Drosophila melanogaster in order to characterize phenotypes caused by Ppt1-deficiency in flies. -

Perkinelmer Genomics to Request the Saliva Swab Collection Kit for Patients That Cannot Provide a Blood Sample As Whole Blood Is the Preferred Sample
Progressive Myoclonic Epilepsy Panel Test Code D4004 Test Summary This test analyzes 18 genes that have been associated with Progressive Myoclonic Epilepsy Turn-Around-Time (TAT)* 3 - 5 weeks Acceptable Sample Types DNA, Isolated Dried Blood Spots Saliva Whole Blood (EDTA) Acceptable Billing Types Self (patient) Payment Institutional Billing Commercial Insurance Indications for Testing The early way to tell the difference is an EEG with background slowing. Symptoms like stimulus induced myoclonic jerks, cognitive decline and motor slowing, generalized tonic-clonic seizures, or visual/occipital seizures help narrow the diagnosis. Most importantly, the presence of slowing on the EEG should raise suspicion for PME and, if present, lead to further testing, including genetic and enzyme testing. Test Description This panel analyzes 18 genes that have been associated with Progressive Myoclonic Epilepsy and/or disorders associated with epilepsy. Both sequencing and deletion/duplication (CNV) analysis will be performed on the coding regions of all genes included (unless otherwise marked). All analysis is performed utilizing Next Generation Sequencing (NGS) technology. CNV analysis is designed to detect the majority of deletions and duplications of three exons or greater in size. Smaller CNV events may also be detected and reported, but additional follow-up testing is recommended if a smaller CNV is suspected. All variants are classified according to ACMG guidelines. Condition Description Progressive myoclonic epilepsies (PME) are a group of more than 10 rare types of epilepsy that are “progressive.” People with PME have a decline in motor skills, balance and cognitive function over time. Myoclonus indicates frequent muscle jerks, both spontaneous and often stimulus induced. -

A Study of Neuronal Ceroid Lipofuscinosis Proteins Cln5 and Cln8
A STUDY OF NEURONAL CEROID LIPOFUSCINOSIS PROTEINS CLN5 AND CLN8 By W A BHAGYA NILUKSHI DE SILVA B. S., University of Colombo, Sri Lanka, 2011 A THESIS Submitted in partial fulfillment of the requirements for the degree MASTER OF SCIENCE Department of Biochemistry and Molecular Biophysics College of Arts and Sciences KANSAS STATE UNIVERSITY Manhattan, Kansas 2015 Approved by: Major Professor Dr. Stella Y. Lee ABSTRACT Neuronal ceroid lipofuscinoses (NCLs) are a group of neurodegenerative lysosomal storage disorders which is the most frequent group of inherited neurodegenerative disorders that affect children leading to severe pathological conditions such as progressive loss of motor neuron functions, loss of vision, mental retardation, epilepsy, ataxia and atrophy in cerebral, cerebella cortex and retina and eventually premature death. Among the many genes that cause NCL, mutations in CLN5 leads to different forms of NCL (infantile, late infantile, juvenile and adult) and mutations in CLN8 leads to progressive epilepsy with mental retardation (EPMR) and a variant late infantile form of NCL. The function(s) of both CLN5 and CLN8 proteins remain elusive. CLN5 is a glycosylated soluble protein that resides in the lysosome. We observed that endogenous CLN5 protein exist in two forms and identified a previously unknown C-terminal proteolytic processing event of CLN5. Using a cycloheximide chase experiment we demonstrated that the proteolytic processing of CLN5 is a post-translational modification. Furthermore treatment with chloroquine showed the processing occurs in low pH cellular compartments. After treatment with different protease inhibitors our results suggested the protease involved in the processing of CLN5 could be a cysteine protease. -

Human Induced Pluripotent Stem Cell–Derived Podocytes Mature Into Vascularized Glomeruli Upon Experimental Transplantation
BASIC RESEARCH www.jasn.org Human Induced Pluripotent Stem Cell–Derived Podocytes Mature into Vascularized Glomeruli upon Experimental Transplantation † Sazia Sharmin,* Atsuhiro Taguchi,* Yusuke Kaku,* Yasuhiro Yoshimura,* Tomoko Ohmori,* ‡ † ‡ Tetsushi Sakuma, Masashi Mukoyama, Takashi Yamamoto, Hidetake Kurihara,§ and | Ryuichi Nishinakamura* *Department of Kidney Development, Institute of Molecular Embryology and Genetics, and †Department of Nephrology, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan; ‡Department of Mathematical and Life Sciences, Graduate School of Science, Hiroshima University, Hiroshima, Japan; §Division of Anatomy, Juntendo University School of Medicine, Tokyo, Japan; and |Japan Science and Technology Agency, CREST, Kumamoto, Japan ABSTRACT Glomerular podocytes express proteins, such as nephrin, that constitute the slit diaphragm, thereby contributing to the filtration process in the kidney. Glomerular development has been analyzed mainly in mice, whereas analysis of human kidney development has been minimal because of limited access to embryonic kidneys. We previously reported the induction of three-dimensional primordial glomeruli from human induced pluripotent stem (iPS) cells. Here, using transcription activator–like effector nuclease-mediated homologous recombination, we generated human iPS cell lines that express green fluorescent protein (GFP) in the NPHS1 locus, which encodes nephrin, and we show that GFP expression facilitated accurate visualization of nephrin-positive podocyte formation in -

Enhanced Expression of Manganese-Dependent Superoxide Dismutase in Human and Sheep CLN6 Tissues
Biochem. J. (2003) 376, 369–376 (Printed in Great Britain) 369 Enhanced expression of manganese-dependent superoxide dismutase in human and sheep CLN6 tissues Claudia HEINE*, Jaana TYYNELA¨†, Jonathan D. COOPER‡,David N. PALMER§,Milan ELLEDER,Alfried KOHLSCHUTTER*¨ and Thomas BRAULKE*1 *Children’s Hospital, University of Hamburg, 20246 Hamburg, Germany, †Institute of Biomedicine/Biochemistry, University of Helsinki, 00014 Helsinki, Finland, ‡Institute of Psychiatry, King’s College London, London SE5 8AF, U.K., §Animal and Food Sciences Division, Lincoln University, Canterbury, New Zealand, and Institute of Inherited Metabolic Disorders, Charles University Prague, 121 11 Prague 2, Czech Republic Neuronal ceroid lipofuscinosis type 6 and its sheep model (OCL6) fluorescence microscopy and immunohistochemical studies re- are lysosomal storage disorders caused by mutations in the CLN6 vealed the presence of MnSOD in mitochondria of CLN6 fi- gene product of unknown function. It has been proposed that mito- broblasts and in CLN6 brain sections within both neurons and chondrial dysfunction, including defects in mitochondrial protein hypertrophic astrocytes. These data suggest that oxidative stress degradation, organelle enlargement and functional changes in oxi- and/or the production of pro-inflammatory cytokines are charac- dative phosphorylation, may contribute to the disease pathology. teristic features of human and sheep CLN6, resulting in elevated To further explore the disease mechanisms underlying CLN6, pro- expression of MnSOD, which may be important for diagnostic tein expression was compared between normal and affected tis- purposes. sues. Using two-dimensional electrophoretic separation of pro- teins, MS and immunoblotting, MnSOD (manganese-dependent superoxide dismutase) was found to be significantly and speci- Key words: fluorescence microscopy, lysosomal storage dis- fically increased in fibroblasts and brain extracts of both human order, manganese-dependent superoxide dismutase (MnSOD), and sheep affected with CLN6. -

Invitae Detect Lysosomal Storage Diseases (Lsds) Genetic Testing Program
Invitae Detect Lysosomal Storage Diseases (LSDs) Genetic Testing Program SPONSORED, NO-CHARGE GENETIC TESTING AND COUNSELING FOR PATIENTS SUSPECTED OF HAVING AN LSD WHAT ARE LSDs? • Progressive, multisystemic inherited metabolic diseases • Associated with high morbidity and mortality • Approximately 50 known LSDs PROGRAM ELIGIBILITY To qualify for no-charge testing through the Invitae Detect The Invitae Detect LSDs program offers sponsored no-charge genetic LSDs program, the individual testing and counseling to diagnose LSDs, bringing patients closer to must be suspected of having an an accurate diagnosis and appropriate clinical management. LSD based on at least one of the following: Influence clinical outcomes Impact quality of life • Clinical features • Suspicion of, or known Diagnose LSDs faster to enable Get patients the right information diagnosis of, a specific LSD improved clinical outcomes. quicker to regain valuable • Family history related to LSDs treatment time. • Lab result suggestive of LSDs Break down barriers to Give patients more • Presumptive positive genetic testing than just test results newborn screening Both testing and counseling If the test is positive, the for this program are sponsored, proband’s blood relatives are no‑charge offerings. eligible for the program using program code LYSO. GENETIC TESTING WITH INVITAE Invitae offers LSDs testing with multiple panels as well as with single genes: • Comprehensive Lysosomal Storage Disorders Panel* • Comprehensive Neuromuscular Disorders Panel • Comprehensive Mucopolysaccharidoses (MPS) Panel • Cardiomyopathy Comprehensive Panel • Or test the following single genes: AGA CLN6 FUCA1 GM2A HEXB LAMP2 NAGA PSAP ARSA CLN8 GAA GNPTAB HGSNAT LIPA NAGLU SGSH ARSB CTNS GALC GNPTG HYAL1 MAN2B1 NEU1 SLC17A5 ASAH1 CTSA GALNS GNS IDS MANBA NPC1 SMPD1 CLN2(TPP1) CTSD GLA GUSB IDUA MCOLN1 NPC2 SUMF1 CLN5 CTSK GLB1 HEXA KCTD7 MFSD8 PPT1 *This panel does not currently test for Gaucher disease. -
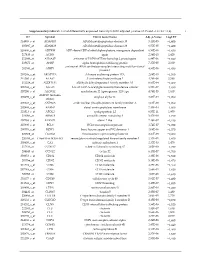
Supplementary Table S1. List of Differentially Expressed
Supplementary table S1. List of differentially expressed transcripts (FDR adjusted p‐value < 0.05 and −1.4 ≤ FC ≥1.4). 1 ID Symbol Entrez Gene Name Adj. p‐Value Log2 FC 214895_s_at ADAM10 ADAM metallopeptidase domain 10 3,11E‐05 −1,400 205997_at ADAM28 ADAM metallopeptidase domain 28 6,57E‐05 −1,400 220606_s_at ADPRM ADP‐ribose/CDP‐alcohol diphosphatase, manganese dependent 6,50E‐06 −1,430 217410_at AGRN agrin 2,34E‐10 1,420 212980_at AHSA2P activator of HSP90 ATPase homolog 2, pseudogene 6,44E‐06 −1,920 219672_at AHSP alpha hemoglobin stabilizing protein 7,27E‐05 2,330 aminoacyl tRNA synthetase complex interacting multifunctional 202541_at AIMP1 4,91E‐06 −1,830 protein 1 210269_s_at AKAP17A A‐kinase anchoring protein 17A 2,64E‐10 −1,560 211560_s_at ALAS2 5ʹ‐aminolevulinate synthase 2 4,28E‐06 3,560 212224_at ALDH1A1 aldehyde dehydrogenase 1 family member A1 8,93E‐04 −1,400 205583_s_at ALG13 ALG13 UDP‐N‐acetylglucosaminyltransferase subunit 9,50E‐07 −1,430 207206_s_at ALOX12 arachidonate 12‐lipoxygenase, 12S type 4,76E‐05 1,630 AMY1C (includes 208498_s_at amylase alpha 1C 3,83E‐05 −1,700 others) 201043_s_at ANP32A acidic nuclear phosphoprotein 32 family member A 5,61E‐09 −1,760 202888_s_at ANPEP alanyl aminopeptidase, membrane 7,40E‐04 −1,600 221013_s_at APOL2 apolipoprotein L2 6,57E‐11 1,600 219094_at ARMC8 armadillo repeat containing 8 3,47E‐08 −1,710 207798_s_at ATXN2L ataxin 2 like 2,16E‐07 −1,410 215990_s_at BCL6 BCL6 transcription repressor 1,74E‐07 −1,700 200776_s_at BZW1 basic leucine zipper and W2 domains 1 1,09E‐06 −1,570 222309_at -

A New Large Animal Model of CLN5
NIH Public Access Author Manuscript Neurobiol Dis. Author manuscript; available in PMC 2009 February 1. NIH-PA Author ManuscriptPublished NIH-PA Author Manuscript in final edited NIH-PA Author Manuscript form as: Neurobiol Dis. 2008 February ; 29(2): 306±315. A new large animal model of CLN5 neuronal ceroid lipofuscinosis in Borderdale sheep is caused by a nucleotide substitution at a consensus splice site (c.571+1G>A) leading to excision of exon 3 Tony Frugier1, Nadia L. Mitchell1, Imke Tammen2, Peter J. Houweling2, Donald G. Arthur3, Graham W. Kay1, Otto P. van Diggelen4, Robert D. Jolly5, and David N. Palmer1,# 1 Lincoln University, Agriculture and Life Sciences Division, Cell Biology Group, PO Box 84, Lincoln 7647, Canterbury, New Zealand 2 Centre for Advanced Technologies in Animal Genetics and Reproduction (Reprogen), Faculty of Veterinary Science, The University of Sydney, PMB3, Camden NSW 2570, Australia 3 Selwyn Rakaia Vet Services, PO Box 52, Dunsandel 8190, New Zealand 4 Department of Clinical Genetics, Erasmus University Medical Centre, Dr. Molewaterplein 50, 3015 GE Rotterdam, The Netherlands 5 Institute of Veterinary, Animal and Biomedical Sciences, Massey University, Private Bag 11222, Palmerston North, New Zealand Abstract Batten disease (neuronal ceroid lipofuscinoses, NCLs) are a group of inherited childhood diseases that result in severe brain atrophy, blindness and seizures, leading to premature death. To date eight different genes have been identified, each associated with a different form. Linkage analysis indicated a CLN5 form in a colony of affected New Zealand Borderdale sheep. Sequencing studies established the disease-causing mutation to be a substitution at a consensus splice site (c.571+G>A), leading to the excision of exon 3 and a truncated putative protein. -

Disorders of Sphingolipid Synthesis, Sphingolipidoses, Niemann-Pick Disease Type C and Neuronal Ceroid Lipofuscinoses
551 38 Disorders of Sphingolipid Synthesis, Sphingolipidoses, Niemann-Pick Disease Type C and Neuronal Ceroid Lipofuscinoses Marie T. Vanier, Catherine Caillaud, Thierry Levade 38.1 Disorders of Sphingolipid Synthesis – 553 38.2 Sphingolipidoses – 556 38.3 Niemann-Pick Disease Type C – 566 38.4 Neuronal Ceroid Lipofuscinoses – 568 References – 571 J.-M. Saudubray et al. (Eds.), Inborn Metabolic Diseases, DOI 10.1007/978-3-662-49771-5_ 38 , © Springer-Verlag Berlin Heidelberg 2016 552 Chapter 38 · Disor ders of Sphingolipid Synthesis, Sphingolipidoses, Niemann-Pick Disease Type C and Neuronal Ceroid Lipofuscinoses O C 22:0 (Fatty acid) Ganglio- series a series b HN OH Sphingosine (Sphingoid base) OH βββ β βββ β Typical Ceramide (Cer) -Cer -Cer GD1a GT1b Glc ββββ βββ β Gal -Cer -Cer Globo-series GalNAc GM1a GD1b Neu5Ac βαββ -Cer Gb4 ββ β ββ β -Cer -Cer αβ β -Cer GM2 GD2 Sphingomyelin Pcholine-Cer Gb3 B4GALNT1 [SPG46] [SPG26] β β β ββ ββ CERS1-6 GBA2 -Cer -Cer ST3GAL5 -Cer -Cer So1P So Cer GM3 GD3 GlcCer - LacCer UDP-Glc UDP Gal CMP -Neu5Ac - UDP Gal PAPS Glycosphingolipids GalCer Sulfatide ββ Dihydro -Cer -Cer SO 4 Golgi Ceramide apparatus 2-OH- 2-OH-FA Acyl-CoA FA2H CERS1-6 [SPG35] CYP4F22 ω-OH- ω-OH- FA Acyl-CoA ULCFA ULCFA-CoA ULCFA GM1, GM2, GM3: monosialo- Sphinganine gangliosides Endoplasmic GD3, GD2, GD1a, GD1b: disialo-gangliosides reticulum KetoSphinganine GT1b: trisialoganglioside SPTLC1/2 [HSAN1] N-acetyl-neuraminic acid: sialic acid found in normal human cells Palmitoyl-CoA Deoxy-sphinganine + Serine +Ala or Gly Deoxymethylsphinganine 38 . Fig. 38.1 Schematic representation of the structure of the main sphingolipids , and their biosynthetic pathways. -

Molecular Pathophysiology Underlying Neuronal Ceroid Lipofuscinoses: Cln2 and Cln5
MOLECULAR PATHOPHYSIOLOGY UNDERLYING NEURONAL CEROID LIPOFUSCINOSES: CLN2 AND CLN5 CARLOS JORGE PEREIRA BESSA Dissertação de doutoramento em Ciências Biomédicas 2009 Carlos Jorge Pereira Bessa Molecular Pathophysiology Underlying Neuronal Ceroid Lipofuscinoses: CLN2 and CLN5 Dissertação de Candidatura ao grau de Doutor em Ciências Biomédicas submetida ao Instituto de Ciências Biomédicas de Abel Salazar da Universidade do Porto. Orientador – Doutora Maria Gil Roseira Ribeiro Categoria – Técnica Superior de Saúde Afiliação – Instituto Nacional de Saúde Dr. Ricardo Jorge, Centro de Genética Médica Jacinto Magalhães Co-orientador – Doutor Jorge Eduardo da Silva Azevedo Categoria – Professor Catedrático Afiliação – Instituto de Ciências Biomédicas Abel Salazar da Universidade do Porto Legal precepts Preceitos legais In accord with the 2nd of the 8th article of Decreto-lei nº 388/70, the experimental results stated bellow in Publications I/II/III were used in this dissertation. The author of this dissertation declares his participation in the outlining and execution of the experimental work, in the data presentation and preparation of the work referenced below under the name Bessa, C / Carlos Bessa: De acordo com o disposto no nº2 do artigo 8º do Decreto-lei nº388/70, nesta dissertação foram utilizados os resultados dos trabalhos publicados abaixo referenciados. No cumprimento do disposto no referido Decreto-lei, o autor desta dissertação declara que interveio na concepção e execução do trabalho experimental, na interpretação e redacção dos resultados publicados sob o nome Bessa, C / Carlos Bessa: Publication I / Publicação I Bessa C, Teixeira CA, Dias A, Alves M, Rocha S, Lacerda L, Loureiro L, Guimarães A, Ribeiro MG. CLN2/TPP1 deficiency: the novel mutation IVS7-10A>G causes intron retention and is associated with a mild disease phenotype.