The Effect of Diuretics on Patients with Heart Failure: a Network Meta- Analysis: Diuretics Effect on Heart Failure Patients
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Stems for Nonproprietary Drug Names
USAN STEM LIST STEM DEFINITION EXAMPLES -abine (see -arabine, -citabine) -ac anti-inflammatory agents (acetic acid derivatives) bromfenac dexpemedolac -acetam (see -racetam) -adol or analgesics (mixed opiate receptor agonists/ tazadolene -adol- antagonists) spiradolene levonantradol -adox antibacterials (quinoline dioxide derivatives) carbadox -afenone antiarrhythmics (propafenone derivatives) alprafenone diprafenonex -afil PDE5 inhibitors tadalafil -aj- antiarrhythmics (ajmaline derivatives) lorajmine -aldrate antacid aluminum salts magaldrate -algron alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol combined alpha and beta blockers labetalol medroxalol -amidis antimyloidotics tafamidis -amivir (see -vir) -ampa ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) subgroup: -ampanel antagonists becampanel -ampator modulators forampator -anib angiogenesis inhibitors pegaptanib cediranib 1 subgroup: -siranib siRNA bevasiranib -andr- androgens nandrolone -anserin serotonin 5-HT2 receptor antagonists altanserin tropanserin adatanserin -antel anthelmintics (undefined group) carbantel subgroup: -quantel 2-deoxoparaherquamide A derivatives derquantel -antrone antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine antineoplastics (arabinofuranosyl derivatives) fazarabine fludarabine aril-, -aril, -aril- antiviral (arildone derivatives) pleconaril arildone fosarilate -arit antirheumatics (lobenzarit type) lobenzarit clobuzarit -arol anticoagulants (dicumarol type) dicumarol -

Partial Agreement in the Social and Public Health Field
COUNCIL OF EUROPE COMMITTEE OF MINISTERS (PARTIAL AGREEMENT IN THE SOCIAL AND PUBLIC HEALTH FIELD) RESOLUTION AP (88) 2 ON THE CLASSIFICATION OF MEDICINES WHICH ARE OBTAINABLE ONLY ON MEDICAL PRESCRIPTION (Adopted by the Committee of Ministers on 22 September 1988 at the 419th meeting of the Ministers' Deputies, and superseding Resolution AP (82) 2) AND APPENDIX I Alphabetical list of medicines adopted by the Public Health Committee (Partial Agreement) updated to 1 July 1988 APPENDIX II Pharmaco-therapeutic classification of medicines appearing in the alphabetical list in Appendix I updated to 1 July 1988 RESOLUTION AP (88) 2 ON THE CLASSIFICATION OF MEDICINES WHICH ARE OBTAINABLE ONLY ON MEDICAL PRESCRIPTION (superseding Resolution AP (82) 2) (Adopted by the Committee of Ministers on 22 September 1988 at the 419th meeting of the Ministers' Deputies) The Representatives on the Committee of Ministers of Belgium, France, the Federal Republic of Germany, Italy, Luxembourg, the Netherlands and the United Kingdom of Great Britain and Northern Ireland, these states being parties to the Partial Agreement in the social and public health field, and the Representatives of Austria, Denmark, Ireland, Spain and Switzerland, states which have participated in the public health activities carried out within the above-mentioned Partial Agreement since 1 October 1974, 2 April 1968, 23 September 1969, 21 April 1988 and 5 May 1964, respectively, Considering that the aim of the Council of Europe is to achieve greater unity between its members and that this -

Azosemide Is More Potent Than Bumetanide and Various Other Loop
www.nature.com/scientificreports OPEN Azosemide is more potent than bumetanide and various other loop diuretics to inhibit the sodium- Received: 21 November 2017 Accepted: 14 June 2018 potassium-chloride-cotransporter Published: xx xx xxxx human variants hNKCC1A and hNKCC1B Philip Hampel 1,2, Kerstin Römermann1, Nanna MacAulay 3 & Wolfgang Löscher1,2 The Na+–K+–2Cl− cotransporter NKCC1 plays a role in neuronal Cl− homeostasis secretion and represents a target for brain pathologies with altered NKCC1 function. Two main variants of NKCC1 have been identifed: a full-length NKCC1 transcript (NKCC1A) and a shorter splice variant (NKCC1B) that is particularly enriched in the brain. The loop diuretic bumetanide is often used to inhibit NKCC1 in brain disorders, but only poorly crosses the blood-brain barrier. We determined the sensitivity of the two human NKCC1 splice variants to bumetanide and various other chemically diverse loop diuretics, using the Xenopus oocyte heterologous expression system. Azosemide was the most potent NKCC1 inhibitor (IC50s 0.246 µM for hNKCC1A and 0.197 µM for NKCC1B), being about 4-times more potent than bumetanide. Structurally, a carboxylic group as in bumetanide was not a prerequisite for potent NKCC1 inhibition, whereas loop diuretics without a sulfonamide group were less potent. None of the drugs tested were selective for hNKCC1B vs. hNKCC1A, indicating that loop diuretics are not a useful starting point to design NKCC1B-specifc compounds. Azosemide was found to exert an unexpectedly potent inhibitory efect and as a non-acidic compound, it is more likely to cross the blood-brain barrier than bumetanide. Te Na+–K+–2Cl− cotransporter NKCC1 (encoded by SLC12A2) plays an important role in Cl- uptake in neu- rons both in developing brain and in adult sensory neurons1,2. -

Simultaneous Determination of 5 Diuretic Drugs Using an HPLC Method by Quantitative Analysis of Multiple Components by a Single Marker
Simultaneous determination of 5 diuretic drugs using an HPLC method by quantitative analysis of multiple components by a single marker Fuchao Chen Hubei University of Medicine Baoxia Fang Hubei University of Medicine Peng Li ( [email protected] ) Hubei University of Medicine Sicen Wang Xi'an Jiaotong University Research Article Keywords: Diuretic drug, Multi-components detection with a single marker, HPLC, Method development Posted Date: April 8th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-397940/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Simultaneous determination of 5 diuretic drugs using an HPLC method by quantitative analysis of multiple components by a single marker Fuchao Chen 1, Baoxia Fang 1, Peng Li 1 *, and Sicen Wang 2 * 1 Sinopharm Dongfeng General Hospital, Hubei University of Medicine, Shiyan, Hubei, 442008, P.R. China. 2 School of Pharmacy, Xi'an Jiaotong University, Xi'an, 710061, Shanxi, PR China. Fu-chao Chen and Baoxia Fang contributed equally to this work and should be considered as equal first authors. Correspondence to: Peng Li, E- mail: [email protected]. Sinopharm Dongfeng General Hospital, Hubei University of Medicine, Shiyan, Hubei, 442008, P.R. China.Sicen Wang, E- mail: [email protected]. School of Pharmacy, Xi'an Jiaotong University, Xi'an, 710061, Shanxi, PR China. Abstract: Background: Loop diuretics are commonly used in clinical practice to manage high fluid loads and to control fluid balance. In this paper, a novel quantitative analysis method for multiple components with a single marker (QAMS) was developed for the simultaneous determination of 5 diuretic drugs furosemide, torasemide, azosemide, etacrynic acid, and bumetanide, by HPLC. -

Tepzz 77889A T
(19) TZZ T (11) EP 2 277 889 A2 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.: 26.01.2011 Bulletin 2011/04 C07K 1/00 (2006.01) C12P 21/04 (2006.01) C12P 21/06 (2006.01) A01N 37/18 (2006.01) (2006.01) (2006.01) (21) Application number: 10075466.2 G01N 31/00 C07K 14/765 C12N 15/62 (2006.01) (22) Date of filing: 23.12.2002 (84) Designated Contracting States: • Novozymes Biopharma UK Limited AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Nottingham NG7 1FD (GB) IE IT LI LU MC NL PT SE SI SK TR (72) Inventors: (30) Priority: 21.12.2001 US 341811 P • Ballance, David James 24.01.2002 US 350358 P Berwyn, PA 19312 (US) 28.01.2002 US 351360 P • Turner, Andrew John 26.02.2002 US 359370 P King of Prussia, PA 19406 (US) 28.02.2002 US 360000 P • Rosen, Craig A. 27.03.2002 US 367500 P Laytonsville, MD 20882 (US) 08.04.2002 US 370227 P • Haseltine, William A. 10.05.2002 US 378950 P Washington, DC 20007 (US) 24.05.2002 US 382617 P • Ruben, Steven M. 28.05.2002 US 383123 P Brookeville, MD 20833 (US) 05.06.2002 US 385708 P 10.07.2002 US 394625 P (74) Representative: Bassett, Richard Simon et al 24.07.2002 US 398008 P Potter Clarkson LLP 09.08.2002 US 402131 P Park View House 13.08.2002 US 402708 P 58 The Ropewalk 18.09.2002 US 411426 P Nottingham 18.09.2002 US 411355 P NG1 5DD (GB) 02.10.2002 US 414984 P 11.10.2002 US 417611 P Remarks: 23.10.2002 US 420246 P •ThecompletedocumentincludingReferenceTables 05.11.2002 US 423623 P and the Sequence Listing can be downloaded from the EPO website (62) Document number(s) of the earlier application(s) in •This application was filed on 21-09-2010 as a accordance with Art. -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -

SUPPLEMENTARY MATERIAL 1: Search Strategy
SUPPLEMENTARY MATERIAL 1: Search Strategy Medline search strategy 1. exp basal ganglia hemorrhage/ or intracranial hemorrhages/ or cerebral hemorrhage/ or intracranial hemorrhage, hypertensive/ or cerebrovascular disorders/ 2. ((brain$ or cerebr$ or cerebell$ or intracerebral or intracran$ or parenchymal or intraparenchymal or intraventricular or infratentorial or supratentorial or basal gangli$ or putaminal or putamen or posterior fossa or hemispher$ or pon$ or lentiform$ or brainstem or cortic$ or cortex$ or subcortic$ or subcortex$) adj5 (h?emorrhag$ or h?ematoma$ or bleed$)).tw 3. ((hemorrhag$ or haemorrhag$) adj6 (stroke$ or apoplex$ or cerebral vasc$ or cerebrovasc$ or cva)).tw 4. (ICH or ICHs or PICH or PICHs).tw 5. 1 or 2 or 3 or 4 6. exp blood pressure/ 7. exp hypertension/ 8. (blood pressure or bloodpressure).tw 9. ((bp or blood pressure) adj5 (lowering or reduc$)).tw 10. ((strict$ or target$ or tight$ or intens$ or below) adj3 (blood pressure or systolic or diastolic or bp or level$)).tw 11. (hypertension or hypertensive).tw 12. ((manage$ or monitor$) adj3 (hypertension or blood pressure)).tw 13. ((intense or intensive or aggressive or accelerated or profound or radical or severe) adj5 ((bp or blood pressure) adj5 (lowering or reduc$ or decreas$ or decrement or dimin$ or declin$))).tw 14. ((standard or normal or ordinary or guideline or guide line or guideline recommend$ or recommend$ or convention$ or usual or established) adj5 ((bp or blood pressure) adj5 (lowering or reduc$ or decreas$ or decrement or dimin$ or declin$))).tw 15. (antihypertensive adj2 (agent$ or drug$ or medicat$)).tw 1 16. 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 17. -
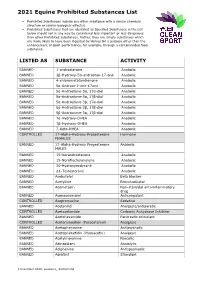
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

Superiority of Long-Acting to Short-Acting Loop Diuretics in The
Advance Publication by J-STAGE Circulation Journal LATE BREAKING CLINICAL TRIAL (JCS 2012) Official Journal of the Japanese Circulation Society http://www.j-circ.or.jp Superiority of Long-Acting to Short-Acting Loop Diuretics in the Treatment of Congestive Heart Failure – The J-MELODIC Study – Tohru Masuyama, MD; Takeshi Tsujino, MD; Hideki Origasa, PhD; Kazuhiro Yamamoto, MD; Takashi Akasaka, MD; Yutaka Hirano, MD; Nobuyuki Ohte, MD; Takashi Daimon, PhD; Satoshi Nakatani, MD; Hiroshi Ito, MD Background: Diuretics are the most prescribed drug in heart failure (HF) patients. However, clinical evidence about their long-term effects is lacking. The purpose of this study was to compare the therapeutic effects of furosemide and azosemide, a short- and long-acting loop diuretic, respectively, in patients with chronic heart failure (CHF). Methods and Results: In this multicenter, prospective, randomized, open, blinded endpoint trial, we compared the effects of azosemide and furosemide in patients with CHF and New York Heart Association class II or III symptoms. 320 patients (160 patients in each group, mean age 71 years) were followed up for a minimum of 2 years. The pri- mary endpoint was a composite of cardiovascular death or unplanned admission to hospital for congestive HF. During a median follow-up of 35.2 months, the primary endpoint occurred in 23 patients in the azosemide group and in 34 patients in the furosemide group (hazard ratio [HR], 0.55, 95% confidence interval [CI] 0.32–0.95: P=0.03). Among the secondary endpoints, unplanned admission to hospital for congestive HF or a need for modification of the treatment for HF were also reduced in the azosemide group compared with the furosemide group (HR, 0.60, 95%CI 0.36–0.99: P=0.048). -

Role of Bumetanide on Insulin Secretion
Wright State University CORE Scholar Browse all Theses and Dissertations Theses and Dissertations 2014 Role of Bumetanide on Insulin Secretion Mohammed Mashari Almutairi Wright State University Follow this and additional works at: https://corescholar.libraries.wright.edu/etd_all Part of the Pharmacology, Toxicology and Environmental Health Commons Repository Citation Almutairi, Mohammed Mashari, "Role of Bumetanide on Insulin Secretion" (2014). Browse all Theses and Dissertations. 2030. https://corescholar.libraries.wright.edu/etd_all/2030 This Thesis is brought to you for free and open access by the Theses and Dissertations at CORE Scholar. It has been accepted for inclusion in Browse all Theses and Dissertations by an authorized administrator of CORE Scholar. For more information, please contact [email protected]. ! ! ! ! ! ! Role%of%Bumetanide%on%Insulin%Secretion% % ! ! ! A!thesis!submitted!in!partial!fulfillment! of!the!requirements!for!the!degree!of!! Master!of!Science! ! ! By! ! MOHAMMED!MASHARI!ALMUTAIRI! ! B.!Pharm,!King!Saud!University,!Riyadh,!Saudi!Arabia!2010! ! ! ! ! ! 2014! ! Wright!State!University! WRIGHT STATE UNIVERSITY GRADUATE SCHOOL Date: July 22, 2014 I HEREBY RECOMMEND THAT THE THESIS PREPARED UNDER MY SUPERVISION BY Mohammed M Almutairi ENTITLED “Role of Bumetanide on Insulin Secretion” BE ACCEPTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF Master of Science. Mauricio Di Fulvio, Ph.D. Thesis Director Norma Adragna, Ph.D. Interim Chair Department of Pharmacology and Toxicology Committee on Final Examination Mauricio Di Fulvio, Ph.D. Norma Adragna, Ph.D. David Cool, Ph.D. Robert E. W. Fyffe, Ph.D. Vice President for Research and Dean of the Graduate School ABSTRACT Almutairi, Mohammed. M.S., Department of Pharmacology and Toxicology, Wright State University, Boonshoft School of Medicine, 2014. -

Decision Trees to Characterise the Roles of Permeability and Solubility on the Prediction of Oral Absorption
Decision trees to characterise the roles of permeability and solubility on the prediction of oral absorption Article (Accepted Version) Newby, Danielle, Freitas, Alex A and Ghafourian, Taravat (2015) Decision trees to characterise the roles of permeability and solubility on the prediction of oral absorption. European Journal of Medicinal Chemistry, 90. pp. 751-765. ISSN 0223-5234 This version is available from Sussex Research Online: http://sro.sussex.ac.uk/id/eprint/64123/ This document is made available in accordance with publisher policies and may differ from the published version or from the version of record. If you wish to cite this item you are advised to consult the publisher’s version. Please see the URL above for details on accessing the published version. Copyright and reuse: Sussex Research Online is a digital repository of the research output of the University. Copyright and all moral rights to the version of the paper presented here belong to the individual author(s) and/or other copyright owners. To the extent reasonable and practicable, the material made available in SRO has been checked for eligibility before being made available. Copies of full text items generally can be reproduced, displayed or performed and given to third parties in any format or medium for personal research or study, educational, or not-for-profit purposes without prior permission or charge, provided that the authors, title and full bibliographic details are credited, a hyperlink and/or URL is given for the original metadata page and the content is not changed in any way. http://sro.sussex.ac.uk Accepted Manuscript Decision trees to characterise the roles of permeability and solubility on the prediction of oral absorption Danielle Newby , Alex. -

READY: Relative Efficacy of Loop Diuretics in Patients with Chronic Systolic Heart Failure—A Systematic Review and Network Meta-Analysis of Randomised Trials
Heart Failure Reviews (2019) 24:461–472 https://doi.org/10.1007/s10741-019-09771-8 READY: relative efficacy of loop diuretics in patients with chronic systolic heart failure—a systematic review and network meta-analysis of randomised trials Tobias Täger1 & Hanna Fröhlich 1 & Mirjam Seiz1 & Hugo A. Katus1 & Lutz Frankenstein1 Published online: 14 March 2019 # Springer Science+Business Media, LLC, part of Springer Nature 2019 Abstract The majority of patients with chronic heart failure (HF) receive long-term treatment with loop diuretics. The comparative effectiveness of different loop diuretics is unknown. We searched PubMed, clinicaltrials.gov, the Cochrane Central Register of Controlled Trials and the European Union Clinical Trials Register for randomised clinical trials exploring the efficacy of the loop diuretics azosemide, bumetanide, furosemide or torasemide in patients with HF. Comparators included placebo, standard medical care or any other active treatment. The primary endpoint was all-cause mortality. Secondary endpoints included cardiovascular mortality, HF-related hospitalisation and any combined endpoint thereof. Hypokalaemia and acute renal failure were defined as additional safety endpoints. Evidence was synthesised using network meta-analysis (NMA). Thirty-four trials reporting on 2647 patients were included. The overall quality of evidence was rated as moderate. NMA demonstrated no significant differences between loop diuretics with respect to all-cause mortality, cardiovascular mortality or hypokalaemia. In contrast, torasemide ranked best in terms of HF hospitalisation, and there was a trend towards benefits with torasemide with regard to occurrence of acute renal failure. Sensitivity analyses excluding trials with a follow-up < 6 months, trials with a cross-over design and those including < 25 patients confirmed the main results.